Why does ionic size change

William Taylor
Published Apr 25, 2026
the ionic radius decreases for metals forming cations, as the metals lose their outer electrons. The ionic radius increases for nonmetals as the effective nuclear charge decreases due to the number of electrons exceeding the number of protons.
How do you determine ionic size?
Ionic radius is determined by measuring the atom in a crystal lattice. Removal of electrons results in an ion that is smaller than the parent element. Addition of electrons results in an ion that is larger than the parent atom.
What does ionic size depend on?
The size of an atom or ion depends on the size of the nucleus and the number of electrons. Generally atoms with higher numbers of electrons have larger radii than those with smaller numbers of electrons. Thus ions will have radii different from the atoms because ions will have either gained or lost electrons.
How is the ionic size trend different from the atomic size trend?
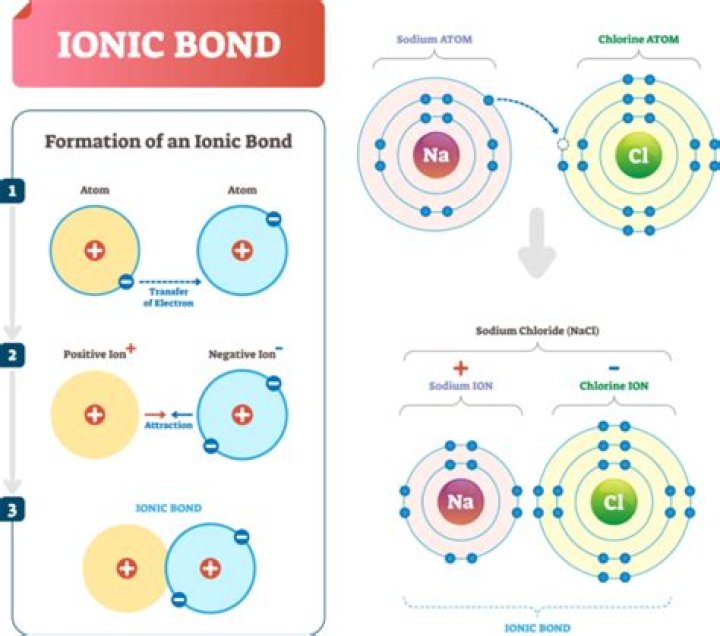
In a neutral atom, the atomic and ionic radius are the same, but many elements exist as anions or cations. If the atom loses its outermost electron (positively charged or cation), the ionic radius is smaller than the atomic radius because the atom loses an electron energy shell.Why does ionic size decrease across a period?
This is because each row adds a new electron shell. Ionic radius decreases moving from left to right across a row or period. More protons are added, but the outer valence shell remains the same, so the positively charged nucleus draws in the electrons more tightly.
What is ionic size in chemistry?
The ionic size is when the atom loses or gains electrons to become negatively charged (anions) or positively charged (cations) ions. When atoms lose or gain electrons, the size of the ion is not the same as the original atom.
How does the size change when an atom forms a cation?
When an atom loses an electron to form a cation, the lost electron no longer contributes to shielding the other electrons from the charge of the nucleus; consequently, the other electrons are more strongly attracted to the nucleus, and the radius of the atom gets smaller.
Which has largest ionic size?
IonRadius (pm)Atomic NumberN3−1467O2−1408F−1339Na+9811How do you arrange ion sizes?
In such a series, size decreases as the nuclear charge (atomic number) of the ion increases. The atomic numbers of the ions are S (16), Cl (17), K (19), and Ca (20). Thus, the ions decrease in size in the order: S2- > Cl– > K+ > Ca2+.
What is atomic size and ionic size?Atomic and ionic radii are distances away from the nucleus or central atom that have different periodic trends. Atomic is the distance away from the nucleus. Atomic radius increases going from top to bottom and decreases going across the periodic table. Ionic radius is the distance away from the central atom.
Article first time published onWhy the size of cation is smaller than parent atom?
This is because the number of protons now exceeds the number of electrons. As a result, there is a net charge, and the atom is no longer neutral. As a result of this electron loss when creating a cation, the cation is smaller in size than its parent atom.
Why are anions larger than cations?
But an anion gains electron attaining a negative charge. This negative charge gives an extra strength to the inner most electrons to shield themselves from the attraction of nucleus and thus anion is bigger in size than cation.
Why are anions larger?
In general, anions are larger than the corresponding neutral atom, since adding electrons increases the number of electron-electron repulsion interactions that take place. Cations are smaller than the corresponding neutral atoms, since the valence electrons, which are furthest away from the nucleus, are lost.
Why are the trends and expectations to the trends in ionization energy observed Check all that apply?
Why are the trends and exceptions to the trends in ionization energy observed? … Ionization energy tends to increase across a period because electrons are added to the same main energy level Ionization energy tends to increase across a period because the nuclear charge increases.
What are the trends in ionization energy?
Ionization energy exhibits periodicity on the periodic table. The general trend is for ionization energy to increase moving from left to right across an element period. Moving left to right across a period, atomic radius decreases, so electrons are more attracted to the (closer) nucleus.
What is the trend going across a period in the periodic table?
Elements in the same period have the same number of electron shells; moving across a period (so progressing from group to group), elements gain electrons and protons and become less metallic. This arrangement reflects the periodic recurrence of similar properties as the atomic number increases.
Why are positive ions smaller than neutral atoms?
This is because it gains one or more electrons than it’s neutral atom to become stable and thus its size increases. A positive ion on the other hand becomes smaller. A cation looses one or more electrons than it’s neutral atoms and thus is smaller in size.
When an atom because an anion its size changes explain how and why its size changes?
An anion has a larger radius than the neutral atom because it gains valence electrons. There are added electron/electron repulsions in the valence shell that expand the size of the electron cloud, which results in a larger radius for the anion.
How does the size change when an atom forms a cation and when an atom forms a anion?
When a neutral atom gains or loses an electron, creating an anion or cation, the atom’s radius increases or decreases, respectively.
What is ionic radius Class 10?
Ionic radius is defined as the distance of the valence shell of electrons from the centre of the nucleus in an ion. Concept: Periodic Trends in Elemental Properties.
What is a cation chemistry?
cation, atom or group of atoms that bears a positive electric charge.
What is the size of H+ ion?
In LiH, the less electronegative lithium gives its electron to hydrogen, and the H- ion, with a filled first-electron shell, has an ionic radius of 1.54 A.
What is atomic size order?
The atomic size or atomic radius is of the order of 10−7cm or 10−9m or 1 nanometer(nm).
Is a negative ion larger or smaller?
The Relative Size of Atoms and Their Ions In each case, the negative ion is much larger than the atom from which it was formed. In fact, the negative ion can be more than twice as large as the neutral atom. The only difference between an atom and its ions is the number of electrons that surround the nucleus.
Are all ions the same size in ionic solids?
IonRadius (pm)Atomic NumberN3−1467O2−1408F−1339Na+9811
What is the smallest ion?
More the nuclear charge on ion, electrons are more strongly attracted and thus size decreases. Hence, among the given ions Al3+ is the smallest ion.
Which of the following has smallest ionic size?
Na+,Mg2+,O2− and F− all are isoelectronic but Mg2+ has 12 protons in his nucleus, so the attraction force on the last shell is maximum and hence, it has the smallest size.
Which atom is the largest?
Cesium (Cs), tucked in the lower left hand corner of the table, has the largest known atoms. The atomic radius of Cs is given variously as 273.1 pm [1], 265 pm [2], 265.5 pm [3] or 260 pm [4].
What is covalent and ionic radii?
Measurement of Radius Nonpolar atomic radii: The radius of an atom is derived from the bond lengths within nonpolar molecules; one-half the distance between the nuclei of two atoms within a covalent bond. … Crystal Radii: The atomic or ionic radius is determined using electron density maps fro X-ray data.
What is the trend between atomic radius and ionic radius for cations positively charged atoms )? Why?
Ionic radii follow the same vertical trend as atomic radii; that is, for ions with the same charge, the ionic radius increases going down a column. The reason is the same as for atomic radii: shielding by filled inner shells produces little change in the effective nuclear charge felt by the outermost electrons.
What is difference between atomic size and atomic radius?
What is Atomic Size? Atomic size is the distance between the centre of the nucleus of an atom and its outermost shell. In basic chemistry, the atomic radius is defined as the shortest distance between the atom’s nuclei and the outermost shell of the atom.



