What type of bond is CCl4

Emma Valentine
Published Mar 01, 2026
Carbon tetrachloride (chemical formula CCl4) is known to be a covalent compound since it features four nonpolar covalent bonds between carbon and chlorine.
Is CCl4 a polar covalent bond?
Carbon tetrachloride is nonpolar. … As a result, CCl4 has no net positive or negative dipole moment. If an atom other than chlorine had been positioned at any other place in a molecule, the molecule would have a dipole moment and CCl4 would be polar as a result.
How many single bonds are in CCl4?
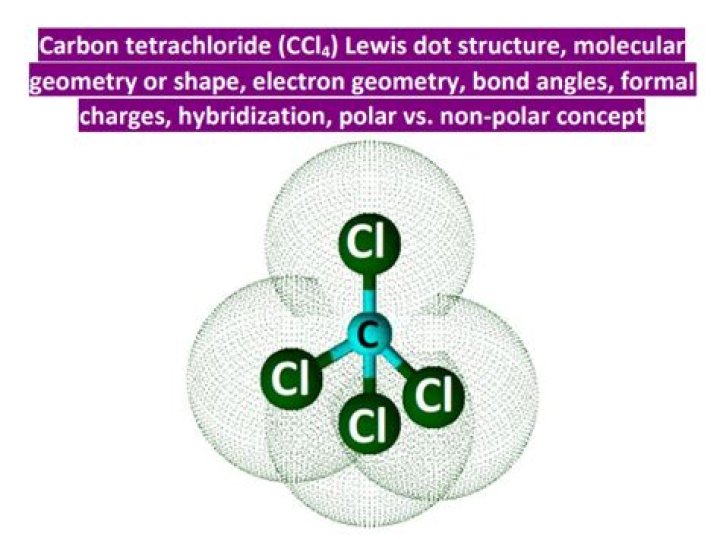
As per the CCl4 lewis structure, carbon is the central atom that has no lone pair present on it because carbon completes its octet with the help of 4 single bonds.
What type of bond is C6H12O6?
Glucose i.e. C6H12O6… there is one ketonic group and five alcoholic group present… All are in a single chain. So all the bonds present in glucose is double bond and covalent bond.What is the electronegativity of ccl4?
According to the Lewis structure, CCl4 is a tetrahedral molecule. The electronegativity for C is 2.5 and Cl is 3.0, resulting in a polar covalent bond.
What type of bond is fe2o3?
It is represented by Fe2O3. The oxidation state of iron oxide is +3 and +2. The bond formed between iron and oxygen is due to the difference in electronegativity between the two atoms. Since iron is metal and oxygen is non-metal therefore the bonding between oxygen and iron is ionic.
How many covalent bonds are in C6H12O6?
The carbon atoms in the glucose ring each have four covalent bonds. The best, or optimum angle, between all these bonds is 109.5o, which results in a perfect tetrahedron.
What type of bond is MgCl2?
MgCl2 is an ionic compound. Magnesium is a metal with a positive charge of +2.Is Cao a covalent bond?
Calcium oxide is ionic because it is formed between a metal and non-metal and bonds formed between a metal and non-metal atoms are ionic. … A bond formed between nonmetal atoms is covalent.
How many electrons does CCl4?Carbon has a valence number of 4 and each of the four chlorine atoms each have a valence number of 7. So the total number of electrons in our diagram of CCl4 should be: 1(4)+4(7) = 32 electrons.
Article first time published onDoes CCl4 have London dispersion forces?
CCl4 is a nonpolar molecule. Its strongest intermolecular forces are London dispersion forces.
Why is CCl4 nonpolar but CH2Cl2 is polar?
Re: Why is CH2Cl2 polar and CCl4 is nonpolar? Answer: In the 4 dipoles cancel each other out making the molecule nonpolar. In , the 2 C-Cl bonds create a dipole towards the Cl since Cl is highly electronegative. This makes the molecule negative towards the Cl and away from the 2 H.
What is bond polarity?
In chemistry, bond polarity is the separation of electric charge along a bond, leading to a molecule or its chemical groups having an electric dipole or dipole moment. Electrons are not always shared equally between two bonding atoms. … Electronegativity measures a particular atom’s attraction for electrons.
Why is CCl4 insoluble in water?
The compound which is made up ionic bond can produce ions in polar solvents like water. But CCl4 is comprising of covalent bond and cannot produce ions. So it is insoluble in water.
What will dissolve in CCl4?
Methane gas has the same structure, making carbon tetrachloride a halomethane. As a solvent, it is well suited to dissolving other non-polar compounds such as fats, and oils. It can also dissolve iodine.
Does CCl4 mix in water?
CCl4 is insoluble in water because it is non polar whereas water is polar. Polar substances are soluble in polar substances and nonpolar in nonpolar.
Is C6H12O6 an ionic or covalent bond?
The difference is EN of C, H and O aren’t great enough to have one atom give up its electrons, so a true ionic bond does not form. This makes all the bonds in C6H12O6 covalent bonds.
What type of bond does CH4 have?
Methane, CH4, is a covalent compound with exactly 5 atoms that are linked by covalent bonds. We draw this covalent bonding as a Lewis structure (see diagram). The lines, or sticks, as we say, represent the covalent bonds. There are four bonds from a central carbon (C) linking or bonding it to four hydrogen atoms (H).
How many bonds does N2 have?
The three bonds appear as the three parallel lines between the N atoms. This is called a triple bond. Each bond is a pair of electrons, one from each connected N atom.
What is the chemical name of fe3o4?
Iron Oxide (fe3o4)
What type of bond is pbo2?
The atoms of lead and oxygen are in their ionic form because the difference in electronegativity between the two is significant.
Why is CaO an ionic bond?
The ionic formula for calcium oxide is simply CaO. Oxygen has six valence electrons and is looking to gain two electrons to complete the octet (8) electron count in the valence shell making it a -2 anion. This one to one ratio of charges makes the formula CaO.
Is SO2 ionic or covalent?
Name of moleculeSulfur dioxide (SO2)SO2 Ionic or Covalent?covalent
What type of bonding exists in MgCl2 and nh3?
covalent bond is your answer.
How would you explain the bonding in MgCl2?
MgCl2 basically they formed by a positive ions and a negative ion. Magnesium has 2+ charge on it Cl has the negative charge on it. So two chlorine charges are going form an ionic bond with magnesium of positive charge and both form MgCl2 , which is a neutral compound.
How many double bonds does CCl4 have?
There are no double bonds. Each chlorine is singly-bonded to the central carbon and each chlorine also features 3 lone pairs (6 total electrons).
How many of each type of bond are found in the Lewis structure for CCl4?
In our example, CCl4, carbon has four bonds around it. Because it normally has four valence electrons, carbon has no charge (4 – 4 = 0). Because each chlorine has one bond and six lone pair electrons (1 + 6 = 7) in this diagram and seven valence electrons, it also has no charge.
What is hybridization of CCl4?
The hybridization of CCl4 is sp3.