What is americium used for

Robert Spencer
Published Mar 17, 2026
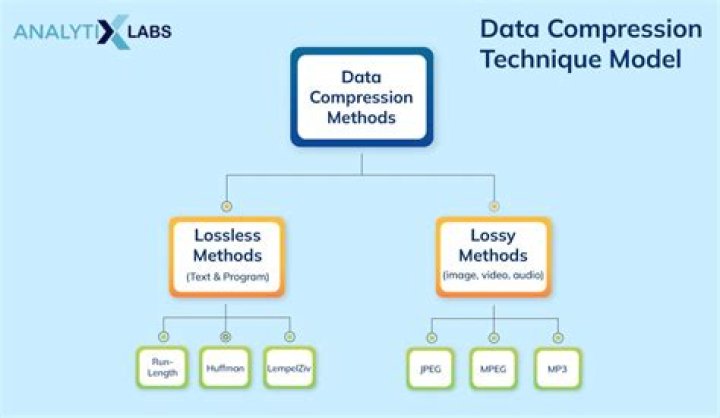
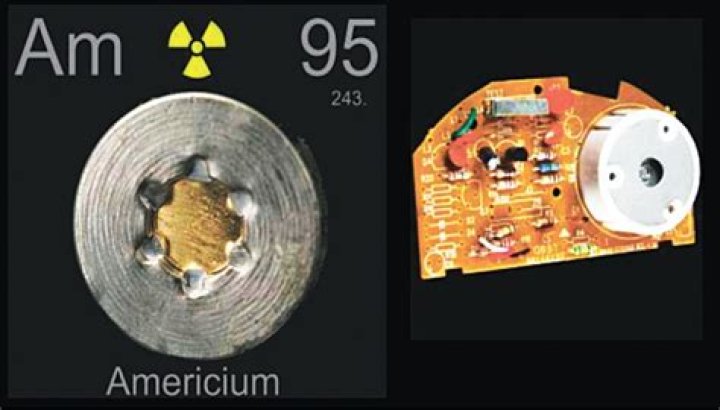
Americium is a silvery, shiny radioactive metal. Americium is commonly used in smoke alarms, but has few other uses. It has the potential to be used in spacecraft batteries in the future. Currently plutonium is used but availability is poor so alternatives are being considered.
What items contain americium?
The two major types of household smoke detectors are ionization smoke detectors and optical smoke detectors. Most common household ionization smoke detectors use Americium-241 in the form of Americium Dioxide as its source of the ionizing radiation that the smoke detector needs to work.
Does the human body use americium?
Americium emits radioactive alpha particles in the bone that can cause damage to the surrounding tissue. The body normally repairs all of the damage, but if it fails, bone cancer could result after many years.
How much does americium cost?
One gram of americium oxide provides enough active material for more than three million household smoke detectors. Its cost is about $1500 per gram.Is americium used in bombs?
Oct. Cesium-137, Cobalt-60, and Americium-241 are considered to be the most likely materials for use in a dirty bomb due to their availability and their relative ease of handling. …
Is americium bad for the environment?
Environmental effects of americium Its isotopes decay very slowly in the environment and as a result they can do harm to plants and animals. When animals are exposed to extreme levels of americium, results may be damage to organs such as the lungs, liver and thyroid.
How is americium used in everyday life?
Americium is a silvery, shiny radioactive metal. Americium is commonly used in smoke alarms, but has few other uses. It has the potential to be used in spacecraft batteries in the future. Currently plutonium is used but availability is poor so alternatives are being considered.
What element has 159 neutrons?
For example, seaborgium 265, which has a nucleus containing 106 protons and 159 neutrons, decays to rutherfordium 261, which decays to nobelium 257.Is it legal to own americium?
Americium is the only man-made element which is legal to posses, and the amazing part is that you can buy it for less than $10 at any hardware store.
Which is the rarest element on the Earth?A team of researchers using the ISOLDE nuclear-physics facility at CERN has measured for the first time the so-called electron affinity of the chemical element astatine, the rarest naturally occurring element on Earth.
Article first time published onIs americium a heavy metal?
AmericiumGroupgroup n/aPeriodperiod 7Blockf-blockElectron configuration[Rn] 5f7 7s2
Why is americium used in smoke detectors?
Ionization smoke detectors use americium as a source of alpha particles. Alpha particles from the americium source ionize air molecules. This makes some particles positively charged and some negatively charged. … Because of this shielding, the smoke detector poses no radiation health risk when they are properly handled.
What is AM 241 used for?
Americium-241 is used as a neutron source in non-destructive testing of machinery and equipment, and as a thickness gauge in the glass industry. However, its most common application is as an ionization source in smoke detectors, and most of the several kilograms of americium made each year are used in this way.
What did the nuclear bomb do to Japan?
The uranium bomb detonated over Hiroshima on 6 August 1945 had an explosive yield equal to 15,000 tonnes of TNT. It razed and burnt around 70 per cent of all buildings and caused an estimated 140,000 deaths by the end of 1945, along with increased rates of cancer and chronic disease among the survivors.
Is there uranium in smoke detectors?
The isotope of americium used in smoke detectors is americium-241, which decays by α emission to neptunium-237 with a half-life of 432.2 years. Plutonium-239 is itself the primary fissile isotope of plutonium (used in nuclear weapons), and is in turn made from uranium. …
What is bismuth used for today?
Bismuth finds its main uses in pharmaceuticals, atomic fire alarms and sprinkler systems, solders and other alloys and pigments for cosmetics, glass and ceramics. It is also used as a catalyst in rubber production.
Is strontium a metal?
A soft, silvery metal that burns in air and reacts with water. Strontium is best known for the brilliant reds its salts give to fireworks and flares. It is also used in producing ferrite magnets and refining zinc.
Is thorium man-made?
Thorium (chemical symbol Th) is a naturally occurring radioactive metal found at trace levels in soil, rocks, water, plants and animals. … There are natural and man-made forms of thorium, all of which are radioactive. In general, naturally occurring thorium exists as Th-232, Th-230 or Th-228.
How was americium produced?
Americium (chemical symbol Am) is a man-made radioactive metal that is solid under normal conditions. Americium is produced when plutonium absorbs neutrons in nuclear reactors or during nuclear weapons tests. Americium-241 is the most common form of Americium.
Where is CF on the periodic table?
californium (Cf), synthetic chemical element of the actinoid series of the periodic table, atomic number 98.
What is the daughter isotope?
In nuclear physics, a decay product (also known as a daughter product, daughter isotope, radio-daughter, or daughter nuclide) is the remaining nuclide left over from radioactive decay.
Why do smoke detectors expire?
If you were to remove a smoke detector from where it sits on the wall or ceiling, you’ll find there are two key dates on the backside. Besides the serial number, you’ll find a manufacture date and a “Replace By” date. Why do these devices need to be replaced every 10 years? It’s because the sensors degrade over time.
Which metal is used for containers that store radioactive sources?
To shield containers that store radioactive materials, metallic lead is typically used.
What is an atomic notation?
Standard nuclear notation shows the chemical symbol, the mass number and the atomic number of the isotope. Example: the isotopes of carbon. The element is determined by the atomic number 6.
How many electrons does Dy have?
Atomic Number66Number of Neutrons97Shell structure (Electrons per energy level)[2, 8, 18, 28, 8, 2]Electron Configuration[Xe] 4f10 6s2Valence Electrons4f10 6s2
What is the chemical symbol for bismuth?
bismuth (Bi), the most metallic and the least abundant of the elements in the nitrogen group (Group 15 [Va] of the periodic table). Bismuth is hard, brittle, lustrous, and coarsely crystalline.
Is Diamond a metal?
Diamond is not considered as a non-metal in the exceptional category as diamond is a form of carbon. It is not classified as an element. … It is an allotrope of carbon.
What is the most abundant thing on earth?
Q: What is the most abundant element on Earth? A: Oxygen, which composes about 49.5% of the total mass of the Earth’s crust, waters and atmosphere, according to the textbook “Modern Chemistry.” Silicon is second at 28%.
Is there synthetic gold?
Chrysopoeia, the artificial production of gold, is the symbolic goal of alchemy. Such transmutation is possible in particle accelerators or nuclear reactors, although the production cost is currently many times the market price of gold.
What is an interesting fact about americium?
Americium (chemical symbol: Am) Americium is a radioactive, soft, silver-white, heavy metal that is denser than lead and slowly tarnishes in air. With a density of about 13.6 g/cc, it is almost twice as heavy as steel. Americium is a transuranic metal, which means it is located after uranium on the periodic table.
How many isotopes does americium have?
Nineteen radioisotopes of americium—223Am, 229Am, 230Am, and those ranging from 232Am to 247Am—have been characterized, with the most stable being 243Am with a half-life of 7,370 years, and 241Am with a half-life of 432.2 years.