What makes an alpha helix

Mia Kelly
Published Feb 23, 2026
An alpha helix is a common shape that amino acid chains will form. The alpha helix is characterized by a tight right-handed twist in the amino acid chain that causes it to form a rod shape. Hydrogen bonds between the hydrogen in an amino group and the oxygen in a carboxyl group on the amino acid cause this structure.
Who described the alpha helix?
PNAS papers by Linus Pauling, Robert Corey, and Herman Branson in the spring of 1951 proposed the alpha-helix and the beta-sheet, now known to form the backbones of tens of thousands of proteins.
What is another name for alpha helix?
The alpha helix is also called a classic Pauling–Corey–Branson α-helix. The name 3.613-helix is also used for this type of helix, denoting the average number of residues per helical turn, with 13 atoms being involved in the ring formed by the hydrogen bond.
What is meant by α helix?
Definition of alpha-helix : the coiled structural arrangement of many proteins consisting of a single chain of amino acids stabilized by hydrogen bonds.Why is alpha helix more stable?
α-helix is right handed and is more stable due to inter molecular H bonding between first and fourth amino acid.
Who discovered the protein?
Proteins were first described by the Dutch chemist Gerardus Johannes Mulder and named by the Swedish chemist Jöns Jacob Berzelius in 1838.
Why is alpha helix right handed?
Despite the fact that, based on the Ramachandran plot, both right-handed and left-handed alpha helices are among the permitted conformations, the right-handed alpha helix is energetically more favorable because of fewer steric clashes between the side chains and the main chain.
How is alpha helix stabilized?
The α helix is stabilized by hydrogen bonds between an amide hydrogen of one amino acid and a carbonyl oxygen four amino acids away.Why Proline is called Helix breaker?
Proline and glycine are sometimes known as “helix breakers” because they disrupt the regularity of the α helical backbone conformation; however, both have unusual conformational abilities and are commonly found in turns.
Which amino acids favor alpha helix?For example, alanine, leucine, lysine and glutamate favor alpha helix; whereas, beta-branched amino acids threonine, valine, and isoleucine favor beta sheet.
Article first time published onWhy is glycine not in alpha helix?
All the amino acids are found in α-helices, but glycine and proline are uncommon, as they destabilize the α-helix. Glycine is exempt from many steric constraints because it lacks a β carbon. … Proline, on the other hand, is too rigid.
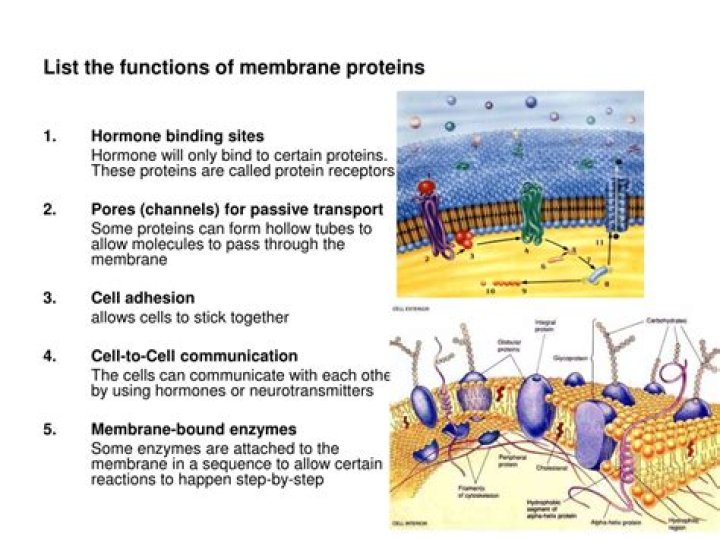
What is the function of alpha helix protein?
α-helical membrane proteins are responsible for interactions between most cells and their environment. [5] Trans-membrane (TM) helices are typically encoded by stretches of 17-25 residues [6], which provide sufficient length to cross the membrane.
What is the polypeptide chain?
A polypeptide is an unbranched chain of amino acids that are linked together by peptide bonds. The peptide bond links the carboxyl group of one amino acid to the amine group of the next amino acid to form an amide. … Short polypeptides may be named based on the number of monomeric amino acids that comprise them.
Is DNA an alpha helix?
The secondary structure of DNA is actually very similar to the secondary structure of proteins. The protein single alpha helix structure held together by hydrogen bonds was discovered with the aid of X-ray diffraction studies.
What holds alpha helix together?
The alpha-helix is a right-handed helical coil that is held together by hydrogen bonding between every fourth amino acid.
Why is collagen left handed?
Left handed helices are formed because of the high content of proline and hydroxyproline rings, with their geometrically constrained carboxyl and (secondary) amino groups along with abundance of glycine. The left handed helices are formed without any intrachain hydrogen bonding.
Why is alpha helix Amphipathic?
As the name implies, an amphipathic (or amphiphilic) helix is an α-helix with both hydrophobic and hydrophilic amino acid residues arranged in such a way as to create two faces on opposite sides of the helix, one face being hydrophobic.
What is alpha helix and beta sheet?
The most common types of secondary structures are the α helix and the β pleated sheet. Both structures are held in shape by hydrogen bonds, which form between the carbonyl O of one amino acid and the amino H of another. Images showing hydrogen bonding patterns in beta pleated sheets and alpha helices.
Who is Pauling in DNA?
In the 1950’s, Linus Pauling became known as the founder of molecular biology due to his discovery of the spiral structure of proteins (Taton, 1964). Pauling’s discoveries contributed to Watson and Crick’s breakthrough of the DNA double helix.
Who is the father of enzymes?
French chemist Anselme Payen was the first to discover an enzyme, diastase, in 1833.
Who is the father of RNA?
Leslie Orgel, 80; chemist was father of the RNA world theory of the origin of life – Los Angeles Times.
Why is proline not in alpha helix?
Proline is formally NOT an amino acid, but an imino acid. … When proline is in a peptide bond, it does not have a hydrogen on the α amino group, so it cannot donate a hydrogen bond to stabilize an α helix or a β sheet. It is often said, inaccurately, that proline cannot exist in an α helix.
Why does alanine form alpha helix?
The helix propensities of nonpolar amino acids are correlated linearly with side-chain entropy, indicating that alanine favors helix formation in part because no reduction in side-chain entropy is required on helix formation (28–30).
Is serine polar or non polar?
Amino acidSingle Letter CodePolarityprolinePnonpolarserineSpolarthreonineTpolartryptophanWnonpolar
Why is it called glycine?
The name comes from the Greek word γλυκύς “sweet tasting” (which is also related to the prefixes glyco- and gluco-, as in glycoprotein and glucose). In 1858, the French chemist Auguste Cahours determined that glycine was an amine of acetic acid.
Why do alpha helices form before beta sheets?
The alpha helix is formed when the polypeptide chains twist into a spiral. This allows all amino acids in the chain to form hydrogen bonds with each other. … The beta pleated sheet is polypeptide chains running along side each other. It is called the pleated sheet because of the wave like appearance.
How many amino acids are in alpha helix?
Any of the 20 amino acids can participate in an α-helix but some are more favored than others. Ala, Glu, Leu, and Met are most often found in helices whereas, Gly, Tyr, Ser, and Pro are less likely to be seen.
Which is more stable alpha helix or beta sheet?
No change was observed upon heating a beta-sheet sample, perhaps due to kinetic effects and the different heating rate used in the experiments. These results are consistent with beta-sheet approximately 260 J/mol more stable than alpha-helix in solid-state PLA.
Why is serine not in alpha helix?
UNFAVORABLE AMINO ACIDS IN ALPHA HELICES Unfavorable because of size/charge/shape of side chains, , which can destabilize helices. They include: proline, glycine, serine, aspartate, asparagine, threonine, valine and isoleucine.
What is asparagine classified?
Asparagine is a non-essential amino acid in humans, Asparagine is a beta-amido derivative of aspartic acid and plays an important role in the biosynthesis of glycoproteins and other proteins. A metabolic precursor to aspartate, Asparagine is a nontoxic carrier of residual ammonia to be eliminated from the body.
What is the pH of cysteine?
The structure of cysteine at pH = 7 shows that the side group is protonated. So we must conclude that even though the pKa is 8.33, the sulfhydryl (−SH) is acting as an acid. The isoelectric point, pI, is the pH at which the zwitterion is the dominant species.