What is epoxide formation

Emily Dawson
Published Feb 28, 2026
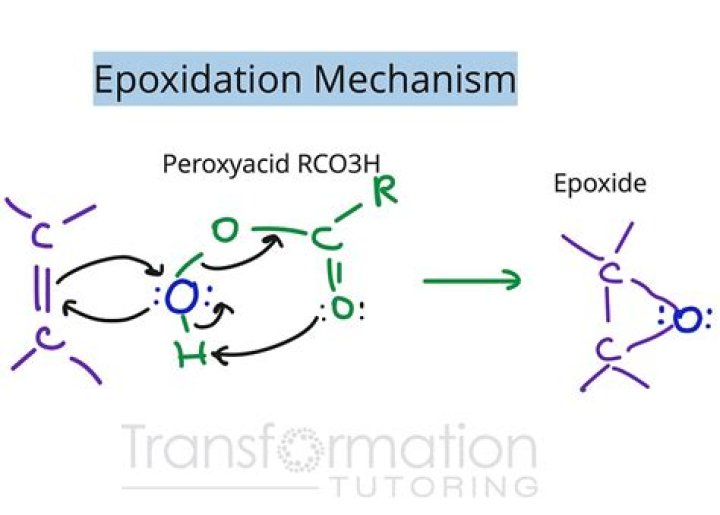
Epoxidation is the chemical reaction which converts the carbon–carbon double bond into oxiranes (epoxides), using a variety of reagents including air oxidation, hypochlorous acid, hydrogen peroxide, and organic peracid (Fettes, 1964).
How are epoxide named?
The root name is based on the longest chain with the two C-O bonds attached. The epoxide prefix is inserted prior to the root name along with both locants e.g. 1,2-epoxypropane. Both locants are included since this method is also used for naming other cyclic ethers.
What is oxirane write the structure?
License. Ethylene oxide is an organic compound with the formula C2H4O. It is a cyclic ether and the simplest epoxide: a three-membered ring consisting of one oxygen atom and two carbon atoms. Ethylene oxide is a colorless and flammable gas with a faintly sweet odor.
What are the reaction of epoxide?
Epoxides are much more reactive than simple ethers due to ring strain. Nucleophiles attack the electrophilic C of the C-O bond causing it to break, resulting in ring opening. Opening the ring relieves the ring strain. The products are typically 2-substituted alcohols.How is epoxide formed?
Aside from ethylene oxide, most epoxides are generated by treating alkenes with peroxide-containing reagents, which donate a single oxygen atom. Safety considerations weigh on these reactions because organic peroxides are prone to spontaneous decomposition or even combustion.
What is the general formula for ether?
Ethers are a class of organic compounds that contain an ether group—an oxygen atom connected to two alkyl or aryl groups. They have the general formula R–O–R′, where R and R′ represent the alkyl or aryl groups.
How do you make an epoxide?
The most important and simplest epoxide is ethylene oxide which is prepared on an industrial scale by catalytic oxidation of ethylene by air. Ethylene oxide is used as an important chemical feedstock in the manufacturing of ethylene glycol, which is used as antifreeze, liquid coolant and solvent.
How do you write the Iupac name for ether?
Systematic (IUPAC) names for ethers use the more complex group as the root name, with the oxygen atom and the smaller group named as an alkoxy substituent. Examples given above are ethoxyethane (diethyl ether), methoxyethane (methyl ethyl ether), 2-methoxy-2-methylpropane (MTBE), and phenoxybenzene (diphenyl ether).How is Tetrahydropyran formed?
The reaction of tertiary 1,4- and 1,5-diols with cerium ammonium nitrate at room temperature gives tetrahydrofuran and tetrahydropyran derivatives in high yield and stereoselectivity. Various fragrant compounds have been synthesized using this method.
What is epoxide give example?epoxide, cyclic ether with a three-membered ring. … Epoxides are easily opened, under acidic or basic conditions, to give a variety of products with useful functional groups. For example, the acid- or base-catalyzed hydrolysis of propylene oxide gives propylene glycol.
Article first time published onWhat is the ring-opening of epoxide?
Epoxide Ring-Opening with Strong Nucleophiles The ring-opening reactions of epoxides occur via SN2 mechanism where the oxygen of the epoxide is the leaving group.
How does epoxide react with ammonia?
Ammonia and amines react with epoxides with the same stereospecificity as anionic nucleophiles. Draw a sawhorse or Newman projection formula for the product of the reaction shown, clearly showing the stereochemistry at both chirality centers.
What is C2H4O called?
Ethylene oxide | C2H4O – PubChem.
Is oxirane an epoxide?
Introduction. The oxirane (epoxide) functional group is arguably the most synthetically useful small-ring heterocycle due to its ease of synthesis and wide-ranging ring-opening reactions, which usually occur with predictable regioselectivity and stereospecificity.
Which reagent can convert epoxide ring into alcohol?
The reaction of epoxides with TMSCN (an organic-soluble and easier-to-handle substitute to traditional cyanide reagents such as KCN) and catalytic ZnI2 gives 1,2-isocyano alcohols.
What is reaction carboxylic acid?
Carboxylic acids react with Thionyl Chloride (SOCl2) to form acid chlorides. Mechanism. Carboxylic acids can react with alcohols to form esters in a process called Fischer esterification. Basic Reaction. Going from reactants to products simplified.
What is a epoxy group?
epoxy group ĕp´ŏksē [key], in chemistry, functional group that consists of an oxygen atom joined by single bonds to two adjacent carbon atoms, thus forming the three-membered epoxide ring. It is the functional group of epoxides.
What is the general formula of alkyl?
An acyclic alkyl has the general formula of CnH2n+1. A cycloalkyl is derived from a cycloalkane by removal of a hydrogen atom from a ring and has the general formula CnH2n-1. Typically an alkyl is a part of a larger molecule. In structural formula, the symbol R is used to designate a generic (unspecified) alkyl group.
What is Ketone formula?
License. In chemistry, a ketone is a functional group with the structure R2C=O, where R can be a variety of carbon-containing substituents. Ketones contain a carbonyl group (a carbon-oxygen double bond). The simplest ketone is acetone (R = R’ = methyl), with the formula CH3C(O)CH3.
What Is carboxylic acid formula?
What is Carboxylic Acid Formula? The general molecular formula for carboxylic acid is CnH2n+1COOH. Carboxylic acids are nothing but organic compounds in which the carbon atom is bonded with an oxygen atom in the form of a double bond.
What is DHP in organic chemistry?
3,4-Dihydropyran (DHP) is a heterocyclic compound with the formula C5H8O. … The isomeric 3,6-dihydropyran has a methylene separating the double bond and oxygen. DHP is used for protecting group for alcohols. It is a colorless liquid.
What is the structure of tetrahydropyran?
Tetrahydropyran (THP) is the organic compound consisting of a saturated six-membered ring containing five carbon atoms and one oxygen atom. It is named by reference to pyran, which contains two double bonds, and may be produced from it by adding four hydrogens.
Is tetrahydropyran polar?
Information on this page: Normal alkane RI, non-polar column, custom temperature program. References. Notes.
How do you name amines?
The amine group is located by the position number. Groups that are attached to the nitrogen atom are located using “N” as the position number. More complex primary amines are named with —NH2 as the amino substituent. Aromatic amines: named as derivatives of the parent compound aniline.
What is alkyl group?
Definition: An alkyl is a functional group of an organic chemical that contains only carbon and hydrogen atoms, which are arranged in a chain. Examples include methyl CH3 (derived from methane) and butyl C2H5 (derived from butane).
What is the Iupac name of Easter?
Condensed Structural FormulaCommon NameIUPAC NameCH3CH2CH2COOCH(CH3)2isopropyl butyrateisopropyl butanoateethyl benzoateethyl benzoate
Where is ethylene found?
Natural sources of ethylene include both natural gas and petroleum; it is also a naturally occurring hormone in plants, in which it inhibits growth and promotes leaf fall, and in fruits, in which it promotes ripening. Ethylene is an important industrial organic chemical.
Is an epoxide aromatic?
Eukaryotes produce aromatic epoxides, followed by the formation of trans-dihydrodiols (35). … In addition, prokaryotic dioxygenases have not been shown to catalyze initiation reactions of aromatic ring structures via formation of aromatic epoxide intermediates (44).
How do you open an epoxide?
Epoxides can also be opened by other anhydrous acids (HX) to form a trans halohydrin. When both the epoxide carbons are either primary or secondary the halogen anion will attack the less substituted carbon and an SN2 like reaction.
What reagent breaks an epoxide?
Epoxides can also be opened by other anhydrous acids (HX) to form a trans halohydrin. When both the epoxide carbons are either primary or secondary the halogen anion will attack the less substituted carbon and an S N2 like reaction.
Is epoxide a racemic?
The racemic epoxide is designated (2R*,3R*)-3-ethyl-2-methyloxirane. When (Z)-2-pentene undergoes the chlorohydrin reaction, there are likewise two possible racemic chlorohydrins formed, which upon base treatment, lead to a single, racemic epoxide. The formation of epoxides by the halohydrin route is stereospecific.