Where are beta turns found

Rachel Hickman
Published Mar 23, 2026
β-Turns are the most abundant structures in the loop segment with a preference of types I′ and II′. β-Hairpins are a key motif in antiparallel β-sheets,227 and they are widely found in a variety of proteins including IgG antibodies,228 cytokine receptors,229 growth factors,230 integrins,231 and viral proteins.
Where do beta turns occur in proteins?
In proteins in general all four beta turn types occur frequently but I is commonest, followed by II, I’ and II’ in that order. Beta turns are especially common at the loop ends of beta hairpins; they have a different distribution of types from the others; type I’ is commonest, followed by types II’, I and II.
What causes beta turns?
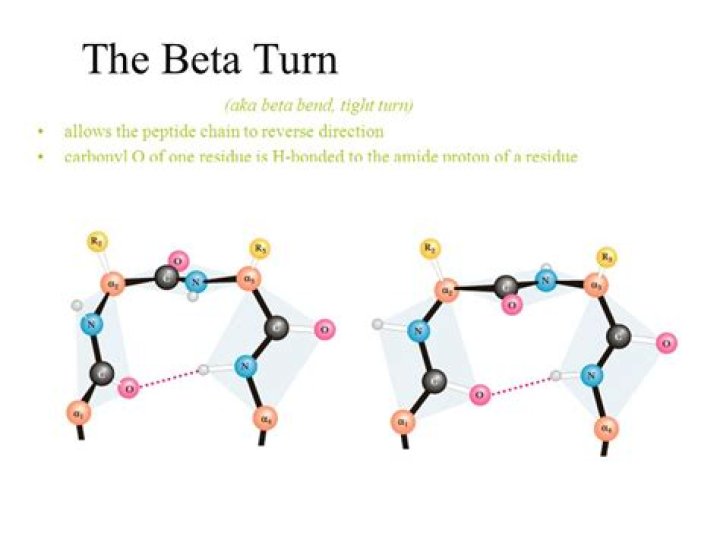
The Beta Turn Turns generally occur when the protein chain needs to change direction in order to connect two other elements of secondary structure. The most common is the beta turn, in which the change of direction is executed in the space of four residues.
Which amino acids are found in beta turns?
Explanation: Glycine and proline are the two amino acids that are found in beta turns. These 180 degree turns are composed of four total amino acids.What is a Type 1 beta turn?
A Type I Turn: Such a structure is known as the Reverse turn or the beta turn (because it is found joining adjacent antiparallel sequences of beta sheet). … Although various amino acids can make up the turn, frequently AA-2 is a proline since it does introduce a sharp bend in the polypeptide chain.
Where might bends or β turns occur?
(a) Bends or turns are most likely to occur at residues 7 and 19 because Pro residues are often (but not always) found at bends in globular folded proteins. A bend may also occur at the Thr residue (residue 4) and, assuming that this is a portion of a larger polypep- tide, at the Ile residue (residue 1).
How do you find the beta turn?
A β-turn is defined by four consecutive amino acids and has a characteristic of the distance between two α-carbons of residues i and i + 3 being less than 7.0 Å (Fig. 26).
Why are glycine and proline found in beta turns?
Proline and glycine residues are statistically preferred at several β-turn positions, presumably because their unique side chains contribute favorably to conformational stability in certain β-turn positions.Do beta turns contain proline?
Proline-containing beta-turns in peptides and proteins: analysis of structural data on globular proteins. Arch Biochem Biophys.
Why is proline found in turns?Proline is unique in that it is the only amino acid where the side chain is connected to the protein backbone twice, forming a five-membered nitrogen-containing ring. … For this reason, Proline can often be found in very tight turns in protein structures (i.e. where the polypeptide chain must change direction).
Article first time published onWhy is glycine found in turns?
Glycine – has no Beta carbon atom, i.e. no side chain. Therefore it is the least sterically hindered as compared to other amino acids. … This conformational flexibility is why you’ll find glycine residues in loop regions of the protein structure, where the polypeptide chain has to make sharp turns.
Which protein is rich in proline?
The proline-rich proteins (PRPs) are a large family of salivary proteins produced by the parotid and submandibular glands and constitute nearly 70% of the total protein of human saliva. Proline accounts for about 25–40% of the amino acids in PRPs (Bennick, 1982), which are encoded by six genes (Azen and Maeda, 1988).
What is a gamma turn?
Gamma-turns are the second most commonly found turns (after beta-turns) in proteins. By definition, a gamma-turn contains three consecutive residues (denoted by i, i + 1, i + 2) and a hydrogen bond between the backbone COi and the backbone NHi+2 (see Fig. 1). There are two types of gamma-turns: classic and inverse7.
How many strands are in a beta sheet?
Here a four-stranded beta sheet containing three antiparallel strands and one parallel strand is drawn schematically. Hydrogen bonds between antiparallel strands are indicated with red lines, those between parallel strands with green lines.
How are beta sheets stabilized?
β-Sheets are formed when several β-strands self-assemble, and are stabilized by interstrand hydrogen bonding, leading to the formation of extended amphipathic sheets in which hydrophobic side-chains point in one direction and polar side-chains in the other (Fig.
How many amino acids are in the beta sheet?
A β-strand is a stretch of polypeptide chain typically 3 to 10 amino acids long with backbone in an extended conformation.
What are turns in polypeptide?
Turns are a fundamental class of polypeptide structure and are defined as sites where the peptide chain reverses its overall direction. In the past 20 years, the peptide field has witnessed major development, stimulated by the discovery of a host of bioactive peptides.
What is a beta helix structure?
A beta helix is a tandem protein repeat structure formed by the association of parallel beta strands in a helical pattern with either two or three faces. … The structure is stabilized by inter-strand hydrogen bonds, protein-protein interactions, and sometimes bound metal ions.
Where might bends or β turns occur B where might intrachain disulfide cross linkages be formed?
An intrachain disulfide cross-linkage could occur between the Cys residues of the peptide located at positions 13 and 24.
What happens when a protein is denatured?
Denaturation involves the breaking of many of the weak linkages, or bonds (e.g., hydrogen bonds), within a protein molecule that are responsible for the highly ordered structure of the protein in its natural (native) state. Denatured proteins have a looser, more random structure; most are insoluble.
Where is proline found?
Proline is often found at the end of α helix or in turns or loops. Unlike other amino acids which exist almost exclusively in the trans- form in polypeptides, proline can exist in the cis-configuration in peptides. The cis and trans forms are nearly isoenergetic.
Why is the amino acid proline often found in β turns?
Statistical analysis revealed that Pro and Gly residues are favored in β-turns presumably due to the cyclic structure of Pro and the flexibility of Gly.
Is glycine in beta sheets?
Background: Glycine is an intrinsically destabilizing residue in beta sheets. In natural proteins, however, this destabilization can be ‘rescued’ by specific cross-strand pairing with aromatic residues.
Is cysteine an amino acid?
Cysteine is a non-essential amino acid important for making protein, and for other metabolic functions. It’s found in beta-keratin. This is the main protein in nails, skin, and hair.
Why is proline common in collagen?
Proline and Hydroxyproline in Collagen Proline is a hydrophobic side chain that is in collagen. Proline is made up specifically of carbons and hydrogens, which also explains how it is so non polar. This is important because fibrous proteins need to be non polar to be able to be structural.
Why is proline CIS?
And proline is the only amino-acid, which can be found in cis conformation (Omega=0 degree), because of the smaller energy difference of proline (it has two carbons bound to the N) between cis and trans states compared to other amino-acid residues (which have a H and a C atom bonded to the peptide N).
Can proline form beta sheets?
Proline is established as a potent breaker of both alpha-helical and beta-sheet structures in soluble (globular) proteins.
Does alpha helix exist proline?
All the amino acids are found in α-helices, but glycine and proline are uncommon, as they destabilize the α-helix.
How is proline synthesized?
Proline in higher plants is synthesized from glutamate and ornithine (Fichman et al., 2014). Glutamate-derived proline requires the bifunctional enzyme 1Δ-pyrroline-5-carboxylate (P5C) synthetase (P5CS), which catalyzes a two-step reaction requiring ATP and NADPH to generate glutamate-γ-semialdehyde (GSA; Figure 1).
Where is glycine found in proteins?
Glycine is the main amino acid in collagen, the main structural protein of connective tissue, such as bone, skin, ligaments, tendons and cartilage.
What are reverse turns?
A reverse turn is region of the polypeptide having a hydrogen bond from one main chain carbonyl oxygen to the main chain N-H group 3 residues along the chain (ie O(i) to N(i+3)). … Reverse turns are very abundant in globular proteins and generally occur at the surface of the molecule.



