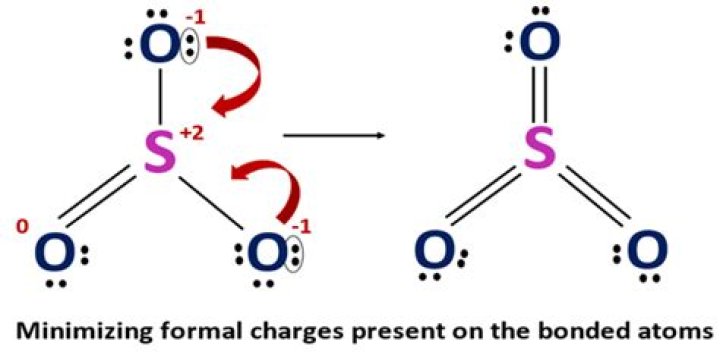
Why the charge of SO3 is 2

Victoria Simmons
Published Apr 22, 2026
Sulfite () is an ion where it has a sulfur atom with oxidation state +4, and 3 oxygen atoms (each being -2) so when these are combined, you get an overall charge of -2. In a more detailed explanation, the sulfite ion has 3 resonance structures.
What is the charge of co3?
The substance with the chemical formula CO3 goes by the name carbonate. Carbonate is made of 1 atom of carbon and 3 atoms of oxygen and has an electric charge of −2. This negative charge means that a single ion of carbonate has 2 more electrons than protons.
What's the charge of phosphate?
Add details and clarify the problem by editing this post. Closed 3 years ago. I am studying chemistry in High School and I am studying my ions(Because I failed too in the beginning, oops) and I have a question on this reasoning: In Phosphate(PO4), it has a charge of -3.
What is the charge of SO2?
The formal charge on the SO2 molecule is zero, but the formal charge on each atom depends on the Lewis structure that you draw.What is SO32?
Sulfite ion, SO32-
What is charge of hydroxide?
We see that hydroxide has a 1- charge, which means the ion has one more electron than there are protons in the nuclei of a hydrogen atom plus an oxygen atom.
What is the charge of N?
NumberElementCharge7nitrogen3-8oxygen2-9fluorine1-10neon0
How do you find the formal charge of sulfur?
Continuing with sulfur, we observe that in (a) the sulfur atom shares one bonding pair and has three lone pairs and has a total of six valence electrons. The formal charge on the sulfur atom is therefore 6 – (6 + 2/2) = 1−.What is the charge of chromate?
Chromate Chemical Formula The chromate ion usually consists of one chromium atom (+6 oxidation state) and four oxide atoms. The overall charge is -2.
What is the charge of no3?For example, NO−3 is the nitrate ion; it has one nitrogen atom and three oxygen atoms and an overall 1− charge.
Article first time published onWhy is the charge of phosphate 3?
So for the whole PO4 group to be stable, the P loses 5 electrons and the 4 Os gain 2 electrons each (total 8 electrons gained). The 5 from the P and another 3 from some other atoms (like hydrogen for example). So the PO4 has an overall 3 electrons more than the protons it has. So it is negatively charged.
What is the charge of hpo3?
In H3PO3 the oxygens will always have a -2 charge and hydrogen is +1. The compound is electrically neutral, so the phosphorus must have an oxidation state of +3.
What is the name of so3 2 negative?
Chemical NameSULFITESReportTRS 952-JECFA69
What is the charge of sulfate?
Charge on Sulfate Sulfate has a 2− charge. In order to understand why this polyatomic ion has a charge, the formal charge of one of the resonance structures can be observed. In the image below, the oxygen atom labeled 1 has 6 valence electrons.
What is the formula for sulfite ion?
Sulfite ion, also written as sulphite, is an ion that is present in many binaries salts largely used in chemical industries. Formula and structure: The sulphite ion chemical formula is SO32-. The molar mass is 80.06 g/mol. It is formed by one centered sulfur cation S4+ and three oxygen anions O2-.
What is the charge of N in N2?
Nitrogen gas has the formula N2. In this state, its charge is zero.
What is the ion for N?
A nitrogen atom must gain three electrons to have the same number of electrons as an atom of the following noble gas, neon. Thus, a nitrogen atom will form an anion with three more electrons than protons and a charge of 3−. The symbol for the ion is N3−, and it is called a nitride ion.
How is nh4 positive?
⇒ The ammonium ion, NH+4 , becomes positive . A covalent bond is formed by two atoms sharing a pair of electrons between the N and the 4th-H forming a Co-ordinate (dative covalent) bond . The hydrogen’s electron is left behind on the chlorine to form a negative chloride ion.
Is OH and HO the same?
HO is a molecule containing positive Hydrogen Oxygen atoms i.e it is a cation. … OH is a molecule containing negative Hydrogen Oxygen atoms i.e it is an anion. Both are ions, HO+ is Hydroxide, OH- is Oxyhydride. Both are acceptable and the only difference is about the charge on oxygen.
What is the charge of ammonium?
Ammonium ion has a formal charge of +1 and amide anion has a formal charge of -1. Carbon has four valence electrons and the Lewis structures of methane, methyl cation, methyl anion, and methyl radical are shown below.
Why does Na ion have a +1 charge?
A sodium atom has one electron in its outer shell. … A sodium atom can lose its outer electron. It will still have 11 positive protons but only 10 negative electrons. So, the overall charge is +1.
What ion is no3?
Nitrate is a polyatomic ion with the chemical formula NO−3. Salts containing this ion are called nitrates.
What is the name of cro2 4?
PubChem CID73415796Molecular FormulaCrO2SynonymsChromium(IV) oxide chromium (IV) oxide UNII-7BHJ7466GL 7BHJ7466GL Q2366389Molecular Weight83.995Component CompoundsCID 23976 (Chromium) CID 190217 (Oxide)
How do you find the charge of dichromate?
Dichromate anion consists of seven oxygen atoms and oxidation state of oxygen is -2. Multiplying seven with -2 we get -14. Adding the total oxidation state of chromium and oxygen, we get -2. So, the ionic charge on dichromate ions is -2.
Why do sulfite and sulfate have the same charge?
Sulfite is an anion composed of sulfur and oxygen atoms. Sulfite anion has one sulfur atom bonded to three oxygen atoms. The charge of Sulfite anion is -2. In sulfite anion, one oxygen atom is bonded to sulfur through a double bond and other two oxygen atom are bonded to sulfur atom through single bonds.
What is the formal charge on Sulphur in so2?
Hence, no electron is gained and no electron is lost. So, neither negative nor positive charge is present on the S atom. Hence, the formal charge on $S$ atom in $S{O_2}$ is zero.
What is the charge of zinc?
As zinc is a metal, it generally forms metallic compounds with other metals. Because it has an ion charge of +2, zinc ions are strong reducing agents and readily form ionic bonds.
What is the charge of N in NO3 1?
B = Number of electrons in a covalent bond. -For Nitrogen: From the structure itself, we find a positive charge on Nitrogen, hence it carries a formal charge of +1.
Is phosphate negative or positive?
The phosphate backbone of DNA is negatively charged due to the bonds created between the phosphorous atoms and the oxygen atoms. Each phosphate group contains one negatively charged oxygen atom, therefore the entire strand of DNA is negatively charged due to repeated phosphate groups.
Why does phosphate have 4 Oxygens?
In the phosphate ion we have a central Phosphorus atom, with five valence electrons. This is bonded to four oxygen atoms, which have six valence electrons. Five P electrons plus 4 times 6 O electrons gives 29 electrons.
What is the ionic charge of sulfide?
The net charge on a sulfide ion, S2, is 2.



