What stabilizes primary protein structure

Christopher Lucas
Published Mar 10, 2026
The primary structure is held together by covalent peptide bonds. … Among these forces, the non-specific hydrophobic interaction is the main force driving the folding of protein, while hydrogen bonds and disulfide bonds are responsible for maintaining the stable structure.
What bonds support the primary protein structure?
What type of bonds are found in the primary structure of a protein? Explanation: Primary structure consists of amino acids joined by peptide bonds. Peptide bonds are between the alpha-carboxyl of one amino acid, and the alpha-amine of the next amino acid.
Why are primary protein structures stable?
Hydrogen bonds form between the oxygen of each C=O. bond in the strand and the hydrogen of each N-H group four amino acids below it in the helix. The hydrogen bonds make this structure especially stable. The side-chain substituents of the amino acids fit in beside the N-H groups.
How does a protein structure become stabilized?
Protein stabilization is based on dampening the molecular mo- tions and therefore eliminating conformational transitions while the protein is still in the native state (i.e. has native, fold- ed structure). … The effect of the solvent on the protein is primarily through hydrogen bonding and the polar interactions.How is the secondary structure of protein is stabilized?
The secondary structure of protein is stabilized by H-bonding.
What stabilizes quaternary protein structure?
The quaternary structure of macromolecules is stabilized by the same non-covalent interactions and disulfide bonds as the tertiary structure, and can also be affected by formulation conditions.
What affects the primary structure of a protein?
The main forces that affect structure are electrostatic forces, hydrogen bonding forces, hydrophobic forces, and disulfide bonds. Each of these affect protein structure in different ways. The electrostatic forces are when two like charges are repelled or two opposite charges are attracted.
Which structure of protein is least stable?
The tertiary structure of the protein The most important feature of tertiary structures is the presence of conserved regions with similar functions known as functional domains. The tertiary structures are less stable, and indeed, most of them change shape during the lifetime of the protein, often multiple times.What does it mean for a protein to be stable?
Publisher Summary. This chapter describes protein stability as the net balance of forces, which determine whether a protein will be its native folded conformation or a denatured (unfolded or extended) state. The net stability of proteins is quite small and is the difference between two large opposing forces.
What type s of bonds forces stabilize protein tertiary structure?Once the nonpolar amino acids have formed the nonpolar core of the protein, weak van der Waals forces stabilize the protein. Furthermore, hydrogen bonds and ionic interactions between the polar, charged amino acids contribute to the tertiary structure.
Article first time published onWhat bonds stabilize tertiary structure?
Tertiary structure is stabilized by multiple interactions, specifically side chain functional groups which involve hydrogen bonds, salt bridges, covalent disulfide bonds, and hydrophobic interactions.
What interactions are responsible for maintaining quaternary protein structure?
Quaternary Structure : Example Question #5 Quaternary structure describes how polypeptide chains fit together to form a complete protein. Quaternary protein structure is held together by hydrophobic interactions, and disulfide bridges.
What bonds hold quaternary structures of a protein together?
The quaternary structure of a protein is the association of several protein chains or subunits into a closely packed arrangement. Each of the subunits has its own primary, secondary, and tertiary structure. The subunits are held together by hydrogen bonds and van der Waals forces between nonpolar side chains.
Which interaction helps stabilize the tertiary level of protein organization?
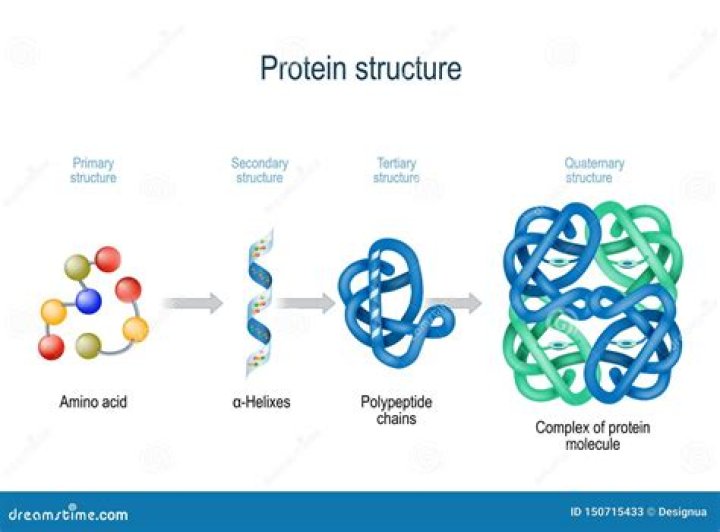
The tertiary structure of a protein refers to the overall three-dimensional arrangement of its polypeptide chain in space. It is generally stabilized by outside polar hydrophilic hydrogen and ionic bond interactions, and internal hydrophobic interactions between nonpolar amino acid side chains (Fig. 4-7).
What causes change in protein structure?
Proteins change their shape when exposed to different pH or temperatures. The body strictly regulates pH and temperature to prevent proteins such as enzymes from denaturing. Some proteins can refold after denaturation while others cannot.
What factors affect protein binding?
Protein binding by this method can be affected by drug stability, radioactive tracer purity, time of equilibration, dilution, temperature, pH, buffer composition, and colloidal osmotic fluid shifts caused by plasma proteins.
When a protein is denatured how is its primary secondary tertiary and quaternary structure affected?
When a protein loses its three-dimensional shape, it may no longer be functional. These unfolded proteins are denatured. Denaturation implies the loss of the secondary structure and tertiary structure (and, if present, the quaternary structure) without the loss of the primary structure.
Why are proteins unstable?
Protein function requires the folded protein form, but this form is unstable mainly because it readily unfolds into a flexible, unstructured form. … Engineering mores stable proteins involves making substitutions that shift the folding–unfolding balance toward the folded form.
What does the primary structure of a protein refer to?
Primary structure. The simplest level of protein structure, primary structure, is simply the sequence of amino acids in a polypeptide chain. … Each chain has its own set of amino acids, assembled in a particular order.
What is the most stable structure?
Triangle is most stable structure. If u apply force on any link or joint the net force is distributed all over the structure ie sides and joints. This means all sides and joint experience force thus the force is distributed and this makes it more rigid and stable structure.
What are primary and secondary structures of proteins?
The primary structure is comprised of a linear chain of amino acids. The secondary structure contains regions of amino acid chains that are stabilized by hydrogen bonds from the polypeptide backbone. These hydrogen bonds create alpha-helix and beta-pleated sheets of the secondary structure.
What is the force that maintains the primary structure?
The primary structure of a protein refers to the sequence of amino acids in the polypeptide chain. The primary structure is held together by peptide bonds that are made during the process of protein biosynthesis.
What forces stabilize proteins?
- Hydrophobic Interactions. Hydrophobic bonds are a major force driving proper protein folding. …
- Van Der Waals forces. The Van der Waals force is a transient, weak electrical attraction of one atom for another. …
- Ionic Bonds- Salt Bridges. …
- Hydrogen bonds.
Which functional group stabilizes proteins?
Both the sulfhydryl groups are responsible for stabilizing a protein structure by making covalent bonds or cross linking between each other.
What bonds are in secondary protein structure?
Secondary structure refers to regular, recurring arrangements in space of adjacent amino acid residues in a polypeptide chain. It is maintained by hydrogen bonds between amide hydrogens and carbonyl oxygens of the peptide backbone. The major secondary structures are α-helices and β-structures.
Which protein is more stable?
Protein #1 will be more stable because its disulfide bond will constraint the UNFOLDED form greatly comparing to the disulfide bond in protein #2. This means that the unfolded protein form of protein #1 will be less stable than the protein #2 leading to a greater stability when folded.