What is K in Amontons law

Victoria Simmons
Published Mar 01, 2026
Amonton’s Law states that the pressure of an ideal gas varies directly with the absolute temperature when the volume of the sample is held constant. P 1/T 1 = P 2/T 2 or P/T = k, where k = nR/V.
Does Charles law have to be in K?
and is called Charles’ law. For this law to be valid, the pressure must be held constant, and the temperature must be expressed on the absolute temperature or Kelvin scale . Therefore, at normal pressure, water freezes at 273.15 K (0°C), which is called the freezing point, and boils at 373.15 K (100°C).
What is the value of the constant k in Boyle's law?
Simply put, Boyle’s states that for a gas at constant temperature, pressure multiplied by volume is a constant value. The equation for this is PV = k, where k is a constant. At a constant temperature, if you increase the pressure of a gas, its volume decreases. If you increase its volume, the pressure decreases.
What is the constant k obtained by dividing the volume by the temperature?
Temperature (K)Volume (mL)3001200.405002000.4010004000.40Which of the following represents Avogadro's law?
Equal volumes of all gases under the same conditions of temperature and pressure contain equal number of atoms.
How does Charles Law keep pressure constant?
To test Charles’ law we need to keep the pressure constant. This means after changing the temperature of a sample of gas we can then adjust the volume of the system until the pressure is the same as it was initially. This can be done using an adjustable cylinder (like a large syringe) attached to a pressure gauge.
Why do we use Kelvin in gas laws?
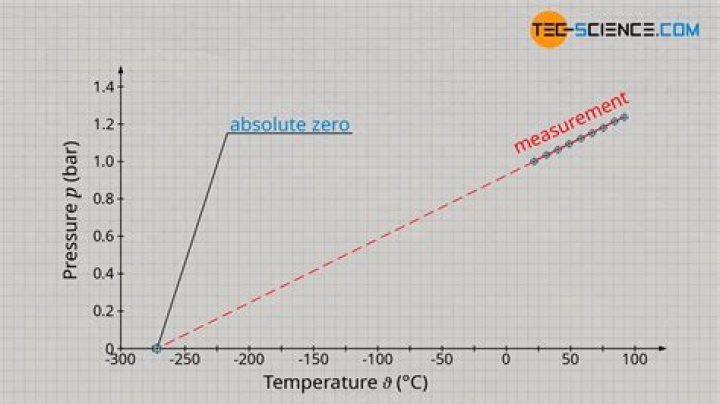
The Kelvin scale is used in gas law problems because the pressure and volume of a gas depend on the kinetic energy or motion of the particles. The Kelvin scale is proportional to the KE of the particles… that is, 0 K (absolute zero) means 0 kinetic energy. 0 °C is simply the freezing point of water.
Is Charles Law in mL or L?
Note: Charles’s Law uses kelvin for temperature units, while the volume units can be any volume unit (mL, L, etc.)What is Charles Law in simple terms?
Charles’s law, a statement that the volume occupied by a fixed amount of gas is directly proportional to its absolute temperature, if the pressure remains constant. … It is a special case of the general gas law and can be derived from the kinetic theory of gases under the assumption of a perfect (ideal) gas.
How is formula derived Charles Law?Charles Law states that the volume of a given mass of a gas is directly proportional to its Kevin temperature at constant pressure. In mathematical terms, the relationship between temperature and volume is expressed as V1/T1=V2/T2.
Article first time published onHow do you verify Charles Law?
The equation for Charles’s law can be expressed as V1/T1=V2/T2. In other words, if a balloon is filled with air, it will shrink if cooled and expand if heated. This happens because the air inside the balloon, which is a gas, takes up a smaller volume when it is cool, and takes up a larger volume when it is heated.
Which of the following needs to remain constant when using the Charles Law?
Temperature and volume. … The volume of an ideal gas is exactly proportional to the absolute temperature under constant pressure, according to Charles law. When the pressure imposed on a sample of a dry gas is maintained constant, the Kelvin temperature and volume will be in direct proportion, according to the law.
What is the value of k in gas?
Since the 2019 redefinition of SI base units, both NA and k are defined with exact numerical values when expressed in SI units. As a consequence, the SI value of the molar gas constant is exactly 8.31446261815324 J⋅K−1⋅mol−1.
Which variable is constant in Boyle's law?
Robert Boyle’s observations are summed up in Boyle’s law, which states that for a given mass of gas at constant temperature, the volume of a gas varies inversely with pressure. Because of the inverse relationship, the product of the two quantities, pressure and volume, is constant.
What is the value of k in ideal gas?
The ideal gas law can be written in terms of Avogadro’s number as PV = NkT, where k, called the Boltzmann’s constant, has the value k = 1.38 × 10 −23 J/K. One mole of any gas at standard temperature and pressure (STP) occupies a standard volume of 22.4 liters.
Which of the following is Avogadro constant?
Avogadro’s number, number of units in one mole of any substance (defined as its molecular weight in grams), equal to 6.02214076 × 1023.
Which answer choice represents Avogadro's number?
Avogadro’s number represents the number of representative particles in a mole of a substance. Avogadro’s number is 6.02 x 1023.
Why is Charles Law in Kelvin?
French physicist Jacques Charles (1746-1823) studied the effect of temperature on the volume of a gas at constant pressure. … The absolute temperature is temperature measured with the Kelvin scale. The Kelvin scale must be used because zero on the Kelvin scale corresponds to a complete stop of molecular motion.
What is K temperature?
kelvinUnit systemSI base unitUnit ofTemperatureSymbolKNamed afterWilliam Thomson, 1st Baron Kelvin
Why is Kelvin used in gas laws instead of Celsius?
We chose 0 as the original base for the Celsius scale because that is the freezing point of water not because of anything to do with gas laws. So that is an arbitrary point that happens to give a more complicated gas law. Hence the Kelvin scale where the zero relates to the gas law and makes it simpler.
What happens to a gas at 0 K?
According to the physical meaning of temperature, the temperature of a gas is determined by the chaotic movement of its particles – the colder the gas, the slower the particles. At zero kelvin (minus 273 degrees Celsius) the particles stop moving and all disorder disappears.
What is V1 in Charles Law?
V1 = 10 liters. T1 = 27° C + 273 ° K = 300 ° K. (temperatures used in Charles Law must be in Absolute Scale)
What is Charles formula?
Definition of Charles Law Formula is, “When the pressure on a sample of a dry gas is held constant, the Kelvin temperature and therefore the volume is going to be in direct proportion.” The equation of the law is PV = k.
What are the variables in Charles Law?
Since pressure is kept constant, the only variable that is manipulated is temperature. This means that we can use Charles’s law in order to compare volume and temperature. Since volume and temperature are on opposite sides of the ideal gas law, they are directly proportional to one another.
What is Charles's law used for?
Charles’ Law is an experimental gas law that describes how gases tend to expand when heated. The law states that if a quantity of gas is held at a constant pressure, there is a direct relationship between its volume and the temperature, as measured in degrees Kelvin. Think of it this way.
What is the relationship of volume and temperature at constant pressure?
The volume of a gas is directly proportional to its temperature when pressure is constant. The ratio of volume to temperature is constant when pressure is constant. This relationship is known as Charles’ law or Gay-Lussac’s law .