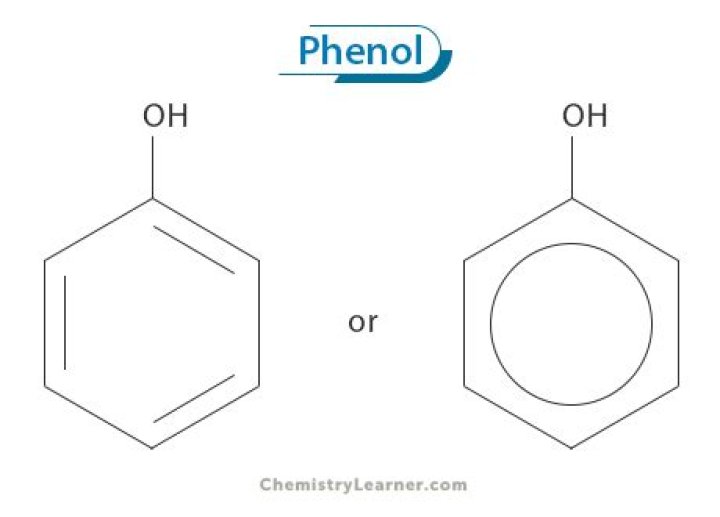
What does phenol look like

William Taylor
Published Mar 15, 2026
Phenols are similar to alcohols but form stronger hydrogen bonds. Thus, they are more soluble in water than are alcohols and have higher boiling points. Phenols occur either as colourless liquids or white solids at room temperature and may be highly toxic and caustic.
How do you identify phenol?
Dissolve the given organic compounds in water. Add neutral solution of ferric chloride slowly dropwise. Observe the change in colour. A red, blue, green or purple colouration indicates the presence of phenol.
What does phenol smell like?
Phenol has a distinct odor that is sickeningly sweet and tarry. You can taste and smell phenol at levels lower than those that are associated with harmful effects. Phenol evaporates more slowly than water, and a moderate amount can form a solution with water.
What is Colour of phenol?
Phenol is both a manufactured chemical and a natural substance. It is a colorless-to-white solid when pure.Where is phenol found?
Phenol is found to be present naturally in coal tar and creosote. It is also produced during natural fires, and through benzene degradation in the atmosphere under the influence of ultraviolet light radiation [14].
What happens to phenol red?
Phenol red is a water-soluble dye used as a pH indicator, changing from yellow to red over pH 6.6 to 8.0, and then turning a bright pink color above pH 8.1. As such, phenol red can be used as a pH indicator dye in various medical and cell biology tests.
Can you purchase phenol?
You can buy over-the-counter phenol spray almost anywhere. The most common brand is Chloraseptic. It contains about 1.4 percent phenol. Phenol spray is safe to use at the recommend dose for a short time.
Why does phenol turn red?
Phenol turns pink in colour on exposure to air due to slow oxidation.Does phenol turn red?
Chemical structure and properties A solution of phenol red is used as a pH indicator, often in cell culture. Its color exhibits a gradual transition from yellow (λmax = 443 nm) to red (λmax = 570 nm) over the pH range 6.8 to 8.2. Above pH 8.2, phenol red turns a bright pink (fuchsia) color. and is orange-red.
What does phenol do in the throat?Phenol is used to relieve pain and irritation caused by sore throat, sore mouth, or canker sores. This medicine is available without a prescription; however, your doctor may have special instructions on the proper use and dose for your medical problem.
Article first time published onIs phenol harmful to humans?
Phenol is highly irritating to the skin, eyes, and mucous membranes in humans after acute (short-term) inhalation or dermal exposures. Phenol is considered to be quite toxic to humans via oral exposure.
Are phenol and phenyl the same thing?
The key difference between phenol and phenyl is that phenol has a hydroxyl group whereas phenyl has no hydroxyl group. Phenol is an aromatic alcohol. It has the chemical formula C6H5OH. … Phenyl is a derivative of phenol; if we remove the hydroxyl group from the phenol molecule, it gives phenyl group.
What products is phenol found in?
Phenol is a very common chemical and is regularly found in the following common products: air fresheners, aftershave, bronchial mists, chloroseptic throat spray, deodorants, feminine powders & sprays, hair spray, decongestants, mouthwash, aspirin, solvents, acne medications, antiseptics, calamine lotions, cleaning …
Is phenol a poison?
Phenol is a severe irritant and highly toxic systemic poison that is absorbed well by inhalation and through the skin.
What does diabetic smell like?
If your breath smells like acetone — the same fruity scent as nail polish remover — it may be a sign of high levels of ketones (acids your liver makes) in your blood. It’s a problem mainly of type 1 diabetes but also can happen with type 2 if you get a serious condition called diabetic ketoacidosis (DKA).
Is Lysol a phenol?
Other members of phenol group: Phenol has several derivates, namely, cresol, creosote, lysol, and dettol. … Cresol is a methyl phenol with meta, ortho, and para isomers. It is used as a disinfectant and antiseptic.
What disinfectants contain phenol?
Phenolic disinfectants (e.g. Meytol, Dettol, etc.) are widely used for domestic purposes.
What is the another name of phenol?
noun Chemistry. Also called carbolic acid, hydroxybenzene, oxybenzene, phenylic acid. a white, crystalline, water-soluble, poisonous mass, C6H5OH, obtained from coal tar, or a hydroxyl derivative of benzene: used chiefly as a disinfectant, as an antiseptic, and in organic synthesis.
Is phenol good for skin?
For natural skincare, plant based phenols are a golden standard. They are rich in antioxidants, as well as anti-inflammatory, anti-bacterial, and anti-septic properties. Said to dramatically repair and rejuvenate the skin, high-end skincare lines are infusing their products with these phenols.
How will you handle phenol Why?
As with any chemical, do not eat, drink, or smoke where phenol is handled, processed, or stored, since the chemical could be ingested. Store phenol in a cool, dry, well-ventilated area, away from heated surfaces or ignition sources. Always wash hands thoroughly after handling phenol, even if gloves are used.
What is the most common use of phenol derivatives in health care?
Phenol has been used to disinfect skin and to relieve itching. Phenol is also used as an oral analgesic or anesthetic in products such as Chloraseptic to treat pharyngitis. Additionally, phenol and its related compounds are used in surgical ingrown toenail treatment, a process termed phenolization.
What is phenol yellow?
Phenol red is a pH indicator that is yellow at a pH below 6.8 and red at a pH above 7.4 with varying shades from yellow to red between those pH levels. If the indicator has turned yellow in the bottle this means it has become contaminated with something that has made the pH more acidic and brought the pH below 6.8.
What is happening when a phenol red broth turns pink?
A pink color in the Phenol Red broth indicates the microorganism does not have the ability to… What metabolic processes are occurring when one observes a yellow Phenol Red broth tube? At what acid pH does Phenol Red turn yellow?
What does phenol red smell like?
Phenol has a distinct odor that is sickeningly sweet and tarry. You can taste and smell phenol at levels lower than those that are associated with harmful effects. Phenol evaporates more slowly than water, and a moderate amount can form a solution with water.
What Colour is phenol red?
Phenol red, also known as phenolsulfonphthalein, is a pH indicator dye that exhibits a gradual transition from yellow to red over a pH range of 6.2 to 8.2 (Figure 2). Above 8.2 the dye turns a bright fuchsia color.
Why is phenol red yellow?
Phenol red is a pH indicator which turns yellow below a pH of 6.8 and fuchsia above a pH of 7.4. If the organism is able to utilize the carbohydrate, an acid by-product is created, which turns the media yellow. … When the organism is able to use the carbohydrate, a gas by-product may be produced.
Why are phenols Coloured?
Alcohols, Phenols and Ethers On long standing in contact with air, phenol gets slowly oxidised. Hence, it get coloured.
Why is phenol kept in brown bottle?
Phenol discolors quickly when in contact with iron or copper. … Phenol also darkens when exposed to light, so whenever possible reagent containers should be kept in the dark and nitrogen-blanketed to minimize color development.
How do you dispose of phenol red?
Offer surplus and non-recyclable solutions to a licensed disposal company. Contact a licensed professional waste disposal service to dispose of this material. Dispose of as unused product. SARA 302: No chemicals in this material are subject to the reporting requirements of SARA Title III, Section 302.
What happens when you mix baking soda calcium chloride and phenol red?
Calcium chloride, baking soda, and water combine to produce carbon dioxide gas. Phenol red is an acid–base indicator that changes color in the presence of acids and bases. Sodium bicarbonate is a base, so it maintains the bright red (basic) color of phenol red at first.
How much phenol is toxic?
Phenol is a general protoplasmic poison (denatured protein) with corrosive local effects. Phenol derivates are less toxic than pure phenol. The lethal dose is between 3 to 30 g, but may be as little as 1 g. Phenol is well absorbed by inhalation, dermal application, and ingestion.