Will AgNO3 dissolve copper

Emily Dawson
Published Mar 28, 2026
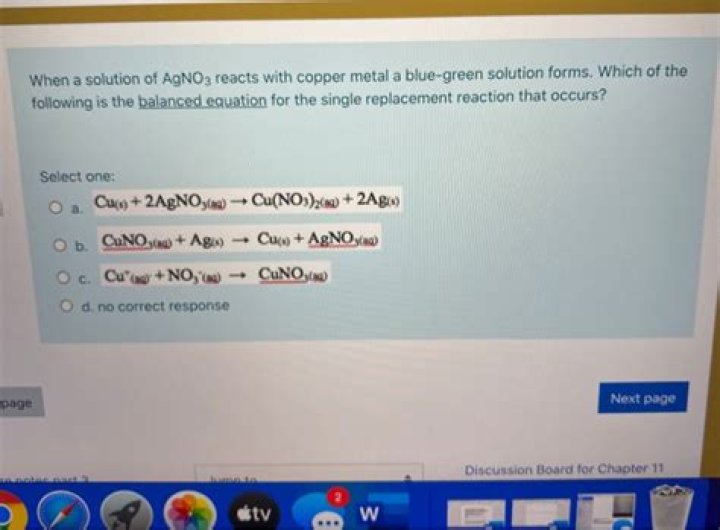
Copper metal dissolves in a silver nitrate solution. The balanced equation for the reaction is: Cu(s) + 2AgNO3(aq) → Cu(NO3)2(aq) + 2Ag(s).
What happens when you mix AgNO3 and CU?
When copper wire is left in an aqueous solution of silver nitrate, precipitates of silver begin to accumulate along the wire. These appear like crystal fractals, and can later be removed, dried and collected.
When Cu reacts with AgNO3 solution what takes place?
reduction of NO3−
What will react with AgNO3?
For example, when an aqueous solution of silver nitrate (AgNO3) is added to the aqueous solution of sodium chloride (NaCl), a white precipitate of silver chloride (AgCl) is formed that is indicated by the following chemical reaction. … Ionic compounds dissociate into ions when dissolved in water (aqueous solution).Is AgNO3 a transition metal?
Properties of Silver Silver is a soft, white, lustrous transition metal. … The metal occurs naturally in its pure, free form (native silver). It also occurs naturally as an alloy with gold and other metals and in minerals such as argentite and chlorargyrite.
Is AgNO3 Cu redox?
Cu + 2AgNO3→ Cu(NO3)2 + 2Ag. The reaction is a redox reaction.
Which will give a white precipitate with AgNO3?
Aryl and vinyl halide’s are not hydrolysed by alcoholic silver nitrate solution. Benzyl chloride gives a ‘white ppt. while ethyl iodide gives yellow ppt with alcoholic silver nitrate solution.
When copper is combined with a solution of silver nitrate the resulting products are copper II nitrate and silver the balanced equation for this reaction is?
The balanced equation for this reaction is: 2AgNO3 (aq) + CuCl2 (s) → 2AgCl(s) + Cu(NO3)2(aq) If 6 moles of silver nitrate react, (a) The reaction consumes _______ moles of copper(II) chloride.What is the products of Cu AgNO3?
When solid copper reacts with aqueous silver nitrate, the products are copper (II) nitrate and solid silver. and carbon monoxide gas produces solid iron and carbon dioxide gas. + __CO 2(g) Page 15 Balancing Chemical Reactions For each of the following, generate a balanced chemical equation.
Is AgNO3 sparingly soluble?AgNO3 is soluble, whereas AgCl is sparingly soluble.
Article first time published onIs kno3 soluble or insoluble?
Potassium nitrate appears as a white to dirty gray crystalline solid. Water soluble.
When copper turning are added to silver nitrate solution a blue Coloured solution is formed after sometime it is because copper?
When copper turnings are added to silver nitrate solution, a blue coloured solution is formed after some time. It is because of copper. oxidises silver from the solution.
Does silver react with silver nitrate?
If you put a silver wire into silver nitrate (which is literally a solid, mp = 210 C), no visible reaction will occur.
What is AgNO3 used for?
Silver nitrate is a natural compound that is used as an antiinfective agent. Silver nitrate topical (for use on the skin) is used to cauterize infected tissues around a skin wound. Silver nitrate can also help create a scab to help stop bleeding from a minor skin wound.
Is AgNO3 conductive?
However, solid silver nitrate, AgNO3, is a good conductor only when melted or dissolved in pure water; as a solid, it is a poor conductor. … This separation of charges accounts for why AgNO3 does conduct electricity in the molten state or in aqueous solutions.
Is cuso4 soluble or insoluble?
Water. Copper sulfate is an inorganic salt that is highly soluble in water. The disassociated copper ions mainly bind to organic matter or remain dissolved in water.
What type of compound is AgNO3?
silver nitrate, caustic chemical compound, important as an antiseptic, in the industrial preparation of other silver salts, and as a reagent in analytical chemistry. Its chemical formula is AgNO3.
What is Ag charge in AgNO3?
Structure of AgNO The nitrogen-oxygen bonds in this ion are similar to each other. The formal charge assigned to the nitrogen atom is -1, whereas each oxygen atom holds a charge of -⅔. The net charge associated by the nitrate ion is -1, which is quenched by the +1 charge held by the Ag+ ion via an ionic bond in AgNO3.
What can silver mix with?
Silver metal will combine with chlorine, bromine, or iodine to form compounds.
Which compound gives white PPT with AgNO3?
In chloroform, the bond between carbon and chlorine is a covalent bond. The compounds in which chlorine is ionically bonded form white precipitate with silver nitrate. Complete step by step solution: We know that chloroform is an organic compound having a molecular formula CHCl3.
Which of the following will not give precipitate with AgNO3?
[Co(NH3)3Cl3] does not give a precipitate with AgNO3 solution because in it all the chlorine atoms are attached by secondary valencies.
Which of the complex produces precipitate with AgNO3?
In the third complex, there are four chloride anions out of which two satisfy the primary valance of the platinum cation, while two are the secondary valance. So this will give curdy white precipitate of silver chloride with silver nitrate.
Does Fe react with cuso4?
Fe + CuSO4(aq) → FeSO4(aq) + Cu(s) The type of reaction is a displacement reaction. In this reaction, the highly reactive metal iron replaces the Copper in copper sulphate by giving up two electrons and gets oxidized and forms a new compound called ferrous sulphate.
When copper II chloride reacts with silver nitrate copper II nitrate and silver chloride are formed write the balanced equation for the reaction given above?
The balanced equation for this reaction is: 2AGNO3 (aq) + CuCl2 (s) → 2AgC1(s) + Cu(NO3)2 (aq) If 4 moles of copper(II) chloride react, The reaction consumes moles of silver nitrate. The reaction produces moles of silver chloride and moles of copper(II) nitrate.
Why is AgNO3 more soluble than Agcl?
In silver nitrate, the NO3− ion is larger and does not allow as close an approach as the chloride ion, so the bond is weaker, easier to break up, and the salt is more soluble.
Is AgNO3 soluble in water at room temperature?
AgNO3 is very soluble in water. This means water molecules, because of their polar nature, can separate the silver ions from the nitrate ions.
Is PO4 soluble?
Phosphates (PO4 3-) and carbonates (CO3 2-) are usually insoluble. Exceptions include salts of the Na+, K+, and NH4 + ions, which are soluble by rule 1.
Is PbI2 insoluble?
Lead iodide appears as a yellow crystalline solid. Insoluble in water and denser than water.