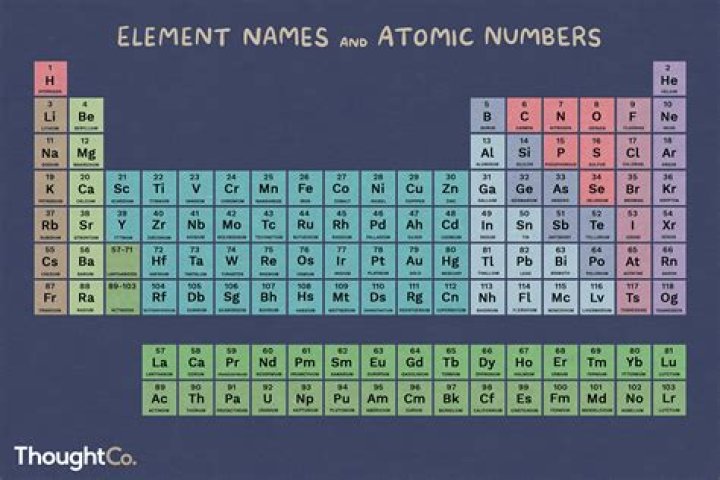
What element number is 90

Emma Valentine
Published Mar 02, 2026
thorium (Th), radioactive chemical element of the actinoid series of the periodic table, atomic number 90; it is a useful nuclear reactor fuel. Thorium was discovered (1828) by Swedish chemist Jöns Jacob Berzelius.
What element has an atomic mass of 9?
Atomic MassName chemical elementnumber14.0067Nitrogen715.9994Oxygen818.9984Fluorine920.1797Neon10
What element has a mass number of 91?
Atomic Mass231.03588uYear Discovered1913
What element has mass number?
NameHydrogenSymbolHAtomic Number1Protons1Neutrons0What is the 92 element?
Uranium is a silvery-white metallic chemical element in the periodic table, with atomic number 92. It is assigned the chemical symbol U. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons. Uranium has the highest atomic weight (19 kg m) of all naturally occurring elements.
What element has an atomic mass of 12?
Atomic NumberSymbolAtomic Weight (amu, g/mol)10Ne20.17911Na22.9897712Mg24.30513Al26.98154
Is thorium man made?
Thorium (chemical symbol Th) is a naturally occurring radioactive metal found at trace levels in soil, rocks, water, plants and animals. … There are natural and man-made forms of thorium, all of which are radioactive. In general, naturally occurring thorium exists as Th-232, Th-230 or Th-228.
What is a mass number in chemistry?
The mass number (symbol A, from the German word Atomgewicht [atomic weight]), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. … The mass number is different for each different isotope of a chemical element.What element has an atomic number of 7?
Atomic NumberSymbolName5BBoron6CCarbon7NNitrogen8OOxygen
How do you find the mass of an element?For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called the mass number. This is because each proton and each neutron weigh one atomic mass unit (amu). By adding together the number of protons and neutrons and multiplying by 1 amu, you can calculate the mass of the atom.
Article first time published onHow do I find the mass number?
Together, the number of protons and the number of neutrons determine an element’s mass number: mass number = protons + neutrons. If you want to calculate how many neutrons an atom has, you can simply subtract the number of protons, or atomic number, from the mass number.
Who discovered element 91?
atomic number91electron configuration of gaseous atomic state[Rn]5f26d17s2
What is 89 on the periodic table?
Actinium gives its name to a block of fifteen elements that lie between actinium and lawrencium in the periodic table with atomic numbers 89 through 103.
What are the 10 elements?
Name of the ElementSymbol of the ElementAtomic NumberNitrogenN7OxygenO8FluorineF9NeonNe10
Who discovered uranium 235?
Uranium-235 has a half-life of 703.8 million years. It was discovered in 1935 by Arthur Jeffrey Dempster.
What are uranium bombs?
The uranium hydride bomb was a variant design of the atomic bomb first suggested by Robert Oppenheimer in 1939 and advocated and tested by Edward Teller. It used deuterium, an isotope of hydrogen, as a neutron moderator in a uranium-deuterium ceramic compact.
What is the half life of uranium?
The half-life of uranium-238 is about 4.5 billion years, uranium-235 about 700 million years, and uranium-234 about 25 thousand years.
How many isotopes does polonium have?
There are 33 known isotopes (atoms of the same element with a different number of neutrons) of polonium, and all are radioactive.
Is radon a form of radiation?
Radon is a naturally occurring radioactive gas without color, odor, or taste that undergoes radioactive decay and emits ionizing radiation.
What is the mass of one atom?
1 Na molar mass:22.99 gTotal:58.44 g
What is element has 14 mass number and 7 of electrons?
The mass number of Nitrogen is 14, and its atomic number is 7.
What are the first 20 elements?
- H – Hydrogen.
- He – Helium.
- Li – Lithium.
- Be – Beryllium.
- B – Boron.
- C – Carbon.
- N – Nitrogen.
- O – Oxygen.
Is atomic mass a mass number?
Atomic mass is also known as atomic weight. Atomic mass is the weighted average mass of an atom of an element based on the relative natural abundance of that element’s isotopes. The mass number is a count of the total number of protons and neutrons in an atom’s nucleus.
What are examples of mass number?
The total number of protons and neutrons in the nucleus of an atom. For example, nitrogen has 7 protons and 7 neutrons in its nucleus, giving it a mass number of 14. Isotopes of elements are distinguished by their mass number; for example, carbon-12 and carbon-14 have mass numbers of 12 and 14 respectively.
How do you find the atomic mass of an element example?
To calculate the atomic mass of a single atom of an element, add up the mass of protons and neutrons. Example: Find the atomic mass of an isotope of carbon that has 7 neutrons. You can see from the periodic table that carbon has an atomic number of 6, which is its number of protons.
What does the atomic mass of an element represent?
The mass number tells us the number (the sum of nucleons) of protons and neutrons in the nucleus of an atom. The atomic number (also known as the proton number) is the number of protons found in the nucleus of an atom. It is traditionally represented by the symbol Z.
What's the most useless element?
Protactinium (formerly protoactinium) is a chemical element with the symbol Pa and atomic number 91.
Which is the rarest element on the Earth?
A team of researchers using the ISOLDE nuclear-physics facility at CERN has measured for the first time the so-called electron affinity of the chemical element astatine, the rarest naturally occurring element on Earth.
What element has a symbol of RN?
radon (Rn), chemical element, a heavy radioactive gas of Group 18 (noble gases) of the periodic table, generated by the radioactive decay of radium. (Radon was originally called radium emanation.)