What is M in Chapman cycle

Sophia Edwards
Published Mar 19, 2026
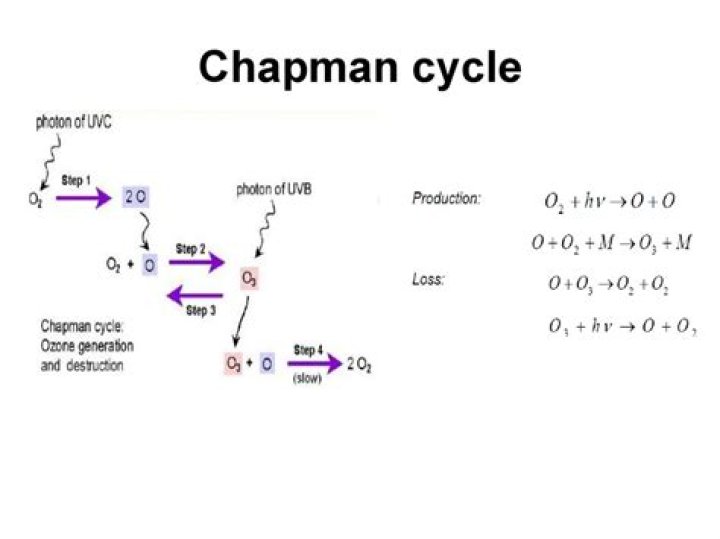
These reactions, proposed by Sydney Chapman in 1930, explain the presence of ozone in the stratosphere. … The �M� in this reaction is any third molecule: M absorbs the heat from this reaction. The increasing temperature profile of the stratosphere results from this reaction.
How is ozone converted to oxygen?
Ozone is extremely valuable since it absorbs a range of ultraviolet energy. When an ozone molecule absorbs even low-energy ultraviolet radiation, it splits into an ordinary oxygen molecule and a free oxygen atom. Usually this free oxygen atom quickly re-joins with an oxygen molecule to form another ozone molecule.
What is the first step in the Chapman cycle?
The Chapman Cycle The ozone layer is created when ultraviolet rays react with oxygen molecules (O2) to create ozone (O3) and atomic oxygen (O). This process is called the Chapman cycle. Step 1: An oxygen molecules is photolyzed by solar radiation, creating two oxygen radicals: hν+O2→2O.
What is Chapman mechanism?
A series of reactions, first proposed by Sidney Chapman in the 1930s, to explain the presence of the ozone layer in the earth’s stratosphere. A theory of upper atmospheric ozone. …Which form of oxygen is involved in the ozone oxygen cycle?
Ozone Cycle The ozone-oxygen cycle involves three forms of oxygen: Atomic oxygen (O) Diatomic oxygen, or oxygen gas (O2) Ozone (O3)
What is oxygen used for?
Common uses of oxygen include production of steel, plastics and textiles, brazing, welding and cutting of steels and other metals, rocket propellant, oxygen therapy, and life support systems in aircraft, submarines, spaceflight and diving.
What is nascent oxygen how it is represented?
Answer: A very reactive and very unstable monoatomic oxygen is considered nascent oxygen and it is represented as [O]. It is a monatomic element. It has a high tendency to react to obtain an oxygen molecule (O2) with the other [O] nascent oxygen.
What are the chemical reactions of ozone in the stratosphere the Chapman cycle?
breaks apart an ozone molecule into a diatomic oxygen molecule and a free oxygen atom. 3. The basic Chapman Cycle for ozone loss is O3 + O –> O2 + O2. Here an ozone molecule reacts with a free oxygen atom to create two diatomic oxygen molecules.Is oxygen endothermic or exothermic?
The reaction between hydrogen and oxygen is exothermic– it releases energy to the surroundings: hydrogen + oxygen → water 2H2 + O2 → 2H2O Fuel cells use the reaction between a fuel and oxygen to produce electrical energy.
How is the ozone layer formed in the stratosphere?Stratospheric ozone is formed naturally through the interaction of solar ultraviolet (UV) radiation with molecular oxygen (O2). The “ozone layer,” approximately 6 through 30 miles above the Earth’s surface, reduces the amount of harmful UV radiation reaching the Earth’s surface.
Article first time published onWhat is Montreal Protocol related to?
The Montreal Protocol, finalized in 1987, is a global agreement to protect the stratospheric ozone layer by phasing out the production and consumption of ozone-depleting substances (ODS). … The United States ratified the Montreal Protocol in 1988 and has joined four subsequent amendments.
What is meant by odd oxygen?
The sum of the concentrations of oxygen atoms and ozone (O3). In the lower atmosphere, the concentration of O3 greatly exceeds that of O, and the odd oxygen level can be approximated by the O3 concentration. Terms O. …
What happens to ozone formation during the night no sun )?
After sunset, when no more sunlight initiates ozone formation, ozone concentrations fall as ozone reacts with other chemicals and rapidly settles onto various surfaces. NOx and VOC concentrations drop as they too participate in other reactions.
Do we still have a hole in the ozone layer?
What we call the “ozone hole” is a thinning of the ozone layer in the stratosphere above Antarctica that develops every September. … The ozone hole reached its maximum extent on October 7, 2021, as calculated by the NASA Ozone Watch team. NASA and NOAA monitor the ozone hole via complementary instrumental methods.
What is meant by the ozone hole?
The term ‘ozone hole’ refers to the depletion of the protective ozone layer in the upper atmosphere (stratosphere) over Earth’s polar regions. … Polar stratospheric clouds create the conditions for drastic ozone destruction, providing a surface for chlorine to change into ozone-destroying form.
Where is the ozone hole?
What we call the ozone hole is a thinning of the protective ozone layer in the stratosphere (the upper layer of Earth’s atmosphere) above Antarctica that begins every September.
What type of UV radiation is responsible for breaking down oxygen molecules to make single oxygen atoms?
UV-C splits oxygen molecules into oxygen atoms. These single atoms then react with other oxygen molecules to produce ozone. So, these reactions increase the amount of ozone in the stratosphere.
Which form of the oxygen changes in the presence of UV light and to what form?
In the stratosphere, the ozone is formed when the ultraviolet rays from the sun splits the oxygen molecule into two oxygen atoms. The these oxygen atom combine with the molecular oxygen(O2) and produce O3 which is called ozone. Therefore O2 changes to O3 in the presence of UV light.
Is ozone an isotope of oxygen?
Ozone was discovered by Christian Friedrich Schönbein in 1840. It is a triatomic molecule and an allotrope of oxygen. Ozone can be very helpful to living organisms because it protects them from harmful ultraviolet light. Ozone is a allotrope of oxygen comprised of three oxygen atoms.
What is nascent oxygen and how is it produced?
Nascent oxygen is produced by a strong oxidizing agent like potassium permanganate and potassium dichromate. When these strong oxidizing agents react with ethanol then there is the formation of nascent oxygen. It reacts with a huge number of atoms and molecules.
How do you make nascent oxygen?
It is the atomic form of oxygen [O] it does not exist independently as a molecule. In chemical reactions, nascent oxygen is produced from strong oxidizing agents like KMnO4 and K2Cr2O7. When potassium permanganate reacts with ethanol, it releases nascent oxygen.
What is called nascent oxygen?
A monoatomic oxygen which is very reactive and very unstable is called nascent oxygen. It is said to be monoatomic. It is written as [O]. It has a high tendency to react with other nascent oxygen to form an oxygen molecule (O2).
Where does oxygen come from?
At least half of Earth’s oxygen comes from the ocean. Scientists estimate that 50-80% of the oxygen production on Earth comes from the ocean. The majority of this production is from oceanic plankton — drifting plants, algae, and some bacteria that can photosynthesize.
How do we get oxygen?
There are two key methods used to obtain oxygen gas. The first is by the distillation of liquid air. The second is to pass clean, dry air through a zeolite that absorbs nitrogen and leaves oxygen. A newer method, which gives oxygen of a higher purity, is to pass air over a partially permeable ceramic membrane.
Where is oxygen commonly found?
Oxygen occurs mainly as an element in the atmosphere. It makes up 20.948 percent of the atmosphere. It also occurs in oceans, lakes, rivers, and ice caps in the form of water. Nearly 89 percent of the weight of water is oxygen.
How flame is produced?
Fire is the rapid oxidation of a material (the fuel) in the exothermic chemical process of combustion, releasing heat, light, and various reaction products. … At a certain point in the combustion reaction, called the ignition point, flames are produced.
Is H2O Endo or Exo?
H2O(l) → H2O(s) Exothermic; energy is released when bonds are being made between water molecules in the phase change.
What do combustion reactions produce?
Most combustion reactions produce carbon dioxide and water, so these chemicals are written as the products on the right of the equation. Charcoal is a fuel that contains carbon atoms but no hydrogen atoms.
How many steps are in the formation of ozone?
Figure Q2-1. Ozone is naturally produced in the stratosphere in a two-step process. In the first step, ultraviolet sunlight breaks apart an oxygen molecule to form two separate oxygen atoms. In the second step, each atom then undergoes a binding collision with another oxygen molecule to form an ozone molecule.
How are individual atoms of oxygen formed in the stratosphere?
How are individual atoms of oxygen formed in the stratosphere? … Individual oxygen atoms are formed when UV rays strike oxygen molecules.
How is ozone formed class 10th?
CBSE NCERT Notes Class 10 Biology Our Environment. Ozone is a molecule formed by three atoms of oxygen. … The UV radiations split apart some molecular oxygen (O2) into free oxygen (O) atoms which then combine with the molecular oxygen (O2) to form ozone (O3).