Why is alkalosis dangerous

Victoria Simmons
Published Mar 27, 2026
Alkalosis is a common finding in hospitalized patients but is rarely life-threatening in itself. While the body’s buffering systems initially correct alkalosis, ongoing alkalosis may cause clinical problems due to associated electrolyte disturbances.
Is alkalosis as serious as acidosis?
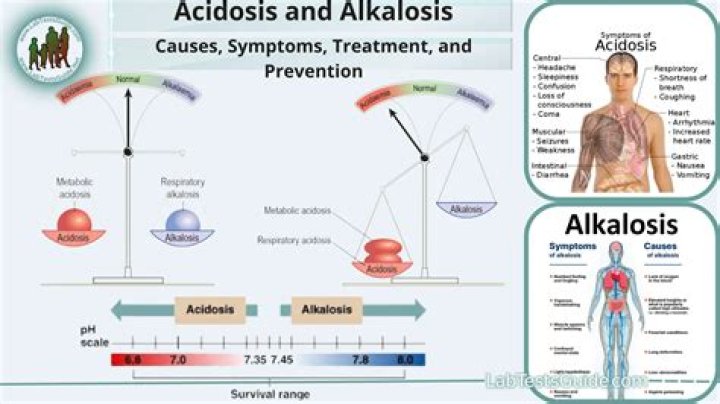
The pH of your blood should be around 7.4. According to the American Association for Clinical Chemistry (AACC), acidosis is characterized by a pH of 7.35 or lower. Alkalosis is characterized by a pH level of 7.45 or higher. While seemingly slight, these numerical differences can be serious.
What happens if body is too alkaline?
An increase in alkaline causes pH levels to rise. When the levels of acid in your blood are too high, it’s called acidosis. When your blood is too alkaline, it is called alkalosis. Respiratory acidosis and alkalosis are due to a problem with the lungs.
How does alkalosis affect the heart?
Respiratory alkalosis resulted in an increased sympathetic influence on the heart activity whereas the shape and duration of the atrial and the ventricular MAPs remained unaffected.When is metabolic alkalosis dangerous?
Metabolic alkalosis is a commonly seen imbalance in the intensive care unit (ICU). Extreme metabolic alkalemia, however, is less common. A pH greater than 7.65 may carry a high risk of mortality (up to 80%).
Does alkalosis cause hypokalemia?
Alkalosis has the opposite effects, often leading to hypokalemia. Potassium disorders also influence acid-base homeostasis. Potassium depletion causes increased H(+) secretion, ammoniagenesis and H-K-ATPase activity.
What are the complications of metabolic acidosis?
- osteoporosis, which is a loss of bone that can increase the risk of fractures.
- improper growth in children, as metabolic acidosis restricts the growth hormone.
- increased kidney damage, which can worsen chronic kidney disease.
- muscle loss or wasting.
Does alkalosis increase heart rate?
Effects on pulse rate were significant increases in the alkalosis and hypercarbia groups, decrease in the acidosis group, while in other conditions no significant changes were recorded.Can alkalosis cause heart failure?
Metabolic alkalosis is a common complication of congestive heart failure (CHF) and its treatment with diuretics. When severe, the alkalosis can have significant adverse effects on cellular function, as shown in Table 1, and contribute to increased mortality.
What happens respiratory alkalosis?Respiratory alkalosis occurs when high levels of carbon dioxide disrupt the blood’s acid-base balance. It often occurs in people who experience rapid, uncontrollable breathing (hyperventilation). Treatment includes supplemental oxygen and therapies to reduce the risk of hyperventilation.
Article first time published onIs it better to be alkaline or acidic?
Science says alkaline foods may be better for you than acidic ones, but not for the reasons you think. According to the alkaline diet, you can lose weight, have more energy, and ward off disease—all by simply eating the “right” foods based on their level of acidity.
How does pH affect the human body?
Water with a pH of between 7.2 and 7.8 is ideal for maintaining good health. When we drink liquids that are too acidic or too alkaline, it can disrupt the body’s delicate balance, which can lead to the development of bacteria, viruses, fungi, yeasts and parasites.
What can combat alkalosis?
Metabolic alkalosis is corrected with the aldosterone antagonist spironolactone or with other potassium-sparing diuretics (eg, amiloride, triamterene). If the cause of primary hyperaldosteronism is an adrenal adenoma or carcinoma, surgical removal of the tumor should correct the alkalosis.
Does hyperkalemia cause acidosis or alkalosis?
In hypokalemia, an intracellular acidosis can develop; in hyperkalemia, an intracellular alkalosis can develop.
Why do Diuretics cause alkalosis?
Severe metabolic alkalosis is much less frequent and, when it occurs, it is in association with loop diuretic use. The generation of a metabolic alkalosis with diuretic therapy is primarily due to contraction of the extracellular fluid space caused by urinary losses of a relatively HCO3 -free fluid.
What conditions cause metabolic alkalosis?
- Loss of stomach acids. This is the most common cause of metabolic alkalosis. …
- Excess of antacids. …
- Diuretics. …
- Potassium deficiency (hypokalemia). …
- Reduced volume of blood in the arteries (EABV). …
- Heart, kidney, or liver failure. …
- Genetic causes.
How does acidosis cause death?
Symptoms of Acidosis Eventually, in severe cases, heart problems may develop and blood pressure can fall, leading to shock, coma, and death. Drowsiness may progress to stupor and coma as the oxygen in the blood becomes inadequate.
Is metabolic acidosis serious?
Metabolic acidosis is a serious electrolyte disorder characterized by an imbalance in the body’s acid-base balance. Metabolic acidosis has three main root causes: increased acid production, loss of bicarbonate, and a reduced ability of the kidneys to excrete excess acids.
What happens if metabolic acidosis goes untreated?
This can cause your body to build a resistance to insulin (the hormone in your body that helps keep your blood sugar level from getting too high or too low). If left untreated for too long or not corrected in time, it can lead to diabetes.
Why does alkalosis increase potassium secretion?
Moreover, in the case of acute metabolic alkalosis, there is inhibition of fractional NaHCO3 and fluid reabsorption in the proximal tubule, leading to increased distal delivery of Na+ and HCO3− and enhanced fluid flow. As already discussed, increased Na+ delivery and fluid flow stimulate K+ secretion.
How does Hypochloremia cause metabolic alkalosis?
Hypochloremia can contribute to the maintenance of metabolic alkalosis by increasing the reabsorption of and reducing the secretion of bicarbonate in the distal tubule. Increased distal reabsorption of bicarbonate.
Why does hyperkalemia lead to acidosis?
Conclusions Hyperkalemia decreases proximal tubule ammonia generation and collecting duct ammonia transport, leading to impaired ammonia excretion that causes metabolic acidosis.
Why does CHF cause alkalosis?
Disturbance in acid-base balance is commonly observed in patients with heart failure. The most common disturbance is metabolic alkalosis combined with hypokalemia, as a result of the excessive use of loop diuretics. Occasionary, hypoxia due to pulmonary edema stimulates ventilation, resulting in respiratory alkalosis.
How does high pH affect heart rate?
This analysis revealed that changes in physiological range of potassium, calcium, and pH could cause large heart rate variations from 60 to 90 bpm.
What happens during respiratory acidosis?
Respiratory acidosis is a condition that occurs when the lungs cannot remove all of the carbon dioxide the body produces. This causes body fluids, especially the blood, to become too acidic.
How is respiratory alkalosis treated?
- Breathe into a paper bag. Fill the paper bag with carbon dioxide by exhaling into it. …
- Get reassurance. The symptoms of respiratory alkalosis can be frightening. …
- Restrict oxygen intake into the lungs. To do this, try breathing while pursing the lips or breathing through one nostril.
Can metabolic acidosis cause coma?
You treat metabolic acidosis by treating what’s causing it. If you don’t restore the balance, it can affect your bones, muscles, and kidneys. In severe cases, it can cause shock or death. DKA can put you in a coma.
Who is at risk for respiratory alkalosis?
Breathing too fast can cause a person to go into respiratory alkalosis. This occurs when a person’s pH level is higher than 7.45. A person may breathe too fast due to anxiety, overdosing on certain medications, or using a ventilator.
Is pneumonia respiratory acidosis or alkalosis?
Respiratory alkalosis is commonly found in patients with asthma, pneumonia & pulmonary embolism.
Why does pulmonary embolism cause respiratory alkalosis?
Thus, most patients with PE present with a lower than normal arterial PCO2 and respiratory alkalosis because of an increased total minute ventilation. Limited data suggest that the increased total minute ventilation occurs because of reflex stimulation of irritant and juxta capillary sensors in the lung.
Are lemons acidic or alkaline?
Because they contain a high amount of citric acid, lemons have an acidic pH. Lemon juice has a pH between 2 and 3, which means it’s 10,000–100,000 times more acidic than water (1, 2, 3). A food’s pH is a measure of its acidity. The pH of lemon juice falls between 2 and 3, meaning it is acidic.