What is a particle diagram

Sophia Edwards
Published Mar 17, 2026
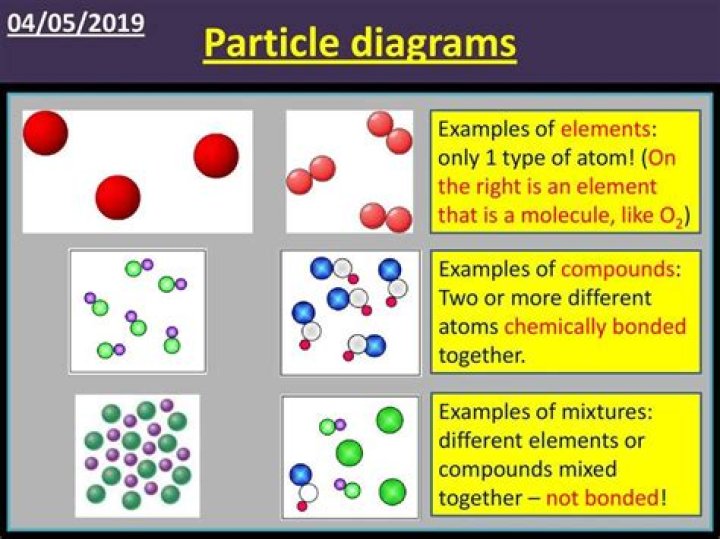
Particle Diagrams. • Elements and compounds can be represented using particle diagrams, which is a box in which colored balls are draw to represent atoms or molecules. • These diagrams can represent elements and compounds, as well as their molecular composition by the types of balls and how they are connected.
What is particle level diagram?
The particle diagrams illustrate volume (size of the box drawn), amount of gas (number of particles drawn), temperature (motion lines on particles), and pressure (collisions or bounces with the container). The activities described here are simple ways to have students practice the skill of drawing particle diagrams.
What do you mean by particulate?
a separate and distinct particle. a material composed of such particles. particulates, … solid or liquid particles suspended in the atmosphere, especially pollutants.
What is the particle model?
The particle theory of matter is a model that describes the arrangement and movement of particles in a substance. The model is used to explain the physical properties of solids, liquids and gases.What can be called a particle?
A particle is a small piece of anything. … These particles are even smaller than atoms. You can also say a crumb or speck of something is a particle. Dust particles are piling up all the time, and vacuuming cleans up lots of particles. Those particles are little bits of things.
What is the molecules of gas?
Gas molecules are made up of a number of atoms bonded to one another. These interatomic bonds are similar to springs connecting atoms of various masses together. This bonding vibrates with a fixed frequency called the natural frequency.
What is the difference between a compound and a mixture?
Difference between Compound and Mixture Compounds are formed because of chemical bonding between two elements. Mixtures are formed when substances are physically mixed with one another. Compounds are basically of three kinds : Ionic, metallic and covalent.
What are the 4 main ideas of the particle model?
- All substances are made of particles.
- The particles are attracted to each other (some strongly, others weakly).
- The particles move around (have kinetic energy).
- As temperature increases, the particles move more (their kinetic energy increases).
Why is a particle diagram important?
Scientists find the particle model useful for two reasons. First, it provides a reasonable explanation for the behaviour of matter. … Thus, the particle model can be used to explain the properties of solids, liquids, and gases. It can also be used to explain what happens in changes of state (Figure 1 on the next page).
How do you identify particles?Charged particles have been identified using a variety of techniques. All methods rely on a measurement of the momentum in a tracking chamber combined with a measurement of the velocity to determine the charged particle mass, and therefore its identity.
Article first time published onWhat are the particles in an element?
A typical atom consists of three subatomic particles: protons, neutrons, and electrons (as seen in the helium atom below). Other particles exist as well, such as alpha and beta particles (which are discussed below). The Bohr model shows the three basic subatomic particles in a simple manner.
What's the difference between a molecule and a particle?
Particle is a general term while molecule is a specific one. Particle refers to an atom, molecule,ion,radical, electron, proton, neutron or a compound… Molecule is a type of particle. It is a combination of two or more same or different atoms.
What are particulates in geography?
Particulate matter – these are tiny particles of solids or liquids suspended in the air. They come from carbon emissions from engines, small bits of metal and rubber from engine wear and braking as well as dust from road surfaces. They can come from natural sources and from building and industry.
What is the difference between particle and particulate?
A particle is matter in form of one “solid” piece. A particulate is matter in the form of several or many separate particles.
How does particulate matter form?
“Particulate matter,” also known as particle pollution or PM, is a complex mixture of extremely small particles and liquid droplets. … These particles can be directly emitted from sources such as forest fires, or they can form when gases emitted from power plants, industries and automobiles react in the air.
What particle diagram represents one substance in the gas phase?
QuestionsAnswerExplanations41 Which particle diagram represents one substance in the gas phase?3gases are spread out they must be the same compounds or elements to be one substance
How do the particles in a solid liquid and gas differ?
Particles in a: gas are well separated with no regular arrangement. liquid are close together with no regular arrangement. solid are tightly packed, usually in a regular pattern.
How do you find the particle number?
N = n × NAwhereN = number of particlesn = moles of particlesNA = Avogadro number = 6.022 × 1023
What is particle molarity?
Each mole contains 6.022 × 10^23 particles, and the mass of a mole of particles is the sum of the atomic weights of its elements. … This is the solution’s particle concentration, measured in molarity (M), or moles per liter.
What is the concentration of particles?
Particle number concentration is the total number of particles per unit volume of air (for example cm-3), whereas particle mass concentration is the total mass of particles per unit volume (for example µg m-3). Mass concentrations are typically dominated by larger particles.
What is material particle?
[mə′tir·ē·əl ′pärd·ə·kəl] (mechanics) An object which has rest-mass and an observable position in space, but has no geometrical extension, being confined to a single point. Also known as particle.
Are electrons particles?
Note that an electron is a fundamental particle; it is not made out of anything else (according to our current experiments and theories). All fundamental particles interact as shapeless points when acting like particles.
Can particles be destroyed?
Yes, particles can be created or destroyed, but it takes energy. If a gamma ray with enough energy passes by a heavy nucleus it can become an electron and and a positron.
What is the difference between H * * * * * * * * * * and heterogeneous mixture?
A homogenous mixture is that mixture in which the components mix with each other and its composition is uniform throughout the solution. A heterogenous mixture is that mixture in which the composition is not uniform throughout and different components are observed.
Is air a pure substance?
A substance that has a fixed chemical composition throughout is called a pure substance such as water, air, and nitrogen. A pure substance does not have to be of a single element or compound.
What is the difference between a physical change and a chemical change?
In a physical change the appearance or form of the matter changes but the kind of matter in the substance does not. However in a chemical change, the kind of matter changes and at least one new substance with new properties is formed. The distinction between physical and chemical change is not clear cut.
What do particles do with density?
Density basically describes how tightly packed the tiny particles that make up any substance/material are. The higher the density, the more closely packed the particles (called atoms and molecules) are. The lower the density, the more spaced out the particles are.
What is meant by density?
density, mass of a unit volume of a material substance. … Density offers a convenient means of obtaining the mass of a body from its volume or vice versa; the mass is equal to the volume multiplied by the density (M = Vd), while the volume is equal to the mass divided by the density (V = M/d).
Why do we compress gases?
The atoms, ions, or molecules that make up the solid or liquid are very close together. There is no space between the individual particles, so they cannot pack together. … Gases are compressible because most of the volume of a gas is composed of the large amounts of empty space between the gas particles.
Students will know and understand that: Gases have mass. The space between gas particles is empty.
One way to calculate mass: Mass = volume × density. Weight is the measure of the gravitational force acting on a mass. The SI unit of mass is “kilogram”.