What do you mean by isomer

Sophia Edwards
Published Mar 14, 2026
The roots of the word isomer are Greek—isos plus meros, or “equal parts.” Stated colloquially, isomers are chemical compounds that have the same parts but are nonetheless not the same. … Such structures also would be analogous to isomers.
What are isomers explain with two examples?
Isomers. 1. Isomers are molecules that have the same molecular formula, but have a different arrangement of the atoms in space.
What is a isomer in biology?
Isomers are molecules with the same chemical formula but have different structures, which creates different properties in the molecules.
What is isomer Class 10 example?
Two or more compound having same molecular formula but different structural Formulas are called isomers and process is called Isomerism.. For Example: Pentane, C5H12, has three chain isomers.What is isomerism Slideshare?
What is Isomerism? The organic compounds having the same molecular formula but different structures are known as Isomers. This phenomenon is known as Isomerism. In other words, the organic compounds having the same molecular formula but different arrangements of carbon atoms in them, are known as Isomers.
What is isomerism class 11th?
Isomerism is the phenomenon in which more than one compounds have the same chemical formula but different chemical structures. Chemical compounds that have identical chemical formulae but differ in properties and the arrangement of atoms in the molecule are called isomers.
What are the 3 types of isomers?
There are three types of structural isomers: chain isomers, functional group isomers and positional isomers. Chain isomers have the same molecular formula but different arrangements or branches. Functional group isomers have the same formula but different functional groups.
Is isomerism important for Class 12?
If you are preparing for class 12th board examination then you don’t need to learn stereoisomerism before learning class 12th organic chemistry. Structural Isomerism is sufficient for understanding class 12th Organic Chemistry.What are isomers Class 12?
CBSE NCERT Notes Class 12 Chemistry Coordination Compounds. Isomerism is the phenomenon in which compounds have same molecular formula but different structures and these different structures are called as isomers. Isomers are those that have different physical and chemical properties.
What is isomer physics?isomer, in nuclear physics, any of two or more nuclides (species of atomic nuclei) that consist of the same number of protons and the same number of neutrons but differ in energy and manner of radioactive decay, and that exist for a measurable interval of time.
Article first time published onWhat is isomerism in protein?
isomerism, the existence of molecules that have the same numbers of the same kinds of atoms (and hence the same formula) but differ in chemical and physical properties.
What are the 4 types of isomers?
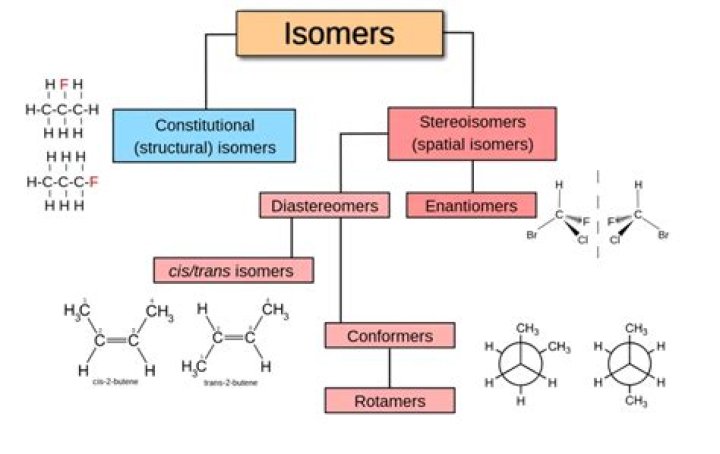
Types of Isomers: Constitutional Isomers, Stereoisomers, Enantiomers, and Diastereomers.
What is isomerism explain its types?
Isomerism is a consequence of the fact that the atoms of a molecular formula can be arranged in different ways to give compounds, called isomers, that differ in physical and chemical properties. There are two types of isomerism: structural isomerism and stereoisomerism, which can be divided into further subtypes.
What is the use of isomerism?
Currently, knowledge of isomerism has helped us in introducing safer and more effective drug alternatives of the newer as well as existing drugs. Many existing drugs have gone chiral switch i.e., switching from racemic mixture to one of its isomers.
What is hybridization Slideshare?
Hybridisation (or hybridization) is the concept of mixing atomic orbitals into new hybrid orbitals (with different energies, shapes, etc., than the component atomic orbitals) suitable for the pairing of electrons to form chemical bonds in valence bond theory.
What is an isomer for kids?
An isomer in chemistry is a chemical that has the same molecular formula as another, but it has a different arrangement of atoms in space. Different isomers have different chemical properties (that is, they may look, or smell, or taste different from each other).
How are isomers named?
The number of carbons are indicated by a prefix and the suffix -ane is added to indicate the molecules is an alkane. The prefix for three carbons is prop so adding -ane, the IUPAC name for C3H8 is propane. Likewise, the prefix for six is hex so the name for the straight chain isomer of C6H14 is called hexane.
What are the two main types of isomerism?
Two main forms of isomerism are structural or constitutional isomerism, in which bonds between the atoms differ; and stereoisomerism or spatial isomerism, in which the bonds are the same but the relative positions of the atoms differ.
What is position isomer?
Positional isomers are constitutional isomers that have the same carbon skeleton and the same functional groups but differ from each other in the location of the functional groups on or in the carbon chain. eg. 1: Propyl bromide (1) and isopropyl bromide (2) are constitutional isomers.
What is isomer of pentane?
Pentane has three structural isomers that are n-pentane, Iso-pentane (methyl butane) and neopentane (dimethylpropane).
What is geometrical isomerism Class 12?
Answer: Geometrical isomerism is a type of stereoisomerism having the same molecular formula and same structure but differ in the relative arrangement of atoms.
What is geometrical isomerism in chemistry?
Geometric isomers are two or more coordination compounds which contain the same number and types of atoms, and bonds (i.e., the connectivity between atoms is the same), but which have different spatial arrangements of the atoms. Not all coordination compounds have geometric isomers.
What is ring chain isomer?
Ring-chain isomerism is a process which involves the transformation of molecule from an open-chain structure into a ring structure. 1 Such isomerization is classified into valence isomerism and additive isomerism.
What is meant by functional isomerism?
Functional isomerism is the type of isomerism in which the compounds that have the same molecular formula, but have different functional groups. … This kind of isomerism deals with the spatial arrangement or orientation of molecules atoms in a compound in space.
What is isomers in radiology?
Isomerism is a term which in general means ‘mirror-image’ and refers to finding normally-asymmetric bilateral structures to be similar. It is used in the context of heterotaxy and is of two types: left isomerism.
What is electron decay?
Title:Electron Decay Abstract: The electron would decay into a photon and neutrino if the law of electric charge conservation is not respected. Such a decay would cause vacancy in closed shells of atoms giving rise to emission of x-rays and Auger electrons.
What is structural isomerism?
In chemistry, a structural isomer (or constitutional isomer in the IUPAC nomenclature) of a compound is another compound whose molecule has the same number of atoms of each element, but with logically distinct bonds between them.
How are isomers formed?
Isomers are compounds that contain exactly the same number of atoms, i.e., they have exactly the same empirical formula, but differ from each other by the way in which the atoms are arranged.