What do u mean by polarity

Victoria Simmons
Published Mar 14, 2026
polarity, in chemical bonding, the distribution of electrical charge over the atoms joined by the bond. … In hydrogen chloride, for example, the hydrogen atom is slightly positively charged whereas the chlorine atom is slightly negatively charged.
What do you mean by polarity in chemistry?
polarity, in chemical bonding, the distribution of electrical charge over the atoms joined by the bond. … In hydrogen chloride, for example, the hydrogen atom is slightly positively charged whereas the chlorine atom is slightly negatively charged.
What is mean by polarity in physics?
noun. Physics. the property or characteristic that produces unequal physical effects at different points in a body or system, as a magnet or storage battery. the positive or negative state in which a body reacts to a magnetic, electric, or other field.
What is polarity in simple words?
1 : the quality or condition inherent in a body that exhibits opposite properties or powers in opposite parts or directions or that exhibits contrasted properties or powers in contrasted parts or directions : the condition of having poles. 2 : attraction toward a particular object or in a specific direction.What is meant by polarity in electrical?
Electrical polarity is a term used throughout industries and fields that involve electricity. … This represents the electric potential at the ends of a circuit. A battery has a positive terminal (+ pole) and a negative terminal (− pole).
What is polarity and non polarity?
When things are different at each end, we call them polar. Some molecules have positive and negative ends too, and when they do, we call them polar. If they don’t, we call them non-polar. Things that are polar can attract and repel each other (opposite charges attract, alike charges repel).
What does polarity mean in water?
Water is a “polar” molecule, meaning that there is an uneven distribution of electron density. Water has a partial negative charge ( ) near the oxygen atom due the unshared pairs of electrons, and partial positive charges ( ) near the hydrogen atoms.
What causes polarity?
Polarity results from the uneven partial charge distribution between various atoms in a compound. Atoms, such as nitrogen, oxygen, and halogens, that are more electronegative have a tendency to have partial negative charges. … A polar molecule results when a molecule contains polar bonds in an unsymmetrical arrangement.What is non polarity?
A nonpolar molecule has no separation of charge, so no positive or negative poles are formed. … In a polar molecule, one side of the molecule has a positive electrical charge and the other side has a negative electrical charge.
What is polarity in chemistry class 11?Define Polarity. “A state or a condition of an atom or a molecule having positive and also negative charges, especially in case of magnetic or an electrical poles.” Polarity, in general, refers to the physical properties of compounds such as boiling point, melting points, and their solubilities.
Article first time published onWhat is polarity and switching?
Reverse Polarity Switching – a clever sounding term for something that’s really very simple. Polarity refers to the direction of a magnetic or electrical field. … Thus each terminal of the battery is said to have a ‘polarity’ and this polarity is either positive or negative.
What is the polarity of a bar magnet?
A bar magnet suspended in Earth’s magnetic field orients itself in a north–south direction. The north-seeking pole of such a magnet, or any similar pole, is called a north magnetic pole. The south-seeking pole, or any pole similar to it, is called a south magnetic pole.
Does AC have polarity?
AC voltage has no polarity. Therefore it does not matter how you connect the wires to the bridge rectifier.
Is water dipolar or polar?
Water molecules are polar, with partial positive charges on the hydrogens, a partial negative charge on the oxygen, and a bent overall structure.
Is glucose polar?
Sugars (e.g., glucose) and salts are polar molecules, and they dissolve in water, because the positive and negative parts of the two types of molecules can distribute themselves comfortably among one another.
How do you show the polarity of water?
Lay the penny on a flat surface and slowly drop water onto it. The water molecules stick together and form a convex shape on the penny, like a bowl placed upside down. This is due to the bonding or attraction of the positive and negative hydrogen molecules found in water. Try the same with oil, which is nonpolar.
What is polar or nonpolar?
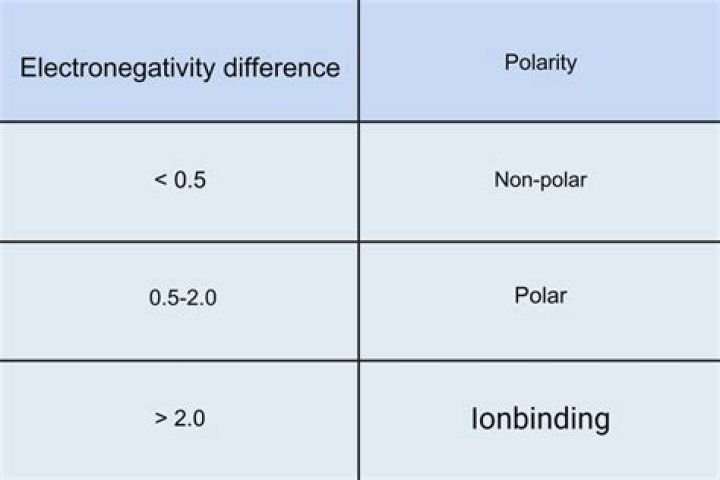
Polar molecules occur when there is an electronegativity difference between the bonded atoms. Nonpolar molecules occur when electrons are shared equal between atoms of a diatomic molecule or when polar bonds in a larger molecule cancel each other out.
Is soap polar or nonpolar?
Soap is effective as a cleaning agent because it is amphiphilic; it is partly polar and partly nonpolar. Soap molecules contain an intensely polar “head” (the ionic part) and a non-polar “tail” (the long hydrocarbon chain, usually 10-18 carbons, depending on which fatty acid is used).
Is CO2 polar or non-polar?
Polar molecules have a non-zero net dipole moment. Both CO2 and H2O have two polar bonds. However the dipoles in the linear CO2 molecule cancel each other out, meaning that the CO2 molecule is non-polar.
Are oils polar or nonpolar?
Oils, by contrast, are nonpolar, and as a result they’re not attracted to the polarity of water molecules. In fact, oils are hydrophobic, or “water fearing.” Instead of being attracted to water molecules, oil molecules are repelled by them.
What are two types of polarity?
4.6. The polarity condition of the electrodes is of two types, (1) straight polarity and (2) reverse polarity.
What causes high polarity?
A molecule may be polar either as a result of polar bonds due to differences in electronegativity as described above, or as a result of an asymmetric arrangement of nonpolar covalent bonds and non-bonding pairs of electrons known as a full molecular orbital.
What causes polarity kid definition?
Chemical polarity is a feature of chemical bonds, where two different atoms in the same molecule have different electronegativity. As a result, the electrons in the bond are not shared equally by the two atoms. This causes an asymmetrical (polar) electric field. … Entire molecules can also be described as polar.
What is a polar molecule class 10?
A polar molecule is a particle-containing polar bond where the whole of all the bond’s dipole minutes isn’t zero. Polar bonds structure when there is a distinction between the electronegativity estimations of the iotas partaking in a bond.
What do polarity symbols mean?
Polarity symbols are a notation for electrical polarity, found on devices that use direct current (DC) power, when this is or may be provided from an alternating current (AC) source via an AC adapter.
What is wrong polarity?
What’s reverse polarity? If your outlet’s polarity is reversed, it means that the neutral wire is connected to where the hot wire is supposed to be. … There is always electricity flowing out of an outlet with reversed polarity, even if an appliance is supposed to be off.
What does polarity sensitive mean?
Electrons flow from negative to positive. In a direct current (DC) circuit, current flows in one direction only, and one pole is always negative and the other pole is always positive. Hence the equipments are polar sensitive.
Why is North Pole magnetic?
The data showed that the position of the north magnetic pole is determined largely by a balance, or tug-of-war, between two large lobes of negative flux at the boundary between Earth’s core and mantle under Canada.
Is north positive or negative?
When magnets are used in magnetic therapy, the poles are often referred to as being positive or negative. Generally, the south pole is termed positive, and the north negative.
How does the oceanic crust record polarity?
When lava is extruded at any mid-ocean ridge, the rock it forms becomes magnetized and acquires the magnetic polarity that exists at the time the lava cools. As the crust moves away from mid-ocean ridges, it contains a continuous record of the Earth’s changing magnetic polarity.
What is positive polarity?
positive polarity in British English noun. grammar. the grammatical characteristic of a word or phrase, such as delicious or rather, that may normally only be used in a semantically or syntactically positive or affirmative context.