Why is sodium sulfide Na2S

Victoria Simmons
Published Mar 27, 2026
NamesChemical formulaNa2SMolar mass78.0452 g/mol (anhydrous) 240.18 g/mol (nonahydrate)Appearancecolorless, hygroscopic solidOdornone
How do you identify Na2S?
Sodium sulfide, anhydrous is a yellow to brick red crystalline mass or fused solid with an odor of rotten eggs. If exposed to moist air it is liable to spontaneous heating and may cause ignition of nearby combustible material.
What is sodium sulfide?
Sodium sulfide is a chemical compound with a chemical formula Na2S. Sodium sulfide is used in the pulp and paper industry, water treatment, textile industry, and various chemical manufacturing processes including the production of rubber chemicals, sulfur dyes and oil recovery.
What ions make up Na2S?
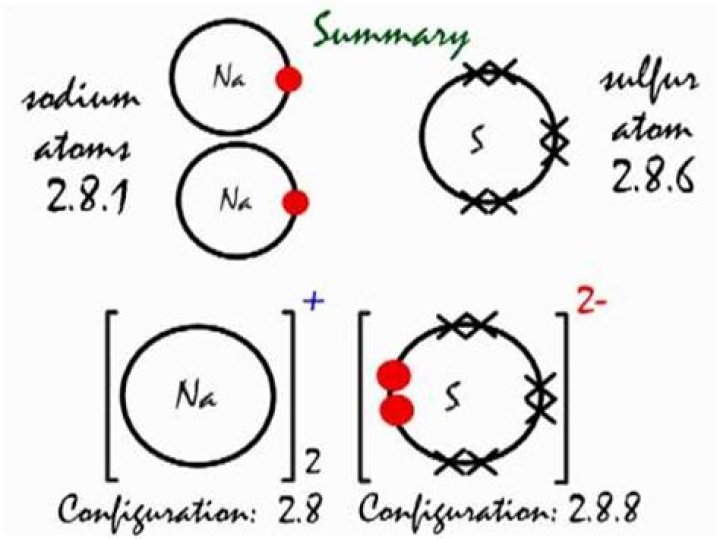
2. Combine these two ions so that the total charge is zero. In this case, it take two 1+ charges per one 2- charge. Thus the formula for sodium sulfide is Na2S.What is the difference between sodium sulfide and sodium sulfate?
The key difference between sodium sulphate and sodium sulphite is that the sodium sulphate has a sulphate anion, which has one sulfur atom and four oxygen atoms whereas sodium sulphite has a sulphite anion, which has one sulfur atom and three oxygen atoms.
Why is Na2S ionic?
Sodium sulfide compound is an ionic compound. There are 2 Na atoms per 1 Sulfide atom. It has a central Sulfur atom encircled by 4 Oxygens in covalent bonds. The Sodium atoms and Sulfur or Oxygen atoms in the compound exchange their electrons.
How many atoms does Na2S?
There are 7 atoms per molecule, 2 Na, 1 S, 4 O.
Why does sodium sulfide have a high melting point?
It takes a lot of energy to overcome the strong electrostatic forces of attraction between oppositely charged ions, so ionic compounds have high melting and boiling points.Why do you need two sodium ions for every sulfide ion in sodium sulfide?
Sulfur, a nonmetal, has a tendency to gain 2 electrons. This results in an ion with a negative 2 charge. Nonmetal ions end in “ide”. To obtain a neutral charge, you need two sodium ions, which provides you with a plus 2 charge balance the negative 2 charge of sulfur.
Why is sodium sulfide important?Sodium sulfide is used in the pulp and paper industry, water treatment, textile industry, and various chemical manufacturing processes including the production of rubber chemicals, sulfur dyes and oil recovery. Along with its hydrate form, sodium sulfide releases hydrogen sulfide (H2S) when in contact with moist air.
Article first time published onIs sodium sulfide harmful?
Corrosive, toxic. Sodium sulfide is a strong base and can cause severe burns on the mucous membrane. Hydrolysis by gastric fluids releases toxic hydrogen sulfide.
What is sulphide used for?
Sodium sulfide (Na2S) is an important industrial chemical, used in manufacture of kraft paper, dyes, leather tanning, crude petroleum processing, treatment of heavy metal pollution, and others.
Is sulphate and sulphide the same justify?
Sulphur exists in nature in the form of sulphate, sulphite, sulphide compounds. … – The oxidation state of sulphur in sulphide is ‘-2’. – The main difference between sulphite, sulphate, and sulphide is the number of oxygen atoms and the oxidation state of the sulphur present in the respective derivatives.
What is the purpose of using anhydrous sodium sulfate in this experiment?
Swirl the contents of the flask. The anhydrous sodium sulfate will absorb the small amount of water that is dissolved in the dichloromethane and small amounts of water from the aqueous layer that may have gotten into the flask by accident.
What is the difference between sulfide and sulfite?
Being an ion, sulfite, like sulfide, has a negative charge. However, the distinguishing factor between sulfide and sulfite is their molecular make ups. Besides one sulfur atom, sulfites have three oxygen atoms. This addition causes the creation of bonds in the ion, another feature sulfide ions do not have.
What is the percent composition of sodium in sodium sulfide na2s?
ElementSymbolMass PercentNatriumNa58.915%SulfurS41.086%
What is the chemical name for na2s?
ChEBI Namesodium sulfide (anhydrous)DefinitionA sulfide salt with formula Na2S. The pentahydrate and (particularly) the nonahydrate are also known. In gel form, sodium sulfide is used to soften toenails to assist in trimming (and so relive pain) of ingrowing toenails.
What is the correct name for BaO?
PubChem CID62392Chemical SafetyLaboratory Chemical Safety Summary (LCSS) DatasheetMolecular FormulaBaOSynonymsBARIUM OXIDE 1304-28-5 oxobarium Baryta Barium oxide (BaO) More…Molecular Weight153.33
Is Na2S binary or polyatomic?
ABsodium iodideNaIsodium oxideNa2Osodium sulfideNa2Ssodium nitrideNa3N
Is FCl ionic or covalent?
FCl: This compound is composed of F (a nonmetal) and Cl (a nonmetal) connected by a covalent bond.
What is the solubility of Na2S?
NamesSolubility in water12.4 g/100 mL (0 °C) 18.6 g/100 mL (20 °C) 39 g/100 mL (50 °C) (hydrolyses)Solubilityinsoluble in ether slightly soluble in alcoholMagnetic susceptibility (χ)−39.0·10−6 cm3/molStructure
Is sodium sulfide covalent ionic metallic or molecular?
Sodium exhibits metallic bonding; i.e. positive ions in an electron sea, which gives rise to demonstrable malleability and ductility. … And sodium sulfide is also non-molecular, ionic solid composed of individual 2×Na+ and S2− ions held together by strong electrostatic interaction between the positive and negative ions.
In what ratio will sodium ions and sulfide ions combine to give sodium sulfide quizlet?
The compound sodium sulfide consists of a ratio of two sodium ions (Na+) to one sulfide ion (S2−). Choose the chemical formula for this compound. C12H22O11 is the molecular formula for _____. Choose the formula for acetic acid.
Why do you need a zinc ion and two fluoride ions for the formula for zinc fluoride?
Both the metal and the nonmetal form ions, which is why it is called an ionic compound. … 1- Why do you need one zinc ion and two fluoride ions for the formula for zinc fluoride? to balance Charge Why do you need two sodium ions for every sulfide ion in sodium sulfide? Sodium is 1+ 3S is at : need to balance charge.
Is FeCl2 a Type 1 binary compound?
A binary ionic compound is composed of ions of two different elements – one of which is a metal, and the other a nonmetal. … Examples of type 1 binary ionic compounds are as FeCl2 and FeCl3, AgCl and AgCl2.
Why does hydrogen sulfide have a low melting point?
In short, because the intermolecular forces holding the molecules together in a rigid, solid structure are much weaker in hydrogen sulfide than in water. The weaker the forces, the less energy is required to break them and the lower the melting point.
Why sodium sulfide does not conduct electricity as a solid but does when melted?
Ionic bonding Solid ionic compounds do not conduct electricity because the ions are held firmly in place. The ions cannot move to conduct the electric current . But when an ionic compound melts, the charged ions are free to move. Therefore, molten ionic compounds do conduct electricity.
What is sodium sulfate used for?
Sodium sulfate is mainly used for the manufacture of detergents and in the Kraft process of paper pulping, although it has many other uses. About half of the world’s production is from the natural mineral form of the decahydrate (mirabilite), and half from by-products of chemical processes.
Is sodium sulfide an antibiotic?
This medication is used to treat several skin problems (acne, acne rosacea, seborrheic dermatitis). Sodium sulfacetamide works by stopping the growth of certain bacteria on the skin that can lead to acne and worsen these other skin conditions. It belongs to a class of drugs known as sulfa antibiotics.
Is sodium sulfide a reducing agent?
Sodium sulfide is the largest consumed reducing agent; around 90% of all sulfur dyes are being reduced by sodium sulfide has adverse effect on environment3, 11.
Can sodium sulfide explode?
SODIUM SULFIDE is a white to yellow crystalline material, flammable. Can explode on rapid heating or when shocked. Violent reaction with carbon, charcoal, diazonium salts, N,N-dichloromethylamine, strong oxidizers, water. On contact with acids it liberates highly toxic and flammable hydrogen sulfide gas.