What is systemic alkalosis

Sophia Edwards
Published Mar 20, 2026
Alkalosis is excessive blood alkalinity caused by an overabundance of bicarbonate in the blood or a loss of acid from the blood (metabolic alkalosis
What do you mean by systemic alkalosis?
Alkalosis occurs when your body has too many bases. It can occur due to decreased blood levels of carbon dioxide, which is an acid. It can also occur due to increased blood levels of bicarbonate, which is a base. This condition may also be related to other underlying health issues such as low potassium, or hypokalemia.
What is the most common cause of alkalosis?
The most common causes are volume depletion (particularly when involving loss of gastric acid and chloride (Cl) due to recurrent vomiting or nasogastric suction) and diuretic use. Metabolic alkalosis involving loss or excess secretion of Cl is termed chloride-responsive.
What are the symptoms of alkalosis?
- Confusion (can progress to stupor or coma)
- Hand tremor.
- Lightheadedness.
- Muscle twitching.
- Nausea, vomiting.
- Numbness or tingling in the face, hands, or feet.
- Prolonged muscle spasms (tetany)
What happens to the body during alkalosis?
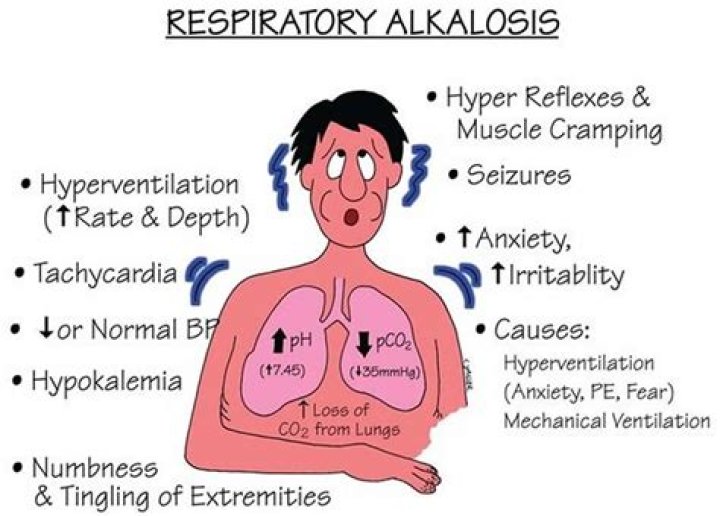
Respiratory alkalosis occurs when you are breathing too fast or too deeply causing your lungs to get rid of too much carbon dioxide. This causes the carbon dioxide levels in the blood to decrease and the blood then becomes alkaline.
How is alkalosis diagnosis?
Diagnosis of Alkalosis A doctor evaluates a person’s acid-base balance by measuring the pH, and levels of carbon dioxide (an acid) and bicarbonate (a base) in the blood. To learn more about the cause of the alkalosis, doctors also measure levels of electrolytes in samples of blood and urine.
How does pH change during alkalosis?
Normal human physiological pH is 7.35 to 7.45. A decrease in pH below this range is acidosis, an increase over this range is alkalosis. Metabolic alkalosis is defined as a disease state where the body’s pH is elevated to greater than 7.45 secondary to some metabolic process.
Is alkalosis fatal?
A person who has a blood pH above 7.45 is considered to be in alkalosis, and a pH above 7.8 is fatal. Some symptoms of alkalosis include cognitive impairment (which can progress to unconsciousness), tingling or numbness in the extremities, muscle twitching and spasm, and nausea and vomiting.What is the main clinical effect of systemic alkalosis?
Metabolic alkalosis can have central nervous system manifestations ranging from confusion to coma, peripheral neuropathic symptoms of tremor, tingling and numbness, muscle weakness and twitching, and arrhythmias, particularly when associated with hypokalemia and hypocalcemia.
What happens if alkalosis is left untreated?Without treatment, you acidosis can lead to shock, coma or even death. Metabolic alkalosis, on the other hand, can cause irritability, muscle cramps and twitches. If left untreated, you can experience long-term muscle spasms.
Article first time published onHow does alkalosis affect potassium?
Alkalosis has the opposite effects, often leading to hypokalemia. Potassium disorders also influence acid-base homeostasis. Potassium depletion causes increased H(+) secretion, ammoniagenesis and H-K-ATPase activity. Hyperkalemia decreases ammoniagenesis and NH4(+) transport in the thick ascending limb.
What is the treatment for alkalosis and acidosis?
Metabolic alkalosis is treated by replacing water and mineral salts such as sodium and potassium (electrolytes) and correcting the cause. Respiratory alkalosis is treated by correcting the cause.
How do you reverse respiratory alkalosis?
- Breathe into a paper bag. Fill the paper bag with carbon dioxide by exhaling into it. …
- Get reassurance. The symptoms of respiratory alkalosis can be frightening. …
- Restrict oxygen intake into the lungs. To do this, try breathing while pursing the lips or breathing through one nostril.
Which condition may result in respiratory alkalosis?
Any lung disease that leads to shortness of breath can also cause respiratory alkalosis (such as pulmonary embolism and asthma).
What causes respiratory alkalosis?
When you breathe faster, the lower carbon dioxide level in your blood can lead to respiratory alkalosis. Respiratory alkalosis is usually caused by over-breathing (called hyperventilation) that occurs when you breathe very deeply or rapidly. Causes of hyperventilation include: Anxiety or panic.
What is a healthy pH level?
A normal blood pH level is 7.40 on a scale of 0 to 14, where 0 is the most acidic and 14 is the most basic. This value can vary slightly in either direction. If the lungs or kidneys are malfunctioning, your blood’s pH level can become imbalanced.
What pH level is harmful to humans?
When measured on the pH scale, anything above a 7 on the pH scale is considered a base. When the pH levels reach or exceed 12.5 pH, it then becomes hazardous. As with all hazardous wastes, when in contact with human or environmental health, it can be very dangerous.
How does the body regulate pH?
- Reduce or Eliminate harmful acidic foods from your diet. Sugar. …
- Choose healthier acidic foods. …
- Increase alkaline foods to 70% of your diet. …
- Incorporate alkalizing lifestyle choices.
When is metabolic alkalosis treated?
Primary hyperaldosteronism Metabolic alkalosis is corrected with the aldosterone antagonist spironolactone or with other potassium-sparing diuretics (eg, amiloride, triamterene). If the cause of primary hyperaldosteronism is an adrenal adenoma or carcinoma, surgical removal of the tumor should correct the alkalosis.
Can you have both respiratory and metabolic alkalosis?
Too much acid and too little base in the blood causes an acidotic pH level, and the result is combined respiratory and metabolic acidosis. For combined respiratory and metabolic alkalosis, the PaCO2 level is decreased and the HCO3 level is elevated.
How do I raise my blood pH?
For example, your doctor may give you sodium bicarbonate (baking soda) to raise the pH of your blood. This can be done either by mouth or in an intravenous (IV) drip. The treatment for other types of acidosis can involve treating their cause.
Can disease live in an alkaline body?
Disease cannot survive in an alkaline state; however, in a low oxygen/low pH (acidic) state, viruses, bacteria, yeast, mold, fungus, Candida and Cancer cells all thrive.
Does apple cider vinegar make your body alkaline?
Apple Cider Vinegar will balance your body’s pH levels But unlike others, apple cider vinegar is the one with an alkalizing effect. Though acidic, ACV promotes an alkaline environment inside your body.
What happens when you have too much acid in your body?
An acidic pH may result in weight problems such as diabetes and obesity. When our body is too acidic, we suffer from a condition known as Insulin Sensitivity. This forces excessive insulin to be produced. As a result, the body is flooded with so much insulin that it diligently converts every calorie into fat.
Why does alkalosis cause low potassium?
A frequently cited mechanism for these findings is that acidosis causes potassium to move from cells to extracellular fluid (plasma) in exchange for hydrogen ions, and alkalosis causes the reverse movement of potassium and hydrogen ions.
What is the most common cause of hypokalemia?
Low potassium (hypokalemia) has many causes. The most common cause is excessive potassium loss in urine due to prescription medications that increase urination. Also known as water pills or diuretics, these types of medications are often prescribed for people who have high blood pressure or heart disease.
Does respiratory alkalosis cause hypokalemia?
Persistent respiratory alkalosis can induce secondary hypocalcemia and hypokalemia that may cause cardiac arrhythmias, conduction abnormalities, and various somatic symptoms such as paresthesia, hyperreflexia, convulsive disorders, muscle spasm and tetany [2].
What medication is used for metabolic alkalosis?
Ammonium chloride (NH4Cl) Ammonium chloride is administered to correct severe metabolic alkalosis related to chloride deficiency. NH4Cl is converted to ammonia and HCl by the liver. By releasing HCl, NH4Cl may help correct metabolic alkalosis.
Is pneumonia respiratory acidosis or alkalosis?
Respiratory alkalosis is commonly found in patients with asthma, pneumonia & pulmonary embolism.
Is COPD respiratory acidosis or alkalosis?
Chronic obstructive pulmonary disease (COPD) is a common group of diseases that are particularly likely to cause respiratory acidosis.
Does asthma cause acidosis or alkalosis?
Respiratory acidosis is a very common acid base disturbance in acute severe asthma and is widely considered to be an ominous finding. Its early recognition and treatment is important and decisive for the final outcome, as it can lead to respiratory failure and arrest if prolonged.