What does ion trapping do

Mia Kelly
Published Feb 25, 2026
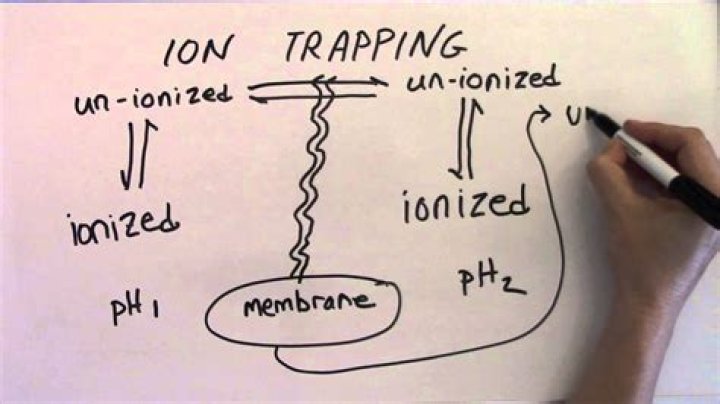
Ion trapping alters the urine pH to inhibit reabsorption of toxins across the renal tubular membranes. The principle behind this approach is to “trap” the toxin in its ionized form in the urine where it can be excreted.
What is ionized and unionized drugs?
Most drugs are weak acids or bases that are present in solution as both the ionized and unionized forms. Ionized molecules are usually unable to penetrate lipid cell membranes because they are hydrophilic and poorly lipid soluble. Unionized molecules are usually lipid soluble and can diffuse across cell membranes.
What is the pH partition theory?
pH partition is the tendency for acids to accumulate in basic fluid compartments, and bases to accumulate in acidic compartments. The reason for this phenomenon is that acids become negatively electric charged in basic fluids, since they donate a proton.
Do acidic drugs ionized in basic media?
Basic drugs get ionized in acidic medium (right shift of eq 2), thus this form is poorly absorbed. Aspirin, an acidic drug is unionized in acidic medium of stomach, so is easily absorbed.How does ion trapping affect drug distribution?
Ion trapping is the reason why basic (alkaline) drugs are secreted into the stomach (for example morphine), where pH is acidic, and acidic drugs are excreted in urine where the conditions are alkaline.
How do you acidify urine poisoning?
Most experts alkalinize the urine by giving an initial intravenous bolus of 1 mEq/kg of sodium bicarbonate and then start a sodium bicarbonate intravenous infusion.
What is an acidic drug?
Abstract. Strong acidic drugs are a class of chemical compounds that normally have high hydrophilicity and large negative charges, such as organophosphatic compounds and organosulphonic compounds.
How do you tell if a drug is ionized?
Ionized (or charged) drugs are not absorbed as efficiently as un-ionized drugs are. Practically speaking, this means that if taken orally, a drug that is a weak acid will be absorbed primarily in the acidic environment; whereas, a drug that is a weak base will be absorbed in the alkaline environment small intestines.What are alkaline drugs?
Specialty. nephrology. Alkalinizing agents are drugs used to manage disorders associated with low pH. For example, they may be used to treat acidosis due to kidney failure. Used for oral or parenteral therapy, sodium bicarbonate is the commonly preferred alkalinizing agent.
Is aspirin ionized in the stomach?Aspirin was not absorbed by the stomach at pH 6.5. These experiments indicate that aspirin can be absorbed to an appreciable extent in its ionized form by the small intestine but not by the stomach.
Article first time published onHow do you tell if a drug is basic or acidic?
The only sure way to know whether a drug is acidic or basic is to learn the functional groups that confer acidity and basicity on a molecule. This should be done even if it means learning the names of the functional groups the way you learned multiplication tables at primary school.
Is pH a pKa?
The pKa is the pH value at which a chemical species will accept or donate a proton. The lower the pKa, the stronger the acid and the greater the ability to donate a proton in aqueous solution.
What could be the reason that irrespective of pH any drug gets absorbed mostly from the intestine?
What could be the reason that irrespective of pH any drug gets absorbed mostly from the intestine? Explanation: Experiments showed that irrespective of the GI pH and the degree of ionization, most of the acidic and basic drugs are more rapidly absorbed from the intestine.
What route of drug absorption has the greatest bioavailability?
Intravenous (IV) drug administration is assumed to be the most dependable and accurate route for drug delivery, with a bioavailability of 100%. Absorption of drugs from tissues and organs (e.g., intramuscular, transdermal, rectal) can also be affected by development ( Table 73.2 ).
What is the significance of pH partitioning in pharmaceutical system?
1957) proposed the pH – partition theory to explain the influence of GI pH and drug pKa on the extent of drug transfer or drug absorption. Brodie reasoned that when a drug is ionized it will not be able to get through the lipid membrane, but only when it is non ionized and therefore has a higher lipid solubility. i.e.
What are limitations of pH-partition theory?
The limitations in the predictive power of pH-partition theory arise because it only accounts for the pH(i)/pH(e)-sensitivity of drug entry into cells but not the drug’s subsequent interactions that, independently, show pH(i)-dependence.
Who poorly developed BBB?
3. Who has poorly developed BBB? Explanation: Infants are newly born. Their BBB is poorly developed.
Is naproxen a weak acid or base?
A naproxen molecule has a carboxylic group and so is a weak acid. It ionises in water to give an equilibrium solution that contains a mixture of unionised molecules, carboxylate ions and hydroxonium ions (figure 2).
Which of the following is a weakly acidic drug?
Weakly acidic drugs (partition coefficient) ibuprofen (3.6), aceclofenac (3.9), glipizide (1.9) and weakly basic drugs olanzapine (3.6), telmisartan (6.0), and sildenafil citrate (1.9) were selected for the study.
What is volume of distribution of drugs?
Volume of distribution (Vd), represents the apparent volume into which the drug is distributed to provide the same concentration as it currently is in blood plasma. It is calculated by the amount of the drug in the body divided by the plasma concentration [19].
Is Honey alkaline or acidic?
Scientists have recorded a pH level of between 3.3 to 6.5 for different types of honey, so honey is therefore acidic.
Is aspirin acidic or basic?
Aspirin itself is an acidic medicine and causes gastric irritation and regurgitation which can lead to low oral pH levels [7].
Is aspirin a weak acid?
Aspirin is a weak acid and it tends to ionize (give up a H atom) in an aqueous medium at high pH. … In a low pH environment like the stomach (pH =2), aspirin is predominantly unionized and crosses membranes into the blood vessels readily.
Does lemon juice make urine alkaline?
Once digested and metabolized, lemon juice produces alkaline byproducts, which make the urine more alkaline. This is why some people think of it as alkalizing, despite its acidic pH before it is digested.
How can I make my urine alkaline fast?
To help make your urine more acid you should avoid most fruits (especially citrus fruits and juices), milk and other dairy products, and other foods which make the urine more alkaline. Eating more protein and foods such as cranberries (especially cranberry juice with vitamin C added), plums, or prunes may also help.
How do I Alkalize my urine?
A diet rich in citrus fruits, most vegetables, and legumes will keep the urine alkaline. A diet high in meat and cranberry juice will keep the urine acidic. Urine pH is an important screening test for the diagnosis of renal disease, respiratory disease, and certain metabolic disorders.
Is lemon water alkaline?
Lemon juice is acidic, with a pH of 3, while water has a pH of around 7, which is neutral. This means it is neither acidic nor alkaline.
What food is high alkaline?
- Green Leafy Vegetables. Green leafy veggies are said to have an alkaline effect in the body. …
- Cauliflower/Broccoli. Cruciferous vegetables like cauliflower and broccoli are known to balance alkaline pH in the system. …
- Citrus Fruits. …
- Root Vegetables. …
- Nuts.
Which medication is used to keep urine alkaline?
Sodium bicarbonate is used intravenously to alkalinize urine.
Why ionized drugs are not absorbed?
The fatty acid chains repel ionized, charged molecules. This means that the ionized molecules cannot pass through the intestinal membrane and be absorbed.
What is good solubility for a drug?
A drug is considered highly soluble when the highest dose strength is soluble in 250 mL or less of aqueous media over the pH range of 1 to 7.5.