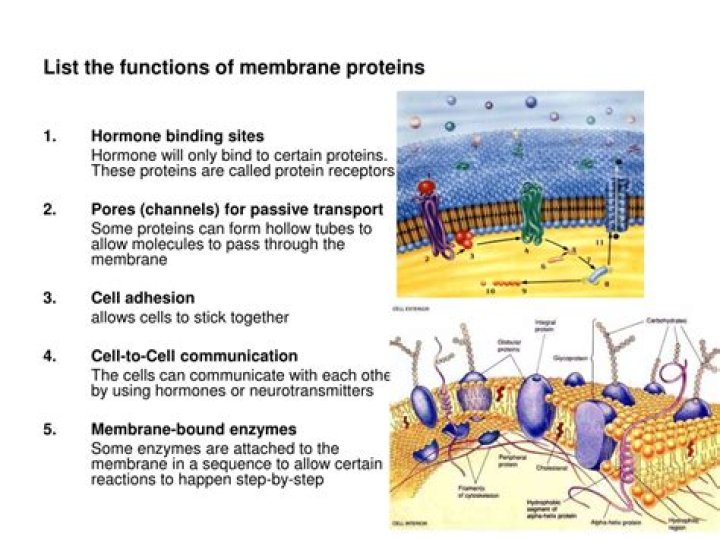
What is the name for Li2O

Victoria Simmons
Published Feb 26, 2026
Lithium oxide | Li2O – PubChem.
What is the systematic name of the following compound \text li }_ 2 \text SLI 2 S?
Dilithium Sulfide | Li2S – PubChem.
What is the correct Iupac name for SrO?
PubChem CID518709StructureFind Similar StructuresMolecular FormulaOSrSynonymsStrontium oxide (SrO) oxostrontium MFCD00049558 Strontium oxide,99% DTXSID00893853 More…Molecular Weight103.62
Is Li2S ionic or covalent?
This short flash animation looks at how the ionic bond forms in Lithium Sulfide (Li2S).What is Li2S charge?
Property NameProperty ValueReferenceFormal Charge1Computed by PubChemComplexity2.8Computed by Cactvs 3.4.8.18 (PubChem release 2021.05.07)Isotope Atom Count0Computed by PubChemDefined Atom Stereocenter Count0Computed by PubChem
What is Be3P2?
Beryllium phosphide | Be3P2 – PubChem.
What is the systematic name of the compound Mn3 PO4 2?
Manganese(II) phosphate | Mn3(PO4)2 – PubChem.
What is Al2S3 name?
Aluminum sulfide | Al2S3 | 99.9 | -100 mesh.What is the name for BeO?
Beryllium oxide (BeO)
What is dilithium sulfide?Lithium sulfide is the inorganic compound with the formula Li2S. It crystallizes in the antifluorite motif, described as the salt (Li+)2S2−. It forms a solid yellow-white deliquescent powder. In air, it easily hydrolyses to release hydrogen sulfide (rotten egg odor).
Article first time published onWhat type of bond is N2O4?
Dinitrogen tetroxide, N2O4 N 2 O 4 is a covalent compound as it is formed by sharing of electrons between nitrogen and oxygen atoms, which are non-metals. Two oxygen share two electrons with one nitrogen each and two oxygen share one electron with each nitrogen.
Is Li2S aqueous?
A Li2S white powder remains insoluble in clear aqueous electrolyte for 12 h. Jacinth solution on the top of bottle is Li2S2 or Li2S4 dissolved in water phase, which is separated from the clear aqueous electrolyte (salt phase) on the bottom.
What is the name for SrS?
NamesChemical formulaSrSMolar mass119.68 g/molAppearancewhite solid (spoiled samples are colored)Odornone (degraded samples smell of hydrogen sulfide)
What ions are present in SrO?
The ionic substance strontium oxide, SrO, forms from the direct reaction of strontium metal with molecular oxygen.
Which of the following is the correct name for n2o2?
PubChem CID6857661StructureFind Similar StructuresMolecular FormulaN2O2Synonymsdioxohydrazine UNII-P6TX5AE4Q8 P6TX5AE4Q8 N-oxonitrous amide 16824-89-8 More…Molecular Weight60.013
What is the name for Mn3 po4 4?
Manganese(IV) Phosphate Mn3(PO4)4 Molecular Weight — EndMemo.
What element is BeO?
Beryllium oxide (BeO) is a white crystalline oxide. It occurs in nature as the mineral “Bromellite”. Historically, beryllium oxide was called glucina or glucinium oxide.
How is BeO made?
Preparation and chemical properties Beryllium oxide can be prepared by calcining (roasting) beryllium carbonate, dehydrating beryllium hydroxide, or igniting metallic beryllium: BeCO3 → BeO + CO. Be(OH)2 → BeO + H2O. … Igniting beryllium in air gives a mixture of BeO and the nitride Be3N2.
Is beryllium a metal or nonmetal?
Beryllium is a silvery-white metal. It is relatively soft and has a low density. Beryllium is used in alloys with copper or nickel to make gyroscopes, springs, electrical contacts, spot-welding electrodes and non-sparking tools. Mixing beryllium with these metals increases their electrical and thermal conductivity.
Is NH4 a 2S?
Ammonium sulfide ((NH4)2S) exhibits high reactivity as a sulfide reagent in anion exchange reactions that transform CoO to cobalt sulfide nanoparticles (NPs).
What's the chemical name for K3N?
Potassium Nitride K3N Molecular Weight — EndMemo.
What is MgS name?
Magnesium sulfide (MgS)