What does v1 t1 v2 t2 mean

Robert Spencer
Published Mar 15, 2026
T1 = -173° C + 273 ° K = 100 ° K (temperatures used in Charles Law must be in Absolute Scale) V2 = 120 liters. T2 = 327° C + 273 ° K = 600 ° K.
What does T1 and t2 mean in Charles Law?
T1 = -173° C + 273 ° K = 100 ° K (temperatures used in Charles Law must be in Absolute Scale) V2 = 120 liters. T2 = 327° C + 273 ° K = 600 ° K.
What is the relationship between V1 and V2?
The relationship for Boyle’s Law can be expressed as follows: P1V1 = P2V2, where P1 and V1 are the initial pressure and volume values, and P2 and V2 are the values of the pressure and volume of the gas after change.
Which gas law is v1 t2 v2?
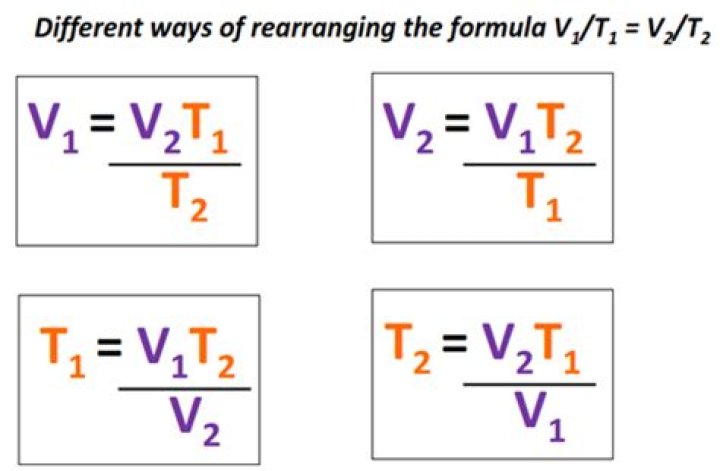
Charles’s Law V1/T1 = V2/T2 At constant amount of gas, as volume increases, its’ temperature increases and vice versa. Volume and temperature are directly proportional. /T1 n1 = P2V2/T2n2 The ratio between the pressure- volume product and the temperature of a system remains constant.What is the name of the following formula P1 T1 P2 t2?
Gay-Lussac s Law states that the pressure of a given mass of gas varies directly with the Kelvin temperature when the volume remains constant. Gay-Lussac s Law is expressed in a formula form as P1/T1=P2/T2. When dealing with Gay-Lussac s Law, the unit of the temperature should always be in Kelvin.
What does P1V1 T1 P2V2 T2 mean?
Gay-Lussac’s law – The pressure of a gas is directly proportional to the. temperature when volume and amount of substance is constant. P1/T1 = P2/T2. Combined gas law: P1V1/T1 = P2V2/T2 Use the gas laws for pressure, volume and temperature calculations. Avagadro’s Law and the Ideal Gas Law.
What is V2 in chemistry?
You can solve for the concentration or volume of the concentrated or dilute solution using the equation: M1V1 = M2V2, where M1 is the concentration in molarity (moles/Liters) of the concentrated solution, V2 is the volume of the concentrated solution, M2 is the concentration in molarity of the dilute solution (after …
How do you solve for V1?
- (C1) (V1) = (C2) (V2)
- Solve for V1 by dividing both sides by C1.
- (C1) (V1)/C1 = (C2) (V2)/C1, or V1 = (C2) (V2)/C1.
How is P1V1 T1 P2V2 T2?
Also same as before, initial and final volumes and temperatures under constant pressure can be calculated. P1V1 / T1 = P2V2 / T2 = P3V3 / T3 etc. Where n is the number of moles of the number of moles and R is a constant called the universal gas constant and is equal to approximately 0.0821 L-atm / mole-K.
What is the combined gas law for V2?Using the Combined Gas Law The easiest way is to derive the Boyle’s, Charles’ or the Gay-Lussac law. If the temperature is kept constant, the combined gas law reverts to P1V1=P2V2. That’s Boyle’s law! When we fix the pressure, we have V1T1=V2T2 which is Charles’ law.
Article first time published onWhat is the final pressure of the gas?
Gas LawsPressure or Volume Constant Temperature; TOriginal pressure x Original Volume=Final pressure x Final VolumeP1V1=P2V2 IsothermicPressure or temperature Constant Volume: VOriginal Pressure x Final Temperature=Final Pressure x Original TemperatureP1T2=P2T1 Isochoric
What is Boyle's Law graph?
The graph of Boyle’s law is called the PV curve. This graph of Boyle’s law is not straight but hyperbolic; this is because the statement says that at a constant temperature the pressure and the volume are inversely proportional. So when the pressure is increased the volume decreases and vice-versa.
What is constant in Charles Law?
Charles’s Law states that the Volume (V) of a gas is directly proportional to the temperature (T). This law is valid as long as the pressure and the amount of gas are constant. The temperature must be an absolute temperature: VT=k(constant) The constant, k, will depend on the number of moles and the pressure.
Does Charles law have to be in liters?
You can input any type of units but you must be consistent. For example, you can’t use cubic inches for volume 1 and liters for volume 2. Similar to Boyle’s Law, every Charles’ Law word problem always gives you three of the four variables you will need.
What happens to moles when pressure increases?
An increase in pressure on an equilibrium system favors the reaction which produces fewer total moles of gas. … This is because the overall number of gas molecules would increases and so would the pressure. A decrease in pressure on an equilibrium system favors the reaction which produces more total moles of gas.
What is r in the ideal gas law?
The factor “R” in the ideal gas law equation is known as the “gas constant”. R = PV. nT. The pressure times the volume of a gas divided by the number of moles and temperature of the gas is always equal to a constant number.
What is T1 in physics?
T1 (longitudinal relaxation time) is the time constant which determines the rate at which excited protons return to equilibrium. It is a measure of the time taken for spinning protons to realign with the external magnetic field.
What is c1 v1 c2 v2?
C1V1=C2V2 is used to calculate an unknown quantity where two solutions/mixtures are proportional … C1V1 = Concentration/amount (start) and Volume (start) C2V2 = Concentration/amount (final) and Volume (final) 1.
How do you find new molarity?
The equation for calculating Molarity from the moles and volume is very simple. Just divide moles of solute by volume of solution.
Does combined gas law have to be in Kelvin?
The relationships between volume and temperature and pressure and temperature expressed in the gas laws are only true when the kinetic energy of the molecules are directly proportional to the temperature. Therefore, when dealing with all gas laws, the temperatures must be expressed in Kelvin.
What happens to the particles in Boyle's law?
According to Boyle’s law, if the temperature of a gas is held constant, increasing the volume of the gas decreases its pressure. … As the volume of a gas increases, its particles have more room to spread out. This means that there are fewer particles bumping into any given area. This decreases the pressure of the gas.
How do you derive the combined gas law?
Derivation of the Combined Gas Law The combined gas law is an amalgamation of the three previously known laws which are- Boyle’s law PV = K, Charles law V/T = K, and Gay-Lussac’s law P/T = K. Therefore, the formula of combined gas law is PV/T = K, Where P = pressure, T = temperature, V = volume, K is constant.
How do you do dilutions?
To make a dilution, you simply add a small quantity of a concentrated stock solution to an amount of pure solvent. The resulting solution contains the amount of solute originally taken from the stock solution but disperses that solute throughout a greater volume.