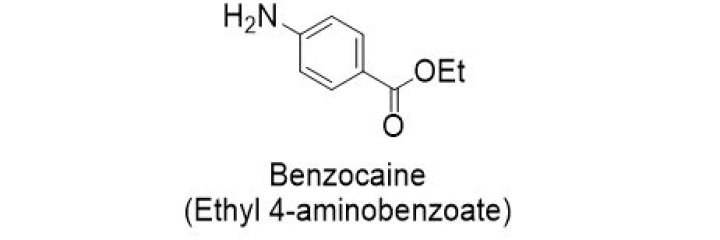
What is benzocaine made of

Victoria Simmons
Published Apr 05, 2026
Benzocaine is a chemical compound derived from para-aminobenzoic acid and ethanol. It is a white, odorless, crystalline powder. It acts on neuronal membranes and blocks nerve signals in the body. Its numbing effects are temporary.
How does Le Chatelier's principle influence the benzocaine synthesis?
Le Chatelier’s Principle can be exploited to push the reaction toward the product side. … In this synthesis of benzocaine, by using microwave heating, we will be able to successfully complete the actual reaction of benzocaine in just a couple of minutes.
Why is absolute ethanol used in the synthesis of benzocaine?
Benzocaine is soluble in Ethanol, Methanol and Diethyl Ether. But it is not very soluble in water. Having water in the solution would decrease the solubility and would create a lot of hassle later. … Using 95 % ethanol would reduce the yield of benzocaine.
How is Novocaine synthesized?
Procaine, the 2-diethylaminoethyl ester of 4-aminobenzoic acid (2.1. 1), better known as novocaine, is synthesized in two ways. The first way consists of the direct reaction of the 4-aminobenzoic acid ethyl ester with 2-diethylaminoethanol in the presence of sodium ethoxide.How is benzocaine metabolized?
Benzocaine is metabolized into at least three compounds by acetylation and hydrolysis. Benzocaine and its acetylated metabolite are rapidly eliminated across the gills while the more polar de-ethylated and de-ethylated-acetylated metabolites are excreted at slower rates primarily in the urine.
How is benzocaine synthesized industrially from P nitrobenzoic acid?
Benzocaine can be prepared by esterification using 4-aminobenzoic acid and ethanol. It can also be prepared by reduction of ethyl 4-nitrobenzoate to the amine. In industrial practice, the reducing agent is usually iron and water in the presence of a little acid.
Is benzocaine soluble in water?
Benzocaine occurs in two forms: a crystalline salt (benzocaine HCl) that is soluble in water at 0.4 g/L, and a nonwater-soluble basic form that must be dissolved in ethyl alcohol at a concentration of 0.2 g/ml before it is sufficiently soluble in water to create a functional induction dose.
How is Le Chatelier's principle used to increase the success of the Fischer esterification reaction?
The Fischer esterification reaction takes advantage of Le Chatelier’s principle to increase the amount of carboxylic acid that is esterified. The equilibrium is shifted towards products by using a large excess of the alcohol (it is used as the reaction solvent), and (in some cases) also removing water as it it formed.How do you synthesize benzocaine from P nitrotoluene?
Principle: In step 1 common reduction of aromatic para-nitrobenzoic acid by tin and hydrochloric acid to para-aminobenzoic acid and in step 2 esterification of para-aminobenzoic acid by sulphuric acid and ethanol to benzocaine occurring called Fischer esterification.
What is lidocaine made from?lidocaine, synthetic organic compound used in medicine, usually in the form of its hydrochloride salt, as a local anesthetic. Lidocaine produces prompter, more intense, and longer lasting anesthesia than does procaine (Novocaine).
Article first time published onWhy is Novacaine no longer used?
Novocaine is no longer used due to the increased time it takes to work, how long it is effective and its chances of causing allergic reactions. Lidocaine and Septocaine are our preferred anesthetics due to their faster onset time, longer acting time and very minimal chance of allergic reactions.
Is it legal to buy Novocaine?
In fact, novocaine is usually sold only to licensed dental and medical professionals. Some websites might advertise novocaine for sale to the general public. But know that, as of this writing, selling novocaine to unlicensed people is illegal.
Why is ethanol a good solvent for Fischer esterification?
2. The ethanol is probably an excess reactant, so it also serves as a solvent. The most important reason for using absolute ethanol is that the Fischer esterification is an equilibrium reaction with a Keq≈4 . … The absolute alcohol contains no water, so the reaction will be able to go further to the right.
How does Fischer esterification work?
Fischer Esterification is an organic reaction which is employed to convert carboxylic acids in the presence of excess alcohol and a strong acid catalyst to give an ester as the final product. This ester is formed along with water.
Why do we use absolute ethanol?
Use as a solvent, a germicide, a beverage, an antifreeze, a fuel, a depressant, and a chemical intermediate for other organic chemicals. … The main uses for industrial ethanol are as an intermediate for the production of other chemicals and as a solvent.
How does benzocaine work chemistry?
Benzocaine is an ester of paraaminobenzoic acid, lacking the terminal diethylamino group of procaine, with anesthetic activity. Benzocaine binds to the sodium channel and reversibly stabilizes the neuronal membrane which decreases its permeability to sodium ions.
How is tetracaine metabolized?
Tetracaine is an amide local anesthetic agent with a longer duration of action than lidocaine and cocaine. It is available as 0.5%, 1%, and 2% solutions for local use. It is metabolized through hydrolysis by plasma cholinesterase.
How do you dissolve benzocaine in water?
A. Dissolve 0.10 g in 5 mL of water, add 3 drops of hydrochloric acid (~70 g/l) TS and 5 drops of iodine TS; a brown precipitate is produced. B.
Is benzocaine the same as lidocaine?
Lidocaine also comes in a systemic injection that’s used to treat cardiac rhythm problems. Benzocaine is more commonly used in throat lozenges and as a spray. Lidocaine and benzocaine are equally effective for pain relief. Both drugs have similar side effects, most commonly stinging or local skin issues.
How do you dissolve Benzocaine powder?
“Benzocaine is sparingly soluble in water; it is more soluble in dilute acids and very soluble in ethanol…” So it won’t do much in water, would dissolve better in vinegar, and pretty well in everclear or vodka.
How do you synthesize P-aminobenzoic acid?
As shown in Scheme 1, in order to prepare p-aminobenzoic acid, toluene is first nitrated to synthesize ortho-nitrotoluene and para-nitrotoluene, which are separated, and then p- Nitrotoluene is oxidized to prepare p-nitrobenzoic acid, and the nitro group of p-nitrobenzoic acid is synthesized by reduction using an …
What does PABA mean?
Para-Aminobenzoic Acid (Paba)
What is the role of h2so4 in ethyl p aminobenzoate benzocaine from P-aminobenzoic acid?
The reaction mixture should dissolve completely in the water, since the ethyl p-aminobenzoate is in the form of the hydrogen salt. … At this point the sulfuric acid is completely neutralized, and the ethyl p-aminobenzoate precipitates.
Why benzocaine precipitates during the neutralization?
Explain why benzocaine precipitates during the neutralization. With the addition of an acid, the amine group on the benzocaine will become protonated. The protonation creates a positive charge on the benzocaine molecule. … Thus, the benzocaine will no longer be soluble and will precipitate during neutralization.
How do you purify Benzocaine?
Purifying the Benzocaine. a) Add a few mL of hot ethanol and heat the mixture on the steam bath until all the oil dissolves. b) Add water (drop wise!) to the alcohol solution until cloudiness just appears and then add a few drops of ethanol. Cool the mixture, with occasional VIGOROUS swirling, in an ice bath.
Is Benzocaine a blood thinner?
Benzocaine, a local anesthetic, can cause a condition in which the amount of oxygen carried through the blood is greatly reduced. This condition, called methemoglobinemia, can be life-threatening and result in death.
How does Le Chatelier's principle relate to esterification?
Le Chatelier’s principle states that an equilibrium reaction can be driven in one direction by changing the concentration, pressure, temperature, or volume of the reagents or products. With a 1:1 mixture of carboxylic acid and alcohol, the esterification reaction reaches equilibrium with about 70% yield of the ester.
How can the Fischer esterification be driven to completion?
This reaction is also known as the Fischer esterification. Esters are obtained by refluxing the parent carboxylic acid with the appropraite alcohol with an acid catalyst. The equilibrium can be driven to completion by using an excess of either the alcohol or the carboxylic acid, or by removing the water as it forms.
What small product is also produced during an esterification reaction?
Esters are formed from an esterification reaction, with simple esters being formed through Fisher esterification. This reaction converts a carboxylic acid and alcohol into an ester with water as a by-product. Fisher esterification is a reversible reaction that proceeds very slowly.
Is lidocaine made from coca plant?
The history of lidocaine began with its synthesis from cocaine. Cocaine is a naturally occurring compound indigenous to the Andes Mountains, West Indies, and Java. It was the first anesthetic to be discovered and is the only naturally occurring local anesthetic; all others are synthetically derived.
Who invented Xylocaine?
Lidocaine, the first amino amide–type local anesthetic, was first synthesized under the name ‘xylocaine’ by Swedish chemist Nils Löfgren in 1943. His colleague Bengt Lundqvist performed the first injection anesthesia experiments on himself. It was first marketed in 1949.