What makes sodium phosphate

Rachel Hickman
Published May 14, 2026
Sodium phosphate is derived from purified, finely ground phosphate rock that is reacted with sulfuric acid to yield phosphoric acid. The Phosphoric acid is then purified and reacted with Sodium Hydroxide to form Sodium Phosphate.
Where is sodium phosphate made?
Description: Sodium phosphates are made from mined phosphate rock. The rock is crushed and mixed with sulfuric acid. Phosphoric acid is then extracted from the mixture.
How do you make sodium phosphate?
- Prepare 800 mL of distilled water in a suitable container.
- Add 70 g of Na2HPO4 to the solution.
- Add 30 g of NaH2PO4 · H2O to the solution.
- Autoclave and store at room temperature.
How is Na3PO4 made?
Production. Trisodium phosphate is produced by neutralization of phosphoric acid using sodium carbonate, which produces disodium hydrogen phosphate. The disodium hydrogen phosphate is reacted with sodium hydroxide to form trisodium phosphate and water.Is sodium phosphate a natural ingredient?
Sodium phosphate is naturally occurring in many foods. It’s also added to foods to maintain freshness, alter texture, and achieve a variety of other effects. Sodium phosphate is considered safe by the FDA but should be avoided by certain people, including those with kidney disease.
What element is sodium phosphate?
Sodium phosphate is a group of chemical compounds. The phosphate atom can hold three other atoms. They can be three hydrogen (phosphoric acid), two hydrogen and one sodium(sodium dihydrogen phosphate), one hydrogen and two sodium (disodium hydrogen phosphate), or three sodium (trisodium phosphate).
What is na3po4 pH?
The aqueous soln is strongly alkaline. pH of 0.1% soln: 11.5; of 0.5% soln: 11.7; of 1.0% soln: 11.9.
What does sodium and phosphorus make?
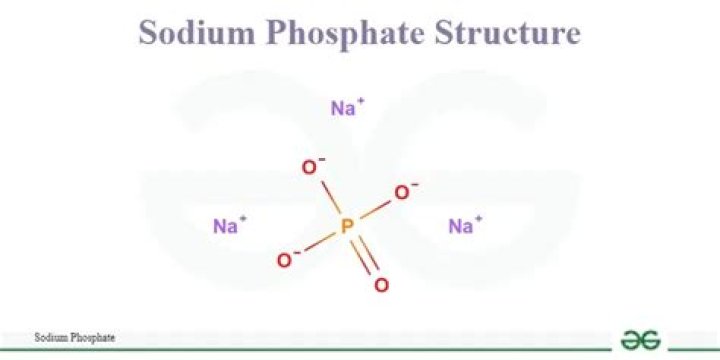
Sodium Phosphate is an ionic compound formed by two ions, Sodium Na+ and Phosphate PO−34 . In order for these two polyatomic ions to bond the charges must be equal and opposite. … This will make the formula for Sodium Phosphate Na3PO4 . I hope this was helpful.What can I use instead of trisodium phosphate?
If you are looking for a more natural trisodium phosphate substitute, borax can be a fine replacement. It doesn’t require all the safety measures of TSP and is inexpensive, easy to use and it won’t hurt the environment. Borax can kill fungus and strip away dirt and grease on porous surfaces such as wood and cement.
Is sodium phosphate a mixture?Sodium Phosphates Description Sodium Phosphates Injection, USP, 3 mmol/mL (millimoles/mL), is a sterile, nonpyrogenic, concentrated solution containing a mixture of monobasic sodium phosphate and dibasic sodium phosphate in Water for Injection.
Article first time published onHow do you make Na2HPO4?
- Prepare 800 mL of distilled water in a suitable container.
- Add 3.569 g of Sodium phosphate dibasic heptahydrate to the solution.
- Add 0.922 g of Sodium phosphate monobasic monohydrate to the solution.
- Adjust solution to final desired pH using HCl or NaOH.
- Add distilled water until volume is 1 L.
What is the common name for sodium phosphate?
SynonymsSourcesphosphoric acid, sodium saltChemIDplussodium orthophosphate Note: (2007-01-29) Not recommended since this is a common name of trisodium phosphate Na3PO4 (CHEBI:37583).ChemIDplus
Why is sodium phosphate bad?
Sodium phosphate can cause serious kidney damage and possibly death. In some cases, this damage was permanent, and some people whose kidneys were damaged had to be treated with dialysis (treatment to remove waste from the blood when the kidneys are not working well).
Why is sodium phosphate in toothpaste?
What is Sodium Phosphate Used In/For? Sodium Phosphate is used as an emulsifier and thickening agent. It is also used as a leavening agent to control the pH balance of processed foods. In toothpaste, this active ingredient is used to protect the tooth enamel from bacteria.
Is sodium phosphate unhealthy?
Eating too many foods containing sodium phosphate additives can raise phosphate levels in the body to an unhealthy level. Studies have linked high levels of phosphate with conditions like heart disease, decreased bone density, premature aging, kidney issues and even early death ( 7 ).
How does Na3PO4 dissociate?
When Na3PO4 N a 3 P O 4 is dissolved in water, it dissociate into its ions (sodium ions and phosphate ions).
Why is Na3PO4 a cleaning agent?
Highly soluble in water producing an alkaline solution. The pH of a 1% solution is between pH 11.8 – 12.0. The versatility of this product coupled with the lack of toxicity make TSP a preferred cleaning agent for a wide range of cleaning purposes.
Is Na3PO4 basic or neutral?
Answer and Explanation: Due to this anion reactivity, a solution of sodium phosphate will be basic.
Can you use vinegar instead of TSP?
Hardware and paint stores carry TSP-alternatives that both clean and dull paint for painting. Some of these cleaners don’t require any rinsing. White vinegar is an effective mildew and mold remover. Apply it full strength, scrub and rinse away mildew stains.
Is Borax the same as TSP?
An Alternative to Cleaning with TSP Also known as sodium borate, borax is a naturally-occurring mineral compound that will clean many of the same surfaces TSP has been used on for years, including mold- and mildew-stained areas—all without causing damage nearby plants.
Does Walmart sell TSP?
Lundmark TSP Hard Surface Cleaner – Walmart.com.
Is phosphate and phosphorus the same thing?
Phosphorus is a mineral that combines with other substances to form organic and inorganic phosphate compounds. The terms phosphorus and phosphate are often used interchangeably when talking about testing, but it is the amount of inorganic phosphate in the blood that is measured with a serum phosphorus/phosphate test.
Where does phosphate come from?
Phosphate reserves are found in Africa, North America, Kazakhstan, the Middle East and Oceania but the world’s largest deposits are located in Morocco, which is also one of the global leaders in phosphate extraction.
What are the composition of phosphate ions?
Formula and structure: The chemical structure of phosphate is PO43-. The molar mass is 94.97 g/mol. It is formed by a centered phosphorous atom that is bond to four oxygen atoms, one of the bonds is a double bond and the other three oxygen atoms have simple bonds.
What is the percent composition of sodium in sodium phosphide?
The percentage composition of sodium phosphate as determined by analysis is 42.1% sodium, 18.9% phosphorus and 39% oxygen.
What does sodium phosphate do?
Sodium phosphate is a saline laxative that is thought to work by increasing fluid in the small intestine. It usually results in a bowel movement after 30 minutes to 6 hours.Do not use this medication in children younger than 5 years of age unless directed by the doctor.
What is sodium pyrophosphate?
Tetrasodium pyrophosphate, also called sodium pyrophosphate, tetrasodium phosphate or TSPP, is an inorganic compound with the formula Na4P2O7. As a salt, it is a white, water-soluble solid. It is composed of pyrophosphate anion and sodium ions. Toxicity is approximately twice that of table salt when ingested orally.
What is sodium acid pyrophosphate made from?
SAPP is manufactured by partially neutralizing food grade phosphoric acid with sodium hydroxide or sodium carbonate to form monosodium phosphate. Dehydration of monosodium phosphate at 250°C will form SAPP. Currently, there is no known natural method for the production of SAPP.
What is NaH2PO4 in chemistry?
Sodium phosphate (NaH2PO4) Sodium orthophosphate, primary. Sodium dihydrogen monophosphate.
How do I make PBS?
- Prepare 800 mL of distilled water in a suitable container.
- Add 8 g of NaCl to the solution.
- Add 200 mg of KCl to the solution.
- Add 1.44 g of Na2HPO4 to the solution.
- Add 245 mg of KH2PO4 to the solution.
- Adjust solution to desired pH (typically pH ≈ 7.4).
Is NaH2PO4 a salt?
NaH2PO4 and NaHPO3 are acid salts. … If these oxoacids will contain more than one acidic hydrogen atom, then only the salt will be an acidic salt whereas if the compound will contain only one acidic hydrogen, the compound will then be called normal or neutral salts.