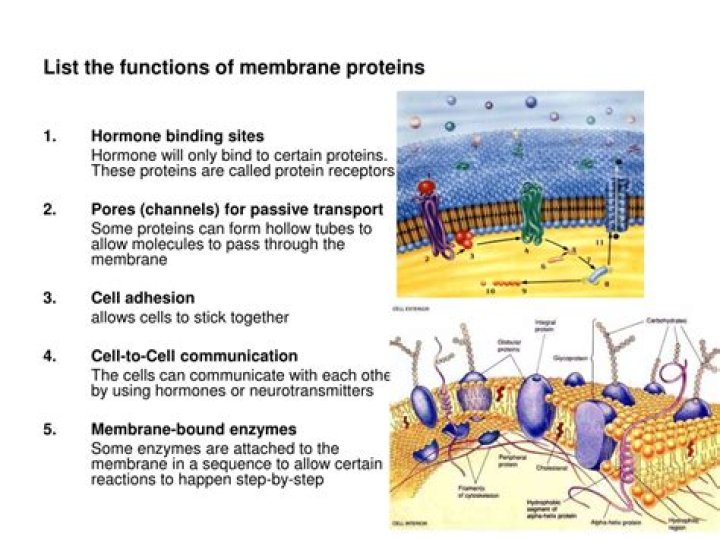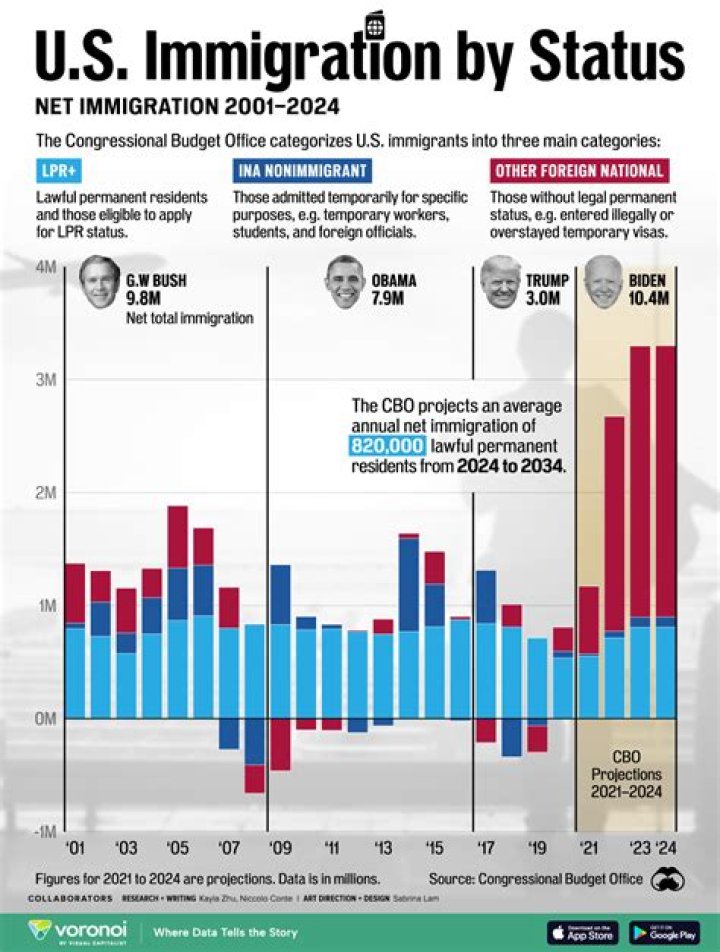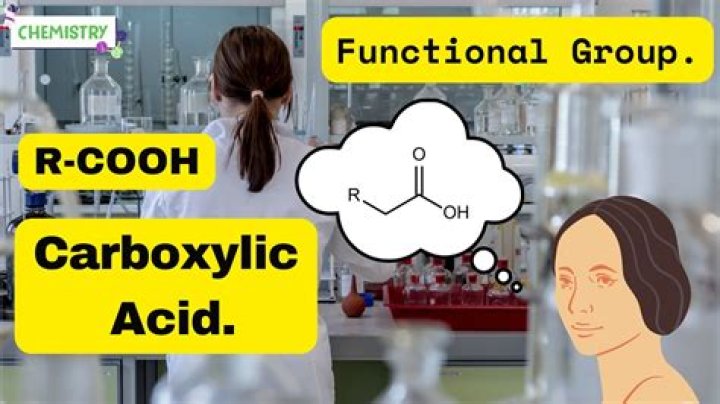
What is Rcooh in chemistry

Mia Morrison
Published Mar 20, 2026
Carboxylic acid (RCOOH; RCO2H): A molecule containing the carboxyl group.
What is the chemical name of RCOOH?
Carboxylic acid (RCOOH; RCO2H): A molecule containing the carboxyl group.
What functional group is Rcoh?
Functional GroupFormulanamingAldehydesRCOH-alKetonesRCOR-oneCarboxylic AcidsRCOOH-oic acidEstersRCOOCRalkyl- -oate
What is Rcoor in chemistry?
Carboxylic acid esters, formula RCOOR′ (R and R′ are any organic combining groups), are commonly prepared by reaction of carboxylic acids and alcohols in the presence of hydrochloric acid or sulfuric acid, a process called esterification.What does the R stand for in R Oh?
Definition. An alcohol molecule has the general formula ROH where R is an alkyl group, O is an oxygen atom and H is a hydrogen atom.
What is a carboxylic acid give an example?
Carboxylic acidpKaAcetic acid (CH3CO2H)4.76Benzoic acid (C6H5CO2H)4.2Formic acid (HCOOH)3.75Chloroacetic acid (CH2ClCO2H)2.86
What is Ester formula?
Esters have the general formula RCOOR′, where R may be a hydrogen atom, an alkyl group, or an aryl group, and R′ may be an alkyl group or an aryl group but not a hydrogen atom. (If it were hydrogen atom, the compound would be a carboxylic acid.) … Esters occur widely in nature.
Is Phenoxide a base?
Sodium phenoxide is a moderately strong base.What is Rcoo?
RCOO-Na+ is formula of soap written everywhere but according to definition, it contains carboxylic acid, not COO only, and Kinross salts if Na is not present. … Generally sodium salts are used, thus formula of soap is written as RCOO-Na+. But it can be RCOO-K+ also.
Is Coo an ester?In a condensation reaction, two molecules join and produce a larger molecule whilst eliminating a small molecule. During esterification this small molecule is water. Esters have characteristic smells and are insoluble in water. The functional group (-COO) is known as the ester link.
Article first time published onWhat compound is Rcoh?
Record InformationDescriptionBelongs to the class of organic compounds known as primary alcohols. Primary alcohols are compounds comprising the primary alcohol functional group, with the general structure RCOH (R=alkyl, aryl).KingdomOrganic compounds
What is aldehyde formula?
The chemical formula for an aldehyde is RCHO. In this formula, R represents a hydrogen atom or carbon/hydrogen chain, CO represents the carbonyl, and H represents the hydrogen attached to the carbonyl chain.
What is the name of Rcoh?
Roh, sometimes spelled Noh, Ro or Lho, is a given name of Sanskrit origins, derived from the male given name Rohit, meaning “rising sun”, or “red horizon”.
What is r in alkyl group?
In organic chemistry, an alkyl substituent is an alkane missing one hydrogen. … Typically an alkyl is a part of a larger molecule. In structural formula, the symbol R is used to designate a generic (unspecified) alkyl group. The smallest alkyl group is methyl, with the formula CH3−.
What is alkyl group?
Definition: An alkyl is a functional group of an organic chemical that contains only carbon and hydrogen atoms, which are arranged in a chain. Examples include methyl CH3 (derived from methane) and butyl C2H5 (derived from butane).
Is ch3 a methyl?
A methyl group is an alkyl derived from methane, containing one carbon atom bonded to three hydrogen atoms — CH3. In formulas, the group is often abbreviated Me. … It is a very stable group in most molecules.
How is ester produced *?
Esters are produced when carboxylic acids are heated with alcohols in the presence of an acid catalyst. The catalyst is usually concentrated sulphuric acid. Dry hydrogen chloride gas is used in some cases, but these tend to involve aromatic esters (ones containing a benzene ring).
What is an ether in chemistry?
ether, any of a class of organic compounds characterized by an oxygen atom bonded to two alkyl or aryl groups. Ethers are similar in structure to alcohols, and both ethers and alcohols are similar in structure to water. … At room temperature, ethers are pleasant-smelling colourless liquids.
What are esters in?
Esters are ubiquitous. Most naturally occurring fats and oils are the fatty acid esters of glycerol. Esters are typically fragrant, and those with low enough molecular weights to be volatile are commonly used as perfumes and are found in essential oils and pheromones.
What Is carboxylic acid used for?
Carboxylic acids and their derivatives are used in the production of polymers, biopolymers, coatings, adhesives, and pharmaceutical drugs. They also can be used as solvents, food additives, antimicrobials, and flavorings.
HOW IS carboxylic acid formed?
The hydrolysis of nitriles, which are organic molecules containing a cyano group, leads to carboxylic acid formation. These hydrolysis reactions can take place in either acidic or basic solutions. The mechanism for these reactions involves the formation of an amide followed by hydrolysis of the amide to the acid.
Why Is carboxylic acid an acid?
The acidity of the carboxylic acids Using the definition of an acid as a “substance which donates protons (hydrogen ions) to other things”, the carboxylic acids are acidic because of the hydrogen in the -COOH group. In solution in water, a hydrogen ion is transferred from the -COOH group to a water molecule.
What type of reaction is RCOOH h2o?
Esterification is the process of combining an organic acid (RCOOH) with an alcohol (ROH) to form an ester (RCOOR) and water; or a chemical reaction resulting in the formation of at least one ester product. Ester is obtained by an esterification reaction of an alcohol and a carboxylic acid.
What is the formula of carboxylate?
Carboxylate salts are salts that have the general formula M(RCOO)n, where M is a metal and n is 1, 2,…; carboxylate esters have the general formula RCOOR′ (or RCO2R′). R and R′ are organic groups; R′ ≠ H.
Why is carboxylate ion stable?
A carboxylate ion is much more stable than the corresponding alkoxide ion because of the existence of resonance structures for the carboxylate ion which disperse its negative charge. … Resonance always stabilizes a molecule or ion, even if charge is not involved.
Is phenoxide a weak base?
The phenoxide ion is a WEAKER base than alkoxide ion, because the phenoxide ion is resonance stabilized, and requires less solvation.
Why is phenoxide more stable?
Phenoxide ion is more stable than phenol due to the delocalisation of negative charge in phenoxide ion. Explanation: Due to delocalisation of negative charge, phenoxide ion is more stable than phenol.
Is phenoxide ion acidic?
When phenoxide is deprotonated, it causes less solvent ORDER, and its acidity is entropically favoured. As a result, rather than being an enthalpy phenomenon, this is an entropy phenomenon. The ability of phenols to lose hydrogen ions and produce phenoxide ions gives them their acidity.
What is the simplest ester?
Methyl formate, also called methyl methanoate, is the methyl ester of formic acid. The simplest example of an ester, it is a colorless liquid with an ethereal odour, high vapor pressure, and low surface tension.
Is methyl acetate an ester?
Methyl acetate is an acetate ester resulting from the formal condensation of acetic acid with methanol. A low-boiling (57 ℃) colourless, flammable liquid, it is used as a solvent for many resins and oils.
Can a secondary alcohol form an ester?
An unexpected new reaction pathway, from which the secondary alcohols can be directly oxidized to corresponding esters, was found.