What is boat conformation

Sophia Edwards
Published Feb 18, 2026
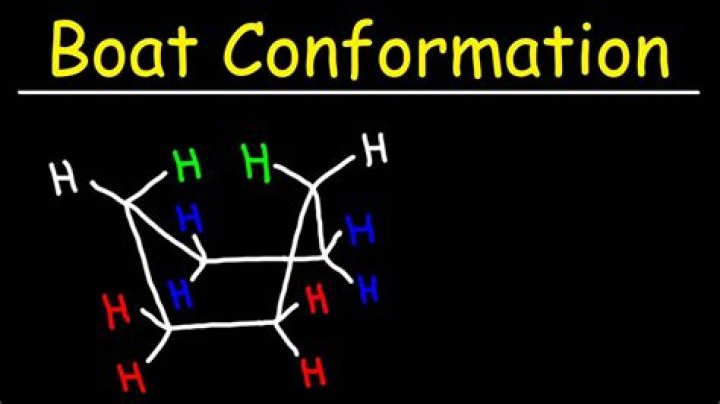
Boat conformation: A six-membered ring conformation in which atoms 1, 2, 4, and 5 are coplanar, and atoms 3 and 6 are bent out of the plane in the same direction. … Note that the bow and stern (front and back ends) of the boat point in the same direction (both up).
How do you draw a cyclohexane boat?
- Draw two horizontal parallel lines. These will form the sides of the boat.
- Add two lines pointing up to make the bow of the boat.
- Add two lines pointing up at the other end to make the stern. And your boat is complete! Ernest Z. · 1 · Jun 28 2015.
Which of the following are conformations of cyclohexane?
The half-chair state (D, in figure below) is the key transition state in the interconversion between the chair and twist-boat conformations. The half-chair has C2 symmetry. The interconversion between the two chair conformations involves the following sequence: chair → half-chair → twist-boat → half-chair′ → chair′.
Is boat or chair conformation more stable?
The chair conformation is more stable than the boat conformation. The boat conformation can sometimes be more stable than it is usually, by a slight rotation in the C-C bonds and is called the skew boat conformation. Nevertheless, the chair conformation is the most stable cyclohexane form.Which one of the conformation of cyclohexane is chiral?
Twist-boat conformation of cyclohexane is chiral.
What is chair and boat conformation of glucose?
In the chair conformation, the 2nd, 3rd, 5th and 6th carbon atoms lie on one plane whereas the 1st carbon is above the plane and 4th carbon is below the plane. In the boat conformation, 1st, 2nd, 4th and 5th carbons are in one plane while the 3rd and 6th carbons are bent in the same plane outside of the plane.
What is chair boat conformation?
The chair conformation is the most stable conformation of cyclohexane. … A second, much less stable conformer is the boat conformation. This too is almost free of angle strain, but in contrast has torsional strain associated with eclipsed bonds at the four of the C atoms that form the side of the boat.
Which statement is true for cyclohexane?
Each carbon atom has one axial and one equilateral C-H bond. The chair being more stable than the boat, there is an energy difference between the chair and the boat conformations of cyclohexane which is 44 kJ/mol. Therefore, the true statement is option D.What is cyclohexane used for?
Most cyclohexane goes into the production of intermediates for nylon, which has a variety of common applications such as clothing, tents and carpets as well as thermoplastics. Cyclohexane is also used as a solvent in chemical and industrial processes and recently has been substituted for benzene in many applications.
How many total conformations are exhibited by cyclohexane?Cyclohexane can form three different conformations. Chair conformation is the most stable, with the lowest energy, and there is no steric or torsional tension.
Article first time published onWhy is twist boat form of cyclohexane more stable than boat form?
The twist relieves some of the torsional strain of the boat and moves the flagpole H further apart reducing the steric strain. Consequently the twist boat is slightly more stable than the boat. Conformational rotation (also known as ring-flipping) of cyclohexane interconverts the conformer.
Which of the following conformations of cyclohexane is chiral twist boat rigid chair boat?
Conformation of cyclohexane that are free of angle strain: Chair conformation is achiral as it possesses a center of symmetry while boat conformation is achiral as it possesses a plane of symmetry. Twist boat conformation is chiral as there is no symmetry element.
Why is the boat conformation less stable?
The boat conformation is again a puckered structure that allows tetrahedral bond angles. The boat conformation suffers from torsional strain, making it less stable (higher in energy) than the chair. … Therefore, at any instant most of the molecules in a cyclohexane sample are in chair conformations.
Which one of the following conformations of cyclohexane is the least stable?
The least stable conformation of cyclohexane is half chair conformation.
Why is chair conformation of cyclohexane more stable than boat conformation?
Answer: Chair conformation of cyclohexane is more stable than boat form because in chair conformaion the C-H bonds are equally axial and equatorial, i.e., out of twelve C-H bonds, six are axial and six are equatorial and each carbon has one axial and one equatorial C-H bond.
Which conformation of cyclohexane has a c3 axis of symmetry?
Answer is “Chair“
Is cyclohexane chiral or achiral?
Both are achiral, since the disubstituted six-membered ring has a plane of symmetry. These isomers are diastereomers of each other, and are constitutional isomers of the 1,2- and 1,3- isomers.
Why does cyclohexane adopt a chair conformation?
Explanation: The carbons in cyclohexane are all sp3 hybridized. The ideal angle between atoms connected to anything that is sp3 hybridized is 109.5 degrees. In a chair conformation, the angles of all the atoms in the ring can adopt this positioning, and thus the molecule does not experience any ring or angle strain.
Who proposed chair conformation of cyclohexane?
Investigations concerning the conformations of cyclohexane were initiated by H. Sachse (1890) and E. Mohr (1918), but it was not until 1950 that a full treatment of the manifold consequences of interconverting chair conformers and the different orientations of pendent bonds was elucidated by D. H. R.
What is chair conformation of glucose?
Glucose, in its most abundant form in solution, is a six-membered ring adopting a chair conformation with all substituents equatorial. … The lower energy chair conformation is the one with three of the five substituents (including the bulky –CH2OH group) in the equatorial position.
What is conformation of ethane?
In the eclipsed conformation, the C-H bonds on the front and back carbons are aligned with each other with dihedral angles of 0 degrees. … In the staggered conformation, the C-H bonds on the rear carbon lie between those on the front carbon with dihedral angles of 60 degrees.
How many equatorial hydrogens are in cyclohexane?
Axial and Equatorial Positions in Cyclohexane. Careful examination of the chair conformation of cyclohexane, shows that the twelve hydrogens are not structurally equivalent. Six of them are located about the periphery of the carbon ring, and are termed equatorial.
What type of hydrocarbon is cyclohexane?
Cyclohexane is an alicyclic hydrocarbon comprising a ring of six carbon atoms; the cyclic form of hexane, used as a raw material in the manufacture of nylon. It has a role as a non-polar solvent. It is a cycloalkane and a volatile organic compound.
Where does cyclohexane come from?
Modern. On an industrial scale, cyclohexane is produced by hydrogenation of benzene in the presence of a Raney nickel catalyst. Producers of cyclohexane account for approximately 11.4% of global demand for benzene. The reaction is highly exothermic, with ΔH(500 K) = -216.37 kJ/mol).
Which is a structural isomer of cyclohexane?
Cyclohexane isomers are compounds that contain atoms or groups of atoms bonded to the cyclohexane ring itself. 1,2-Dichlorocyclohexane and 1,4-dichlorocyclohexane represent what are called constitutional isomers, which are compounds that have the same chemical formula, but the atoms are connected differently.
Which statement is incorrect about cyclohexane?
Which statement about cyclohexane is incorrect? Each C atom is sp3 hybridized. Cyclohexane suffers ring strain. The cyclohexane ring can flip between chair and boat conformers.
Which statement is true about a cyclohexane and 1 hexene?
Cyclohexane and 1-hexene are constitutional isomers. Thus, the statement is true.
Which conformation of methylcyclohexane is the most stable?
The most stable conformation of methylcyclohexane is the chair conformation in which the methyl group is equatorial. The alternative chair conformation, in which the methyl group is axial, is 7.3 kJ/mol higher in energy. This difference corresponds to a equatorial:axial conformer ratio of 19:1 at 25 °C.
What is flagpole interaction in boat conformation of cyclohexane?
In the boat conformation the two bonds shown in red (2) are called flagpole bonds. The hydrogen atoms on the flagpole bonds are called flagpole hydrogens. … Consequently, the boat conformation of cyclohexane ring is less stable than the chair conformation.
Which one of the following is the most energetic conformation of cyclohexane?
Most stable and least energetic conformation of cyclohexane is half-chair.
Which conformation of cyclohexane has highest stability?
The chair form is the most stable conformation of cyclohexane.