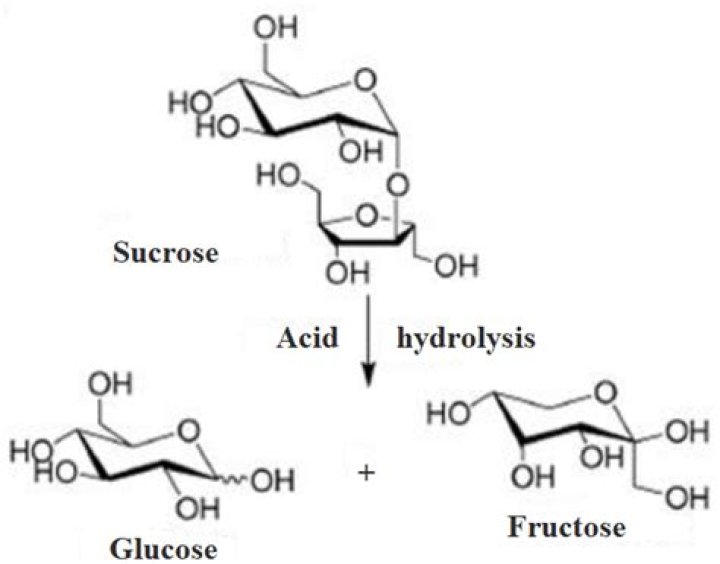
Which of the following is a product of the hydrolysis of sucrose

Robert Spencer
Published Apr 13, 2026
Therefore, the answer is – the products of hydrolysis of sucrose are glucose and fructose.
What is the product of hydrolysis of the following sugar?
Name of the sugarHydrolysis ProductD. Raffinoseiv. Glucose + Glucose
What is sucrose hydrolysis?
In molecular terms, hydrolysis of sucrose involves (a) breaking of a covalent bond in sucrose and hence formation of fructose and glucose, (b) a change in the number of hydrogen bonds between fructose and H2O and between glucose and H2O, and (c) a change in the hydrogen bond equilibrium between H2O and HCl and between …
What is the equation of hydrolysis of sucrose?
The rate equation for the hydrolysis of sucrose to fructose and glucose, C12H22O11(aq)+H2O(l)→2C6H12O6(aq), C 12 H 22 O 11 ( a q ) + H 2 O ( l ) → 2 C 6 H 12 O 6 ( a q ) , is [sucrose]/t = k[C12H22O11] k [ C 12 H 22 O 11 ] .What happens in hydrolysis of sucrose?
When sucrose is hydrolyzed it forms a 1:1 mixture of glucose and fructose. … It is called invert sugar because the angle of the specific rotation of the plain polarized light changes from a positive to a negative value due to the presence of the optical isomers of the mixture of glucose and fructose sugars.
What are the products of hydrolysis reaction?
A hydrolysis reaction is a reaction in which one molecule breaks apart to form multiple smaller molecules. Acidic hydrolysis of an ester gives a carboxylic acid and an alcohol. Basic hydrolysis (saponification) of an ester gives a carboxylate salt and an alcohol.
How does hydrolysis of sucrose change the measurement?
On hydrolysis, the enthalpy decreases by 14.4 kJ/mol of sucrose at 310 K, and the heat capacity, Cp, increases by 61 J mol-1 K-1 of sucrose in the solution. … Cp of the solution increases with temperature less rapidly before sucrose hydrolysis than after it.
What is an example of a hydrolysis reaction?
An example of a hydrolysis reaction is the manufacturing of soap. To obtain soap, a triglyceride is hydrolysed. Fatty acids and glycerin are produced during the reaction. Glycerin then reacts with fatty acids and turn them to a specific type of salt which is known as soap.What causes hydrolysis of sucrose?
The enzyme known as invertase (E.C. 3.2. 1.26 – β-D-fructofuranosidase) catalyzes the sucrose hydrolysis producing an equimolar mixture of glucose and fructose named inverted sugar. … Temperature of 70ºC , pH 6 and agitation of 170 rpm were the established conditions for the hydrolysis process.
What are the types of hydrolysis?2 There are three main types of hydrolysis: salt, acid, and base hydrolysis. Hydrolysis can also be thought of as the exact opposite reaction to condensation, which is the process whereby two molecules combine to form one larger molecule.
Article first time published onCan sucrose be hydrolyzed?
Hydrolysis. Hydrolysis breaks the glycosidic bond converting sucrose into glucose and fructose. Hydrolysis is, however, so slow that solutions of sucrose can sit for years with negligible change. If the enzyme sucrase is added, however, the reaction will proceed rapidly.
What are the final hydrolysis products of carbohydrates?
The final product of the digestion of carbohydrates are monosaccharides such as glucose and fructose. Glucose is used by your cells during respiration to produce energy. Any excess glucose that is not immediately required for cell respiration will be stored.
What does hydrolysis of sucrose yields quizlet?
On hydrolysis, sucrose will yield one molecule of glucose and one molecule of fructose.
What is the hydrolysis reaction?
Thus, a hydrolysis reaction is the cleavage of chemical bonds by the addition of water or a base that supplies the hydroxyl ion ( OH−). A chemical bond is cleaved, and two new bonds are formed, each one having either the hydrogen component (H) or the hydroxyl component (OH) of the water molecule.
Which of the following is an example of hydrolysis quizlet?
hydrolysis is the chemical reaction that breaks a covalent bond by means of the addition of hydrogen to the atom on one side of the original bond and a hydroxyl group to the atom on the other side. water is added in this reaction. an example of hydrolysis is sucrose is separated into glucose and fructose.
What are the three types of hydrolysis?
‘ There are three types of hydrolysis reactions: salt, acid, and base reactions. A salt hydrolysis involves the reaction between organic compounds and water. Acid and base hydrolysis involve the use of water as a catalyst to drive the hydrolysis reaction.
What is required for hydrolysis?
Hydrolysis usually requires the use of an acid or base catalyst and is used in the synthesis of many useful compounds. The term “hydrolysis” literally means to split with water; the inverse process, when water is formed in a reaction, is called condensation.
Which substances are produced by the hydrolysis of an oil?
Hydrolysis can break down a fat or oil and release the triglycerol and fatty acids. The acids can be separated and identified and this information can be used to identify the original fat or oil.
What is inversion of sucrose?
The inversion of sucrose is a reaction that takes place between a molecule of. water and a molecule of the sugar, and proceeds at a noticeable rate only in the. presence of a catalyst (H + ions or invertase).
What is lipid hydrolysis?
The degradation of lipids such as triglycerides is accomplished by extracellular hydrolyzing enzymes, called lipases (esterases), that cleave the ester bonds in this molecule by the addition of water to form the building blocks glycerol (an alcohol) and fatty acids.
What is hydrolysis of sugar?
Hydrolysis is the process of cleavage of molecules with the help of water molecules. It can be carried out in an acidic or basic medium. Hydrolysis of sugars breaks them into smaller molecules by cleaving the glycosidic bonds that hold the monomer units together.
Which of the following best describes the hydrolysis of carbohydrates?
3. Which of the following best describes the hydrolysis of carbohydrates? The addition of a water molecule breaks a bond between sugar monomers.
Which of the following is a characteristic of the sugar alcohols?
Characteristics of Sugar Alcohols They are not as sweet as general sugars and do not contain as many calories as sugars. Their metabolism is not possible by oral bacteria. They do not turn brown in color on heating like general sugars. Dissolution of sugar alcohols is an endothermic reaction.
Which enzyme is used in enzymatic cholesterol determinations to form hydrogen peroxide?
One unit of cholesterol oxidase activity is defined as the amount of enzyme which catalyzes the formation of 1μmol hydrogen peroxide/min at 37°C.
Which of the following defines the term glycolysis quizlet?
What is glycolysis? Glycolysis is a process by which the cell produces ATP, pyruvate, and NADH from sugars.



