What element number is 18

Emily Dawson
Published Mar 01, 2026
atomic number18atomic weight[39.792, 39.963]melting point−189.2 °C (−308.6 °F)boiling point−185.7 °C (−302.3 °F)density (1 atm, 0° C)1.784 g/litre
What is special about Group 18 on the periodic table?
The noble gases are the chemical elements in group 18 of the periodic table. They are the most stable due to having the maximum number of valence electrons their outer shell can hold. Therefore, they rarely react with other elements since they are already stable.
How many protons does element 18 have?
That would be an isotope of the noble gas argon, Ar, since the element has 18 protons — atomic number 18. Specifically, this is argon-39 or 39/18Ar, if you prefer. The atomic mass (upper number) is simply the sum of the protons and neutrons in the nucleus (18p + 21n).
What Krypton is used for?
Krypton is used commercially as a filling gas for energy-saving fluorescent lights. It is also used in some flash lamps used for high-speed photography. Unlike the lighter gases in its group, it is reactive enough to form some chemical compounds.Is argon gas harmful to humans?
Inhalation: This gas is inert and is classified as a simple asphyxiant. Inhalation in excessive concentrations can result in dizziness, nausea, vomiting, loss of consciousness, and death. Death may result from errors in judgment, confusion, or loss of consciousness which prevent self-rescue.
What are the Group 18 elements called?
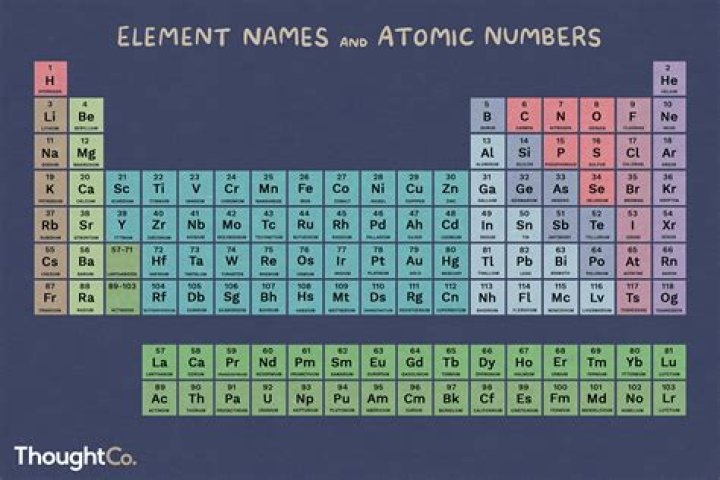
noble gas, any of the seven chemical elements that make up Group 18 (VIIIa) of the periodic table. The elements are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn), and oganesson (Og).
Is argon gas toxic?
A: Argon is an inert, non-toxic gas. It’s denser than oxygen and present in 1 percent of the air we breathe. It’s an extremely stable element with non-reactive properties.
Why are the element of group 18 known as noble gas?
The group 18 elements are helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). These elements are non-reactive and are called noble gases as they have their outermost orbit complete. Due to stable electronic configuration they hardly react with other elements.Is Group 18 A metal?
Group 18 of the periodic table contains the noble gases. These are the elements in the 18th column of the periodic table, at the far right. They are all nonmetals, and are found in their standard state as monatomic gases.
How many isotopes does krypton have?Natural krypton is a mixture of six stable isotopes: krypton-84 (56.99 percent), krypton-86 (17.28 percent), krypton-82 (11.59 percent), krypton-83 (11.5 percent), krypton-80 (2.29 percent), and krypton-78 (0.36 percent).
Article first time published onIs mercury a metal?
Elemental or metallic mercury is a shiny, silver-white metal, historically referred to as quicksilver, and is liquid at room temperature. It is used in older thermometers, fluorescent light bulbs and some electrical switches.
What is use for xenon?
Xenon is used in certain specialised light sources. It produces a beautiful blue glow when excited by an electrical discharge. Xenon lamps have applications as high-speed electronic flash bulbs used by photographers, sunbed lamps and bactericidal lamps used in food preparation and processing.
How many electrons does element 18 have?
So… for the element of ARGON, you already know that the atomic number tells you the number of electrons. That means there are 18 electrons in an argon atom.
What is the name of the element that has 18 protons?
Argon is a chemical element with the symbol Ar and atomic number 18.
What element has 18 protons and 22 neutrons and 18 electrons?
The chemical symbol for Argon. A 2 dimensional representation of a Argon atom with 18 protons and 22 neutrons in the nucleus and 18 electrons orbiting the nucleus.
Can oxygen be flammable?
Oxygen is not flammable, but it can cause other materials that burn to ignite more easily and to burn far more rapidly. The result is that a fire involving oxygen can appear explosive-like.
Is Neon a gas?
neon (Ne), chemical element, inert gas of Group 18 (noble gases) of the periodic table, used in electric signs and fluorescent lamps. Colourless, odourless, tasteless, and lighter than air, neon gas occurs in minute quantities in Earth’s atmosphere and trapped within the rocks of Earth’s crust.
Is it safe to work with aluminum?
Although not specifically recognized by OSHA, NIOSH, or other governmental entities as a hazardous metal, scientific studies have found that aluminum is harmful to workers.
Can argon tanks explode?
Tanks of argon may explode in the heat of a fire due to increased pressure within the tank.
Is acetylene a flammable gas?
Hazard Class: 2.1 (Flammable) Acetylene is a FLAMMABLE GAS. Stop flow of gas or let fire burn itself out. POISONOUS GASES ARE PRODUCED IN FIRE, including flammable Hydrogen gas. CONTAINERS MAY EXPLODE IN FIRE.
Which element is a noble gas and has 18 elements?
Noble gases2Neon (Ne) 103Argon (Ar) 184Krypton (Kr) 365Xenon (Xe) 54
Why is Group 18 not reactive?
Noble gases are nonreactive, nonmetallic elements in group 18 of the periodic table. Noble gases are the least reactive of all elements. That’s because they have eight valence electrons, which fill their outer energy level.
What is the charge of Group 18 on the periodic table?
Group 153-Group 162-Group 171-Group 18No common ions
What is the valence electrons of Group 18?
Periodic table groupValence ElectronsGroup 16 (VI) (chalcogens)6Group 17 (VII) (halogens)7Group 18 (VIII or 0) (noble gases)8**
How many valence electrons does Group 18 have?
Noble Gases The Group 18 elements are the noble gases. Atoms of the noble gases have 8 valence electrons, except for helium, which has 2.
How do you remember the elements in Group 18?
The group includes Helium (He), Neon (Ne), Argon (Ar), Krypton (Kr), Xenon (Xe), and the radioactive Radon (Rn). Mnemonic for Group 18: He Never Arrived; Kara Xero Run pe out.
How many shells does krypton have?
Atomic Number36Mass Number84Number of Neutrons48Shell structure (Electrons per energy level)[2, 8, 18, 8]Electron Configuration[Ar] 3d10 4s2 4p6
Is Diamond a metal?
Diamond is not considered as a non-metal in the exceptional category as diamond is a form of carbon. It is not classified as an element. … It is an allotrope of carbon.
Does mercury destroy gold?
Mercury dissolves many metals such as gold and silver to form amalgams.
Why is gold Au in the periodic table?
Gold symbol on the periodic table is Au that was used because of its Latin name aurum. It is an expensive metal, with a high boiling point, that is used as money and/ or jewelry.
What family is gold in?
Gold is a noble metal and falls into the transitional metal family on the periodic table in group number 11. The actual discoverer of gold is unknown as the metal has been in use by humans for over 5,000 years. Gold is extremely dense, weighing in at 19.3 grams per cubic centimeter.