What is the bicarbonate buffer system and what is it used for

Robert Spencer
Published Mar 10, 2026
The bicarbonate buffering system maintains optimal pH levels and regulates the carbon dioxide concentration that, in turn, shifts any acid–base imbalance. Renal physiology controls pH levels through several powerful mechanisms that excrete excess acid or base.
What is the bicarbonate buffer system used for?
In the human stomach and duodenum, the bicarbonate buffer system serves to both neutralize gastric acid and stabilize the intracellular pH of epithelial cells via the secretion of bicarbonate ion into the gastric mucosa.
What is the primary role of bicarbonate?
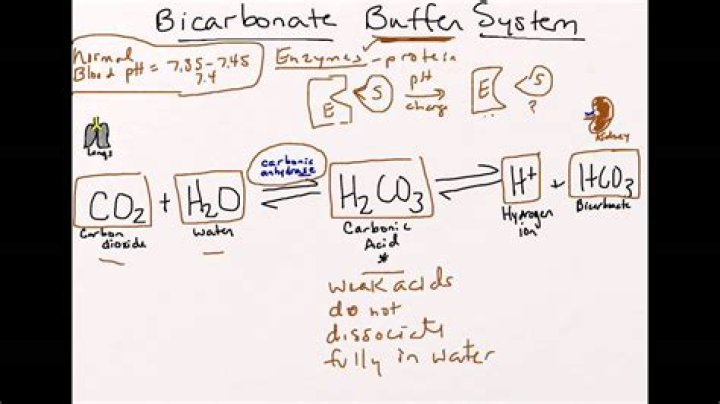
Bicarbonate is an essential component of the physiological pH buffering system in the human body. Up to ¾ of the carbon dioxide in the human body is converted to carbonic acid which is quickly turned to bicarbonate. Bicarbonate is an alkali so helps to keep the acid-base balance of the body stable.
How does the bicarbonate buffer system work quizlet?
The bicarbonate buffer system is the most important buffer in the extracellular fluid. If too much acid (lactic acid) is present, the bicarbonate ions take up hydrogen ions released by the acid and become carbonic acid. … Thus the excess acid is neutralized when bicarbonate ions are used in the buffering process.How does buffer system work?
Buffers work by neutralizing any added acid (H+ ions) or base (OH- ions) to maintain the moderate pH, making them a weaker acid or base. … Thus the breaking of the buffer is its capacity, or in other words, it is the amount of acid or base, a buffer can absorb before breaking its capacity.
What are the functions of bicarbonate and carbonic acid in blood quizlet?
A. Because of its high concentration in blood, the bicarbonate/carbonic acid pair is the most important buffer system in the blood. This buffer system is also effective in the lungs and in the kidneys in helping to regulate body ph.
What are the 3 major buffer systems?
The three major buffer systems of our body are carbonic acid bicarbonate buffer system, phosphate buffer system and protein buffer system.
How does the bicarbonate buffer system regulate pH?
The buffer that maintains the pH of human blood involves carbonic acid (H2CO3), bicarbonate ion (HCO3–), and carbon dioxide (CO2). When bicarbonate ions combine with free hydrogen ions and become carbonic acid, hydrogen ions are removed, moderating pH changes.What is the function of the carbonic acid bicarbonate buffer in blood quizlet?
By altering the reabsorption and excretion of hydrogen ions and bicarbonate ions, the kidneys control the pH of body fluids. The bicarbonate buffer system is one of the chemical buffer systems of the body.
What is the most common buffer system in the body?Bicarbonate buffer is the most important buffer system in blood plasma (generally in the extracellular fluid). This buffer consists of weak acid H2CO3 (pK1 = 6,1) and conjugated base HCO3– (bicarbonate).
Article first time published onWhat is the most important buffer for humans?
The body’s chemical buffer system consists of three individual buffers out of which the carbonic acid bicarbonate buffer is the most important. Cellular respiration produces carbon dioxide as a waste product. This is immediately converted to bicarbonate ion in the blood.
How does a buffer prevent pH from changing?
Buffers are solutions that resist changes in pH, upon addition of small amounts of acid or base. The can do this because they contain an acidic component, HA, to neutralize OH- ions, and a basic component, A-, to neutralize H+ ions.
Which systems regulate the bicarbonate buffer system?
The level of bicarbonate in the blood is controlled through the renal system, where bicarbonate ions in the renal filtrate are conserved and passed back into the blood. However, the bicarbonate buffer is the primary buffering system of the IF surrounding the cells in tissues throughout the body.
Why is buffer system important in the body?
Buffering in blood is crucial to our survival. The pH of blood must be kept constant for normal body functions to work. If blood becomes too acidic, or too basic, then enzymes and proteins are unable to function.
What are the components of bicarbonate buffer system?
The body’s chemical buffer system consists of three individual buffers: the carbonate/carbonic acid buffer, the phosphate buffer and the buffering of plasma proteins.
What is the main buffer system of the extracellular fluid ECF )?
Bicarbonate/carbonic acid represents the most important buffer system of blood and ECF.
Which buffer system S is are not important urine buffers?
The phosphate buffer system is NOT an important blood buffer as its concentration is too low. The concentration of phosphate in the blood is so low that it is quantitatively unimportant. Phosphates are important buffers intracellularly and in urine where their concentration is higher.
Which of the following buffer systems is an important buffer in ICF and in urine?
The phosphate buffer system does not have a role in regulating ECF pH, but it is a very effective buffer system in urine and the intracellular fluid (ICF). The protein buffer system is an abundant and powerful means of regulating pH inside cells, and has a limited role in buffering the blood plasma.
Which of the following describes the primary role of the carbonic acid-bicarbonate buffer system?
The primary role of the carbonic acid-bicarbonate buffer system is to (b.) buffer carbonic acid formed by carbon dioxide.
How would the bicarbonate buffer system work if sodium hydroxide were added to a solution?
How would the bicarbonate buffer system work if sodium hydroxide were added to a solution? A hydrogen on carbonic acid would dissociate and join the hydroxyl group on the base to form water and sodium bicarbonate. a strong base (sodium hydroxide) would be converted to a weak base (sodium bicarbonate).
Why is bicarbonate the best buffer?
The bicarbonate buffering system is important in the body because it: manages the acid and base imbalances produced by both normal and abnormal physiology. assists in the handling of carbon dioxide, the waste product of cellular respiration.
What is produced when a strong acid reacts with the bicarbonate buffer system in the human body?
The hydrogen ions combine with bicarbonate, forming carbonic acid, which dissociates into CO2 gas and water.
Why does bicarbonate increase pH?
If bicarbonate is reabsorbed and/or acid is secreted into the urine, the pH becomes more alkaline (increases). When bicarbonate is not reabsorbed or acid is not excreted into the urine, pH becomes more acidic (decreases).
How do you know if a buffer is effective?
A buffer is most effective when the amounts of acid and conjugate base are approximately equal. As a general rule of thumb, the relative amounts of acid and base should not differ by more than tenfold.
What does low pH mean?
A lower pH means that your blood is more acidic, while a higher pH means that your blood is more basic. The pH of your blood should be around 7.4. … Alkalosis is characterized by a pH level of 7.45 or higher.
How do the kidneys reabsorb bicarbonate?
About 85 to 90% of the filtered bicarbonate is reabsorbed in the proximal tubule and the rest is reabsorbed by the intercalated cells of the distal tubule and collecting ducts.