What is a quaternary structure in biology

Robert Spencer
Published Apr 13, 2026
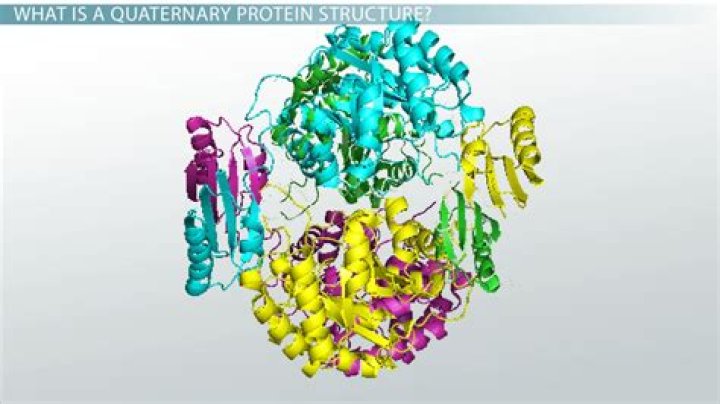
The quaternary structure of a protein is the association of several protein chains or subunits into a closely packed arrangement. Each of the subunits has its own primary, secondary, and tertiary structure. … The subunits in a quaternary structure must be specifically arranged for the entire protein to function properly.
What is quaternary structure and example?
Description and examples The quaternary structure refers to the number and arrangement of the protein subunits with respect to one another. Examples of proteins with quaternary structure include hemoglobin, DNA polymerase, ribosomes, antibodies, and ion channels.
What is the function of quaternary structure?
Quaternary structure is an important protein attribute that is closely related to its function. Proteins with quaternary structure are called oligomeric proteins. Oligomeric proteins are involved in various biological processes, such as metabolism, signal transduction, and chromosome replication.
Which best defines quaternary structure?
Quaternary structure describes how polypeptide chains fit together to form a complete protein. Quaternary protein structure is held together by hydrophobic interactions, and disulfide bridges. … “The four parts of a protein’s amino acid sequence” does not refer to anything in particular.Why is it called quaternary structure?
Many proteins are made up of a single polypeptide chain and have only three levels of structure (the ones we’ve just discussed). However, some proteins are made up of multiple polypeptide chains, also known as subunits. When these subunits come together, they give the protein its quaternary structure.
Is hemoglobin A heterodimer?
A tetramer can be assembled as dimer of dimers with two homodimer subunits (such as sorbitol dehydrogenase), or two heterodimer subunits (such as hemoglobin).
What happens quaternary structure?
Quaternary structure is the interaction of two or more folded polypeptides. Many proteins require the assembly of several polypeptide subunits before they become active. If the final protein is made of two subunits, the protein is said to be a dimer.
Why does hemoglobin have a quaternary structure?
The structure for hemoglobin is very similar to myoglobin except that it has a quaternary structure due to the presence of four protein chain subunits. … Each hemoglobin molecule can bind to a total of four oxygen molecules.Why are quaternary structures so important to cell function?
Functions of Quaternary Structure As mentioned above, quaternary structure allows a protein to have multiple functions. It also allows for a protein to undergo complicated conformational changes. This has several mechanisms. First, an individual subunit can change shape.
Why do proteins adopt quaternary structure?Finally, the quaternary structure of a protein refers to those macromolecules with multiple polypeptide chains or subunits. The final shape adopted by a newly synthesized protein is typically the most energetically favorable one. … Rather, the atoms within these proteins remain capable of making small movements.
Article first time published onWhat is the difference between tertiary and quaternary structure?
Tertiary structure refers to the configuration of a protein subunit in three-dimensional space, while quaternary structure refers to the relationships of the four subunits of hemoglobin to each other.
What types of forces give rise to quaternary structures?
The quaternary structure of macromolecules is stabilized by the same non-covalent interactions and disulfide bonds as the tertiary structure, and can also be affected by formulation conditions.
How do you determine quaternary structure?
The quaternary structure (QS) of a protein is determined by measuring its molecular weight in solution. The data have to be extracted from the literature, and they may be missing even for proteins that have a crystal structure reported in the Protein Data Bank (PDB).
Are all enzymes quaternary?
Explanation: It would completely depend on the enzyme we’re considering. Some are only monomeric (e.g. trypsin), some contain several subunits which interact to form a quaternary structure. … In the case of trypsin, a serine protease, we could say the enzyme only has a tertiary structure.
Does insulin have a quaternary structure?
Quaternary Structure For example, insulin (a globular protein) has a combination of hydrogen bonds and disulfide bonds that cause it to be mostly clumped into a ball shape.
Is myoglobin a Heterotetramer?
Hemoglobin and myoglobin are heterotetramers. … Both hemoglobin and myoglobin contain a prosthetic group called heme, which contains a central iron atom. 7. Each iron atom can form six coordination bonds: One of these bonds is formed between iron and oxygen.
Does myoglobin bind oxygen reversibly?
Myoglobin is a hemoprotein found in the skeletal muscle of mammals that functions in oxygen storage and diffusion. … The heme in myoglobin can reversibly bind a O2 molecule to regulate the transportation of O2 from red blood cells to mitochondria when skeletal muscles are metabolically active.
Is a dimer a quaternary structure?
The protein quaternary structure of the complex is divided into five categories, namely, monomer, dimer, trimer, tetramer, and other subunit classes.
Do all proteins have quaternary structure Why or why not?
Proteins made from a single polypeptide will not have a quaternary structure. In proteins with more than one subunit, weak interactions between the subunits help to stabilize the overall structure. Enzymes often play key roles in bonding subunits to form the final, functioning protein.
What is meant by quaternary structure of a protein?
The quaternary structure of a protein is the association of several protein chains or subunits into a closely packed arrangement. Each of the subunits has its own primary, secondary, and tertiary structure. The subunits are held together by hydrogen bonds and van der Waals forces between nonpolar side chains.
What type of bonds occur in tertiary and quaternary structures to cause a globular look?
Hydrogen bonds between back bone atoms are important in maintaining secondary structures, and those between side chains are involved in maintaining the tertiary structure. Examples of finding and visualizing both types in globular proteins are at hydrogen bonds.
Are all globular proteins quaternary?
As in all proteins, the primary structure of globular proteins consists of a polypeptide, or chain of amino acids joined via peptide bonds. … Some globular proteins, such as hemoglobin, also exhibit quaternary structure, in which multiple polypeptide chains are clustered together to form a functional protein.
What is the difference between the primary secondary tertiary and quaternary structure of a protein?
A protein’s primary structure is defined as the amino acid sequence of its polypeptide chain; secondary structure is the local spatial arrangement of a polypeptide’s backbone (main chain) atoms; tertiary structure refers to the three-dimensional structure of an entire polypeptide chain; and quaternary structure is the …
Which is a property of tertiary structure and quaternary structure?
23. _____ Which is a property of tertiary structure and quaternary structure? a) Both structures are stabilized by numerous covalent hydrophobic and hydrophilic interactions.
Are enzymes tertiary or quaternary?
Enzymes are mainly globular proteins – protein molecules where the tertiary structure has given the molecule a generally rounded, ball shape (although perhaps a very squashed ball in some cases).
What bonds form tertiary structure?
The tertiary structure of a protein consists of the way a polypeptide is formed of a complex molecular shape. This is caused by R-group interactions such as ionic and hydrogen bonds, disulphide bridges, and hydrophobic & hydrophilic interactions.
Which of the following is associated with the quaternary structure of proteins?
The interaction of protein subunits is associated with the quaternary structure of proteins.
What are the forces that stabilize the quaternary structure of a protein?
The stabilizing forces that hold the polypeptide subunits together are the same forces that are responsible for tertiary structure stabilization. A major force stabilizing the quaternary structure is the hydrophobic interaction among nonpolar side chains at the contact regions of the subunits.
Which is true about the quaternary structure of proteins?
-Quaternary structure is defined as the 3D structure of proteins with four subunits. -Quaternary structure requires covalent interactions between polypeptide chains. -Quaternary structure is stabilized by the same types of noncovalent interactions as tertiary structure. … -Water is released when a peptide bond is formed.
What is the quaternary structure of a protein quizlet?
The Quaternary structure of the protein is two or more polypeptide chains bonded together, and maintained by the same interactions as the tertiary structure.
What is an example of a tertiary protein structure?
Protein tertiary structure. For example, amide hydrogen atoms can form H‐bonds with nearby carbonyl oxygens; an alpha helix or beta sheet can zip up, prompted by these small local structures. Hydrophobic interactions among the amino acid side chains also determine tertiary structure.