What shape is an p orbital

Emily Dawson
Published Mar 22, 2026
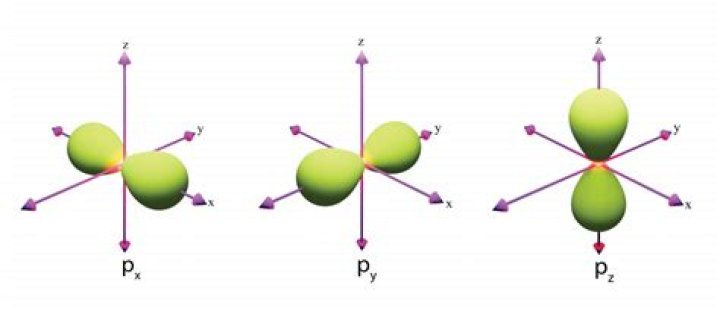
A p orbital has the approximate shape of a pair of lobes on opposite sides of the nucleus, or a somewhat dumbbell shape. An electron in a p orbital has equal probability of being in either half. The shapes of the other orbitals are more complicated.
What shape do the p orbitals have?
Each orbital type has a unique shape based on the energy of its electrons. The s orbital is a spherical shape. The p orbital is a dumbbell shape. There are three p orbitals that differ in orientation along a three-dimensional axis.
What does P mean in orbitals?
You might expect that the ‘s’ stands for ‘spherical’ and ‘p’ stands for ‘polar’ because these imply the shapes of the s and p orbitals, but unfortunately, the letter designations have nothing to do with the orbital shapes.
Why are p orbitals shaped the way they are?
That Said, electrons suffer the actions of forces (attraction towards the nucleus, due to rotation, etc)…the variation of this forces in time is called a potential. The orbital are shaped in the way they are allowed to, by this forces. This is explicit calculated solving Schrodinger equations, for one electron atoms.Why is p orbital dumbbell-shaped?
The p orbital is a dumbbell shape because the electron is pushed out twice during the rotation to the 3p subshell when an opposite-spin proton aligns gluons with two same-spin protons.
What is the shape of 2p orbital?
p-orbital is dumb-bell shaped.
How many orbitals are there in P shell?
This number divides the subshell into individual orbitals which hold the electrons; there are 2l+1 orbitals in each subshell. Thus the s subshell has only one orbital, the p subshell has three orbitals, and so on.
What is the shape of orbitals containing 2p electrons?
Each 2p orbital has two lobes. There is a planar node normal to the axis of the orbital (so the 2px orbital has a yz nodal plane, for instance). The higher p-orbitals ( 3p, 4p, 5p, 6p, and 7p) are more complex still since they have spherical nodes as well.What are the 4 shapes of orbitals?
There are four basic types of orbitals: s, p, d, and f. An s orbital has a spherical shape and can hold two electrons. There are three p orbitals, each of which has the same basic dumbbell shape but differ in its orientation in space. The p orbitals can hold up to six electrons.
Are p orbitals directional?In a p orbital, the probability of finding an electron at a particular distance is greatest along the x, y, or z axis. A p orbital is directional.
Article first time published onWhere are the p orbitals located?
The p orbitals at the second energy level are called 2p x , 2p y and 2p z . There are similar orbitals at subsequent levels: 3p x , 3p y , 3p z , 4p x , 4p y , 4p z and so on. All levels except for the first level have p orbitals.
Which orbitals are spherically symmetrical?
All s orbitals are spherically symmetrical. That is, an electron that occupies an s orbital can be found with the same probability at any orientation (at a given distance) from the nucleus.
What is the shape of ap orbital how many p orbitals are there in a sublevel?
This gives us that a sublevel has 3 p orbitals.
How many p orbitals are in the principal energy level?
Each principal energy level above the first contains one s orbital and three p orbitals. A set of three p orbitals, called the p sublevel, can hold a maximum of six electrons. Therefore, the second level can contain a maximum of eight electrons – that is, two in the s orbital and 6 in the three p orbitals.
What is the shape of 5p orbital?
Each 5p orbital has a planar node normal to the axis of the orbital (so the 5px orbital has a yz nodal plane, for instance) and three spherical nodal surfaces. The higher p-orbitals (6p, and 7p) are more complex since they have spherical nodes as well while lower p orbitals (2p, 3p, and 4p) have fewer.
What is the shape of 4s orbital?
The shape of the 4s orbital. That on the left is sliced in half to show the two spherical nodes of the 4s orbital. The shape on the right shows the nodal structure of the 4s-orbital. While still spherical, the higher s-orbitals (5s, 6s, and 7s) are more complex since they have more spherical nodes.
How many types of p orbitals are there?
p orbitals There are three types of p orbital. Each points in a different direction.
What are the 7 orbitals?
The shape of the seven 7f orbitals (cubic set). From left to right: (top row) 7fy 3, 7fz 3, 7fx 3, (middle row) 7fy(z 2-x 2), 7fz(x 2-y 2), and 7fx(z 2-y 2) (bottom row) 7fxyz. For each, the green zones are where the wave functions have positive values and the white zones denote negative values.
How many p orbitals are in the N 3 shell?
There are nine orbitals in the n = 3 shell. There is one orbital in the 3s subshell and three orbitals in the 3p subshell. The n = 3 shell, however, also includes 3d orbitals. The five different orientations of orbitals in the 3d subshell are shown in the figure below.
Which of the following is correct with respect to p orbitals?
Explanation – p-orbitals are d. umb bell shaped orbitals. They, therefore, have a strong directional character.
Which orbitals are non directional what is their shape?
s orbitals are non-directional because they have spherical symmetry which means that the probability of finding an electron at a particular distance from the nucleus is same in all directions as compared with a p orbital which has somewhat a dumbbell like shape.
Which orbital is more directional in nature?
P orbital possess equal Electron Density on both sides of the orbital , but in case of sp orbital electron density isn’t equal and that’s the reason why sp is more directional than p . That’s also the reason for their different Electronegativity.
How is a 2p orbital different from a 2s orbital?
The 2 s and 2 p orbitals differ in shape, number, and energy. A 2 s orbital is spherical, and there is only one of them. A 2 p orbital is dumbbell-shaped, and there are three of them oriented on the x, y, and z axes. The 2 p orbitals have higher energy than the 2 s orbital.
Is P or P3 bigger?
Phosphorus is a Group 15 element so its only ionic state is P3-. Anions of a given element have a larger radius than the neutral atom, so P3- will increase in size compared with the P atom.
What is phosphorus P 15 electron configuration?
Answer:The electronic configuration of phosphorus is 1s^2 2s^2 2p^6 3s^2 3p^3.
What is meant by 3p3?
#33 what is meant by 3p3? 3rd orbital in p sublevel 3 electrons. #34a give the electron configuration for Na. 1s2 2s2 2p6 3s1.
Are all orbitals the same size?
Atoms are roughly spherical because when all of the different shaped orbitals are overlapped, they take on a spherical shape. … All orbitals in a given atom are roughly the same size.
What orbital is spherically symmetric around the nucleus of the atom?
The hydrogen atom wavefunctions, ψ(r,θ,φ), are called atomic orbitals.
Why is orbital is spherically symmetrical?
The orbitals are segregated by the amount of angular momentum that they carry. No angular momentum, ℓ=0, are called s-orbitals, and are spherically symmetric.
What is the Atom shape?
Originally Answered: What is the shape of an atom? The easy answer is that atoms are not spherical in shape. Sphere is a solid object, atoms to not have well defined boundaries with “solid cutoffs”. Atoms are more like clouds consisting distributions of electrons, neutrons and protons.