What food contains trypsin

Emma Valentine
Published Apr 25, 2026
Trypsin inhibitor is present in various foods such as soybeans, grains, cereals and various additional legumes. The main function of trypsin inhibitors in these foods is to act as a defense mechanism. By having this harmful component wild animals learn that any food that contains trypsin inhibitor is a food to avoid.
What substrate does trypsin break down?
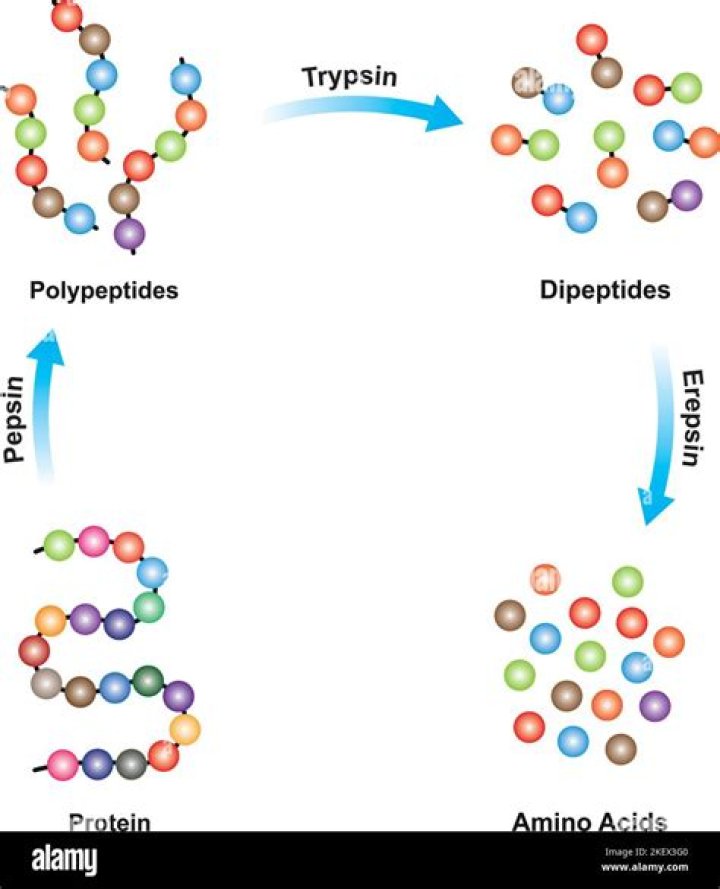
The function of Trypsin is to break down peptides using a hydrolysis reaction into amino acid building blocks. This mechanism is a general catalytic mechanism that all Serine proteases use. The active site where this mechanism occurs in Trypsin is composed of three amino acids and called a catalytic triad.
Which enzymes break down which foods?
- amylase and other carbohydrase enzymes break down starch into sugar.
- protease enzymes break down proteins into amino acids.
- lipase enzymes break down lipids (fats and oils) into fatty acids and glycerol.
What is the product of trypsin digestion?
The products of trypsin digestion are amino acids and various polypeptides. A rare, hereditary trypsinogen deficiency has been reported and results in the significant impairment of protein digestion.What foods contain papain?
- Kiwifruit.
- Ginger.
- Asparagus.
- Sauerkraut.
- Kimchi.
- Yogurt.
- Kefir.
What protein in milk is broken down by trypsin?
When casein (a protein in milk) is hydrolysed, the milk turns from cloudy to clear. Trypsin is one of the enzymes able to do this. To investigate the effect of temperature on the activity of trypsin – using casein as the substrate. 5cm³ distilled water in a fifth test tube and 5cm³ casein in a final test tube.
Where is trypsin found in the body?
It has long been known that trypsin is produced as a zymogen (trypsinogen) in the acinar cells of the pancreas, is secreted into the duodenum, is activated into the mature form of trypsin by enterokinase, and functions as an essential food-digestive enzyme.
How many amino acids are in trypsin?
Trypsin consists of a single polypeptide chain of 223 amino acids. The active site of trypsin include His46 and Ser183. The native form of trypsin is referred to as β-trypsin, which can be autolyzed to form α-trypsin and held together by disulfide bridges.Is trypsin acidic or alkaline?
The purified trypsin belonged to an alkaline protease, as it was highly active between pH 8.0 and 10.0 with the optimum pH of 9.0.
Is trypsin present in gastric juice?(i) pepsin, lipase, and rennin. (ii) trypsin, lipase, and rennin.
Article first time published onWhat does trypsin do in cell culture?
Trypsinization is the process of cell dissociation using trypsin, a proteolytic enzyme which breaks down proteins, to dissociate adherent cells from the vessel in which they are being cultured. When added to a cell culture, trypsin breaks down the proteins which enable the cells to adhere to the vessel.
Does trypsin digest carbohydrates?
Amylase, maltase, and lactase in the mouth digest carbohydrates. Trypsin and lipase in the stomach digest protein. Bile emulsifies lipids in the small intestine. No food is absorbed until the small intestine.
What is the means of trypsin?
Definition of trypsin : a proteolytic enzyme that is secreted in the pancreatic juice in the form of trypsinogen, is activated in the duodenum, and is most active in a slightly alkaline medium.
What enzymes break down vegetables?
Amylase helps to break down complex carbs, like those found in bread and cereals, while maltase helps to break down the malt sugar found in carbohydrate foods, like starchy grains and veggies.
What are proteins broken down to?
Once a protein source reaches your stomach, hydrochloric acid and enzymes called proteases break it down into smaller chains of amino acids. Amino acids are joined together by peptides, which are broken by proteases.
Does Mango have papain?
22.14, actinidin, Actinidia anionic protease, proteinase A2 of Actinidia chinensis) is a type of cysteine protease enzyme found in fruits including kiwifruit (genus Actinidia), pineapple, mango, banana and papaya. … This enzyme is part of the papain-like peptidase C1 family.
Why do Pineapples have bromelain?
Pineapples contain bromelain, a group of digestive enzymes that breaks down proteins. This may aid digestion, especially in those with pancreatic insufficiency.
What is papain used for?
Papain is sometimes used for sore throat. It is also used for insect bites, wound healing, diarrhea, and many other conditions, but there is no good scientific evidence to support these uses. In manufacturing, papain is used in cosmetics, toothpaste, contact lens cleaners, meat tenderizers, and meat products.
How do you get trypsin?
Preparation. Trypsin can be made from bacterial or fungal sources but it is most often extracted from the pancreas of pigs (called porcine trypsine). It can also be made from other meat-producing animal sources. Most commercially sold trypsin supplements are combined with other enzymes.
What happens if you drink trypsin?
It can cause side effects such as pain and burning. When taken by mouth: Not enough is known about the safety of trypsin for its other uses. Trypsin has been used in combination with other enzymes in clinical studies with no reports of serious adverse effects.
Can trypsin digest itself?
“Without efforts to stabilize it, trypsin will eventually digest itself,” says Tracy Adair-Kirk, Principal Scientist at MilliporeSigma. This is particularly undesirable for applications where autolysis may contaminate and confound experimental results.
Is trypsin can digest collagen?
Trypsin-2 is a neutral serine protease that directly degrades the triple helix of type II collagen [21-23].
What happens when trypsin is added to milk?
Trypsin can be used to break down casein in breast milk. If trypsin is added to a solution of milk powder, the breakdown of casein causes the milk to become translucent. The rate of reaction can be measured by using the amount of time needed for the milk to turn translucent.
Which enzyme helps in digestion of milk in stomach?
Chymosin, known also as rennin, is a proteolytic enzyme related to pepsin that synthesized by chief cells in the stomach of some animals. Its role in digestion is to curdle or coagulate milk in the stomach, a process of considerable importance in the very young animal.
What affects trypsin?
Trypsin is a serine protease which is secreted by the pancreas and is most active in the pH range between 7 and 9 at 37°C. It reacts with peptide bonds between the carboxylic acid group of lysine or arginine and the amino group of the adjacent amino acid residue.
What enzymes does trypsin activate?
Trypsinogen is activated by enterokinase, which cleaves an amino-terminal activation peptide (TAP). Active trypsin then cleaves and activates all of the other pancreatic proteases, a phospholipase, and colipase, which is necessary for the physiological action of pancreatic triglyceride lipase.
Is trypsin a liquid?
This liquid formulation of trypsin contains EDTA and phenol red. Gibco Trypsin-EDTA is made from trypsin powder, an irradiated mixture of proteases derived from porcine pancreas. Due to its digestive strength, trypsin is widely used for cell dissociation, routine cell culture passaging, and primary tissue dissociation.
What animals have trypsin?
Since its initial discovery, trypsin has been identified in all animals, including insects, fish, and mammals. Trypsin from each source can differ slightly in activity, but the natural substrate for the enzyme is generally any peptide that contains Lys or Arg.
How does trypsin digest casein?
Trypsin works in the small intestine, after acid and pepsin in the stomach have commenced the work of breaking down the proteins. This experiment uses milk which contains the protein casein. As the casein in milk break down, the smaller molecules become soluble, thereby reducing the opacity of the fluid.
Which peptide bonds does trypsin break?
Trypsin cleaves the peptide bond between the carboxyl group of arginine or the carboxyl group of lysine and the amino group of the adjacent amino acid. The rate of cleavage occurs more slowly when the lysine and arginine residues are adjacent to acidic amino acids in the sequence or cystine.
Which juice contains trypsin?
pancreas secrete pancreatic juice, which contains trypsin enzyme!