What liquids have a low pH

Christopher Lucas
Published Mar 22, 2026
Acidic liquids have low pH levels. Many fruit juices qualify as acidic liquids. Some of the more acidic fruit juices are orange juice, lemon juice, grapefruit juice, tomato juice and red currant juice.
What is the lowest level of pH?
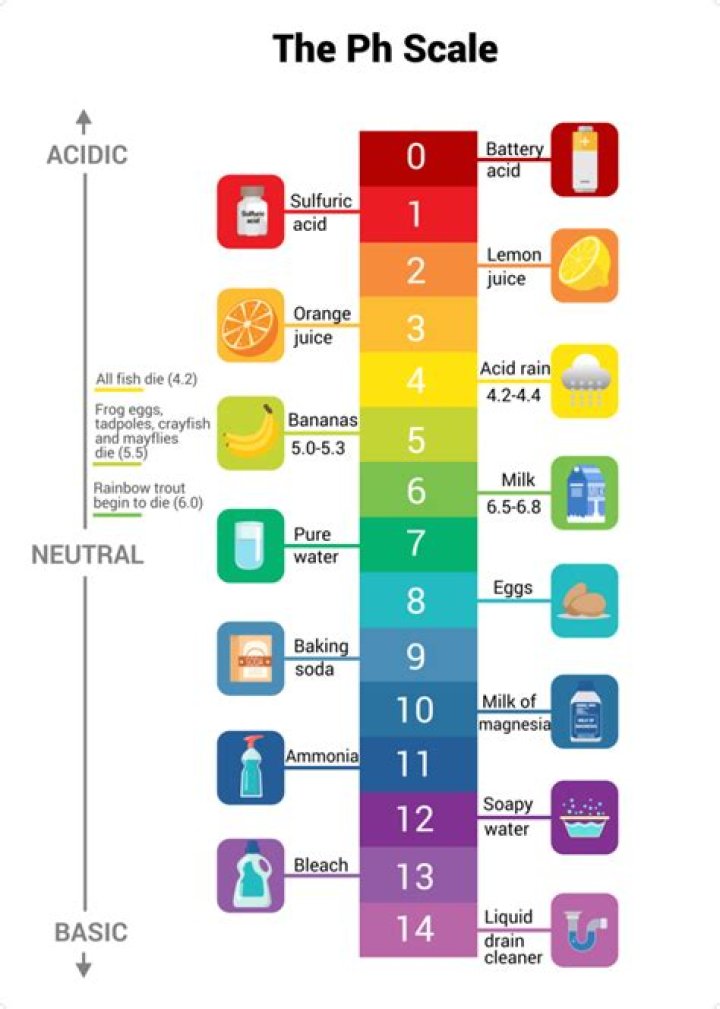
The range goes from 0 – 14, with 7 being neutral. pHs of less than 7 indicate acidity, whereas a pH of greater than 7 indicates a base.
Is Acid high or low pH?
The pH scale ranges from 0 to 14, with 7 being neutral. pHs less than 7 are acidic while pHs greater than 7 are alkaline (basic).
What substance has a pH lower than 7?
A pH less than 7 is acidic. A pH greater than 7 is basic.What things are basic pH?
Basic substances include things like baking soda, soap, and bleach. Distilled water is a neutral substance.
Does everything have a pH?
pH numbers are a measure of the concentration of hydrogen ions in a solution. Only solutions have pH – solids, gases and non-aqueous liquids don’t.
What liquids have pH?
pH ValueH+ Concentration Relative to Pure WaterExample010 000 000battery acid11 000 000gastric acid2100 000lemon juice, vinegar310 000orange juice, soda
Can something have a pH?
Yes, the pH scale can go above 14 and below 0, but it’s not super common that the pH of a substance ever is. If it is, it’s either very acidic or very basic. So basically the negative log of the concentration of hydrogen ions (in mol/L).What substances have a pH of 7?
Increasing pH (Decreasing Acidity)Substances7 (neutral)Water, tears7.5Human blood8Seawater9Baking soda, antacids
Which substance has a pH that is greater than pure water?SubstancepHstomach acid1.7lemon juice2.2vinegar2.9soda3.0
Article first time published onWhich substances has a pH greater than 7?
If a substance has a pH that is greater than 7, it is alkaline. This is consistent with answer choice “e”.
Which solution has the lowest pH value?
From all the above options vinegar has the lowest pH-value that means it is the most acidic from the above and the washing soda is most basic. Hence, the correct option is (b)- vinegar.
What is the pH of baking soda?
Baking soda, also known as sodium bicarbonate, is a base. This means that when people dissolve baking soda in water, it forms an alkaline solution. For example, a 0.1 molar solution of baking soda has a pH of around 8.3. Lemon juice contains citric acid and has a pH of around 3.
What is high and low pH?
The pH scale ranges from 0 to 14, with 7 being neutral. pHs less than 7 are acidic while pHs greater than 7 are alkaline (basic).
What causes low pH in water?
Oftentimes, acidic water is due to industrial pollution, with low pH water often being found near mining sites, chemical dumps, power plants, confined animal feeding operations, and landfills (2). Acidic water has a pH of 6.5 or less and can be caused by natural phenomena, as well as industrial pollution.
What is the pH of sodium hydroxide?
pHproduct14sodium hydroxide13lye12.4lyme11ammonia
What types of substances are acidic?
Increasing pH (Decreasing Acidity)Substances0 (most acidic)Hydrochloric acid (HCl)1Stomach acid2Lemon juice3Cola, beer, vinegar
What has the highest pH?
The highest pH value which be attained is 14, which indeed it really tough to attain. PH of pure water at 208k is 7, meaning there is a 10^-7 mol/dm^3 concentration of H+ ions. Thus is due to the fact that water auto disassociate into H+ and OH- ions.
What is the pH of milk?
Cow’s milk Milk — pasteurized, canned, or dry — is an acid-forming food. Its pH level is below neutral at about 6.7 to 6.9. This is because it contains lactic acid. Remember, though, that the exact pH level is less important than whether it’s acid-forming or alkaline-forming.
What is the pH of apple juice?
FoodspHReferenceapple juice, Delicious3.55-3.7910apple juice, Delicious4.04-4.2410apple juice, Delicious4.01-4.3311apple juice, Golden Delicious3.78-3.9410
Why is Coke acidic?
A big component of the taste and acidity of Coca-Cola comes from Phosphoric Acid. Phosphoric Acid leaches calcium out of bones and teeth, and can contribute to kidney disease and stones.
Is lemon juice basic or acidic?
Lemon juice has a pH between 2 and 3, which means it’s 10,000–100,000 times more acidic than water (1, 2, 3). A food’s pH is a measure of its acidity. The pH of lemon juice falls between 2 and 3, meaning it is acidic.
Does oil have pH?
Because only aqueous solutions have pH levels, vegetable oil has no pH value. Likewise, other oils such as animal and petrochemical oils also have no pH value. Acidity as it relates to flavor should not be confused with an oil’s fatty acid content. … These acids, again, have nothing to do with pH levels.
Can solid things have pH?
pH of a solid cannot be determined nor pH is defined for pure organic solvents e.g. benzene. However you can measure pH of a suspension in water (at least in principle).
What does a negative pH mean?
In practice, any acid that yields a concentration of hydrogen ions with a molarity greater than 1 will be calculated to have a negative pH. … This is because glass pH electrodes suffer from a defect called ‘acid error’ which causes them to measure a higher pH than the real pH.
Is water the only substance with a pH of 7?
As such, no substance (other than pure water or certain balanced solutions in water) have a pH of 7. Not even water has a pH of 7: only under certain conditions of temperature is the pH exactly 7.
What is a pH of 6?
A pH of 7 indicates a neutral solution, a pH greater than 7 indicates a basic solution, and a pH of less than 7 indicates an acidic solution. The farther a pH value is from 7, the more strongly acidic or basic it is. For example, water with a pH of 6 is mildly acidic, and water with a pH of 3 is highly acidic.
What is the pH of vinegar?
Vinegar is mildly acidic with a pH of 2–3. Apple cider vinegar is slightly more alkaline than pure vinegar because it contains more alkaline nutrients. However, it’s still acidic.
Is a pH of 0 possible?
The pH value can be less than 0 for very strong acids, or greater than 14 for very strong bases. The pH scale is traceable to a set of standard solutions whose pH is established by international agreement.
How is negative pH possible?
Is this possible? It’s possible. If the molarity of hydrogen ions is greater than 1, you’ll have a negative value of pH. For example, you might expect a 12 M HCl solution to have a pH of -log(12) = -1.08.
What is a weak acid?
Weak acids are acids that don’t completely dissociate in solution. In other words, a weak acid is any acid that is not a strong acid. The strength of a weak acid depends on how much it dissociates: the more it dissociates, the stronger the acid.