What group is lanthanum in

Emily Dawson
Published Mar 15, 2026
GroupLanthanides920°C, 1688°F, 1193 KPeriod63464°C, 6267°F, 3737 KBlockd6.15Atomic number57138.905State at 20°CSolid139La
What family does lanthanides belong to?
The lanthanide and actinide series make up the inner transition metals. The lanthanide series includes elements 58 to 71, which fill their 4f sublevel progressively.
What family is Group 4?
Group 4 is the second group of transition metals in the periodic table. It contains the four elements titanium (Ti), zirconium (Zr), hafnium (Hf), and rutherfordium (Rf). The group is also called the titanium group or titanium family after its lightest member.
Where is lanthanum found on the periodic table?
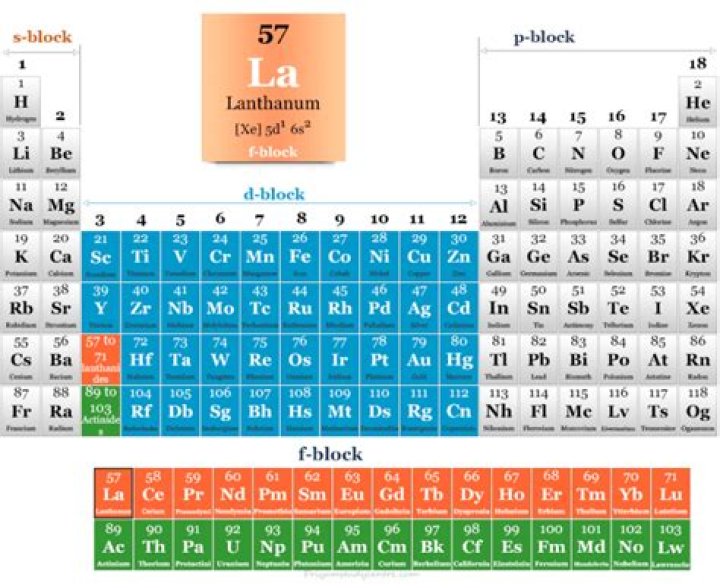
Lanthanum is the first element and prototype of the lanthanide series. In the periodic table, it appears to the right of the alkaline earth metal barium and to the left of the lanthanide cerium.Where is in on the periodic table?
Element NameSymbolAtomic NumberHydrogenH1IndiumIn49IodineI53IridiumIr77
Who is modern periodic table?
In the 1860s, a scientist named Dmitri Mendeleev also saw the need to organize the elements. He created a table in which he arranged all of the elements by increasing atomic mass from left to right across each row.
What is mn on the periodic table?
manganese (Mn), chemical element, one of the silvery white, hard, brittle metals of Group 7 (VIIb) of the periodic table.
Where do lanthanide and actinide really belong?
All actinides belong in the 3B group. The lanthanides and actinides are arranged below with a notation in the main body cell rather than making room for all these elements in the 3B group in the main body of the periodic table.What family is bromine in?
bromine (Br), chemical element, a deep red noxious liquid, and a member of the halogen elements, or Group 17 (Group VIIa) of the periodic table.
What is the luster of lanthanum?Lanthanum is a relatively soft metal that when freshly cut looks has a medium gray luster similar to iron.
Article first time published onHow is lanthanum processed?
Today, lanthanum is primarily obtained through an ion exchange process from monazite sand ((Ce, La, Th, Nd, Y)PO4), a material rich in rare earth elements that can contain as much as 25% lanthanum. … Lanthana (La2O3) is used to make the glass used in camera lenses and in other special glasses.
What is ND in chemistry?
neodymium (Nd), chemical element, a rare-earth metal of the lanthanide series of the periodic table.
What period and group is hydrogen in?
Group1−259.16°C, −434.49°F, 13.99 KPeriod1−252.879°C, −423.182°F, 20.271 KBlocks0.000082Atomic number11.008State at 20°CGas1H, 2H
What family is chlorine in?
Chlorine is in group 17 of periodic table, also called the halogens, and is not found as the element in nature – only as a compound. The most common of these being salt, or sodium chloride, and the potassium compounds sylvite (or potassium chloride) and carnallite (potassium magnesium chloride hexahydrate).
What are the family in periodic table?
Families of the Periodic Table. On the periodic table, there are families which are groups of elements with similar properties. These families are alkali metals, alkaline earth metals, transition metals, post-transition metals, metalloids, halogens, noble metals, and noble gases.
Is mercury a metal?
Elemental or metallic mercury is a shiny, silver-white metal, historically referred to as quicksilver, and is liquid at room temperature. It is used in older thermometers, fluorescent light bulbs and some electrical switches.
What period is K in?
Group163.5°C, 146.3°F, 336.7 KPeriod4759°C, 1398°F, 1032 KBlocks0.89Atomic number1939.098State at 20°CSolid39K
What type of solid is mn?
Atomic Mass54.93804uElectron Configuration[Ar]4s23d5Oxidation States+7, +4, +3, +2Year Discovered1774
How can I memorize the periodic table?
- Break down the table into sections. …
- Spread out the memorization process. …
- Learn the elements in a song. …
- Make nonsense words made from element symbols. …
- Use color to learn element groups.
How many periods are there in modern periodic table?
There are currently seven complete periods in the periodic table, comprising the 118 known elements. Any new elements will be placed into an eighth period; see extended periodic table.
How many periods are there in the periodic table?
The periodic table of the elements contains all of the chemical elements that have been discovered or made; they are arranged, in the order of their atomic numbers, in seven horizontal periods, with the lanthanoids (lanthanum, 57, to lutetium, 71) and the actinoids (actinium, 89, to lawrencium, 103) indicated …
What family is barium in?
Group 2A — The Alkaline Earth Metals. Group 2A (or IIA) of the periodic table are the alkaline earth metals: beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).
Which group is CA in?
calcium (Ca), chemical element, one of the alkaline-earth metals of Group 2 (IIa) of the periodic table.
What period is bromine in?
Group17Melting pointPeriod4Boiling pointBlockpDensity (g cm−3)Atomic number35Relative atomic massState at 20°CLiquidKey isotopes
Where are the metalloids located?
Metalloids lie on either side of the dividing line between metals and nonmetals. This can be found, in varying configurations, on some periodic tables. Elements to the lower left of the line generally display increasing metallic behaviour; elements to the upper right display increasing nonmetallic behaviour.
Where are metals located on the periodic table and what are their properties?
The metals are on the bottom left in the periodic table, and the nonmetals are at the top right. The semimetals lie along a diagonal line separating the metals and nonmetals.
Are lanthanides and actinides metals or nonmetals?
Summary. Lanthanides and actinides are elements with unfilled f orbitals. Lanthanides are all metals with reactivity similar to group 2 elements. Actinides are all radioactive elements.
What is lanthanum used in?
Lanthanum and other rare-earth compounds are used in carbon arc lighting, specifically in the film and television industry for studio lighting and projection. Lanthanum oxide (La2O3), also called lanthana, improves the alkali resistance of glass and is used in making camera lenses and in other special glasses.
What does lanthanum react with?
Lanthanum reacts with water to form lanthanum hydroxide plus hydrogen gas. Lanthanum is chemically reactive and forms compounds with carbon, nitrogen, boron, selenium, silicon, phosphorus, sulfur, and with the halogens.
What does a stand for in the periodic table?
The chemical elements of the periodic chart sorted by:SymbolName chemical element- ElectronegativityArArgon- DensityAsArsenic- Melting pointAtAstatine- Boiling pointAuGold
What is the atomic mass of polonium?
Thus polonium-210 (mass number 210 and atomic number 84, i.e., a nucleus with 84 protons) decays by alpha emission to lead-206 (atomic number 82).