What charge do protons have

Robert Spencer
Published May 05, 2026
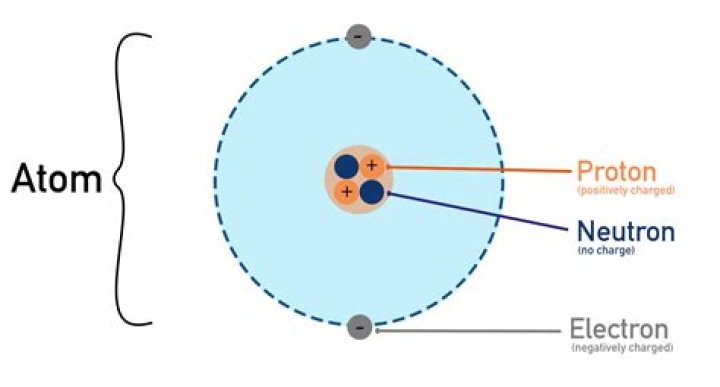
A proton carries a positive charge (+) and an electron carries a negative charge (-), so the atoms of elements are neutral, all the positive charges canceling out all the negative charges. Atoms differ from one another in the number of protons, neutrons and electrons they contain.
What is the charge of a proton?
proton, stable subatomic particle that has a positive charge equal in magnitude to a unit of electron charge and a rest mass of 1.67262 × 10−27 kg, which is 1,836 times the mass of an electron.
What charge does the electron have?
electron, lightest stable subatomic particle known. It carries a negative charge of 1.602176634 × 10−19 coulomb, which is considered the basic unit of electric charge. The rest mass of the electron is 9.1093837015 × 10−31 kg, which is only 1/1,836the mass of a proton.
What charge does a neutrons have?
neutron, neutral subatomic particle that is a constituent of every atomic nucleus except ordinary hydrogen. It has no electric charge and a rest mass equal to 1.67493 × 10−27 kg—marginally greater than that of the proton but nearly 1,839 times greater than that of the electron.Why are protons charged?
Proton is made up of two up quarks(+4/3) and one down quark(-1/3) and due to this the net charge is +1 on protons and that is the reason protons are positively charged. While neutrons are made up of one Up quark(+2/3) and two down quarks(-2/3) due to this the net charge is 0(zero) on a neutron. Because of this n…
What is the charge of a single proton quizlet?
The charge on a single proton is +1.6 x 10 -19 Coulombs.
Does proton have a positive charge?
A proton carries a positive charge (+) and an electron carries a negative charge (-), so the atoms of elements are neutral, all the positive charges canceling out all the negative charges. Atoms differ from one another in the number of protons, neutrons and electrons they contain.
What is proton neutron electron?
Atoms are made of extremely tiny particles called protons, neutrons, and electrons. Protons and neutrons are in the center of the atom, making up the nucleus. Electrons surround the nucleus. Protons have a positive charge. Electrons have a negative charge.What are protons electrons?
Electrons are a type of subatomic particle with a negative charge. Protons are a type of subatomic particle with a positive charge. Protons are bound together in an atom’s nucleus as a result of the strong nuclear force. … The positive charge on a proton is equal in magnitude to the negative charge on an electron.
What is the charge of a proton in coulombs?One coulomb is equal to the amount of charge from a current of one ampere flowing for one second. One coulomb is equal to the charge on 6.241 x 1018 protons. The charge on 1 proton is 1.6 x 10-19 C.
Article first time published onWhy do electrons and protons have the same charge?
In other words, particle is modifying space or time or universe strings to demonstrate charge. And it does it to full saturation. This is why all electrons and all protons that we did measure have the same value.
Do neutrons have a positive charge?
Among atomic particles, the neutron seems the most aptly named: Unlike the positively charged proton or the negatively charged electron, neutrons have a charge of zero.
Which particle has a positive charge?
There are three subatomic particles: protons, neutrons and electrons. Two of the subatomic particles have electrical charges: protons have a positive charge while electrons have a negative charge.
What is the charge of 5 protons?
5 protons, 5 neutrons, 5 electrons Charge -1, 18 electrons, mass number 36.
Do protons have a negative charge?
For example, electrons have negative charge and protons have positive charge, but neutrons have zero charge. The negative charge of each electron is found by experiment to have the same magnitude, which is also equal to that of the positive charge of each proton.
Why do neutrons have no charge?
Like all hadrons, neutrons are made of quarks. A neutron is made of two down quarks and one up quark. One up quark has a charge of +2/3, and the two down quarks each have a charge of -1/3. The fact that these charges cancel out is why neutrons have a neutral (0) charge.
What is the total charge of 1 proton 1 neutron and 1 electron?
Elementary ParticleChargeMassProton+11Neutron01Electron−1~0
Why are protons considered positive?
The charge on the proton has exactly the same value as that of the electron, but is positive rather than negative. It is the balance between these equal yet opposite charges of the protons and electrons that assures the electric neutrality of the atom.
What is the ionic charge of an ion with 13 protons and 10 electrons?
The aluminum ion has 10 electrons and 13 protons, so it has lost three electrons, and has a charge of 3+ .
Does a proton have a mass of 1 amu?
Protons – Protons carry a single positive charge that have a mass of one approximately one atomic mass unit. … An electron is very small, about 1/1,837th the mass of a proton. Atomic Weights Atomic mass of an atom is expressed in atomic mass units (amu) which is approximately 1.66×10-24 grams.
What type of charge does an electron have quizlet?
-Electrons have a negative charge.
Why do electrons have a charge?
I can’t really answer this question, but based on experiments carried out by scientists, electrons have a negative charge because they are attracted to anodes (a positively charged electrode). We know that opposite charges attract. Electrons are known as fundamental particles. Electron does not acquire charge.
Why are electrons negatively charged?
Electric charge is a physical property of matter. It is created by an imbalance in a substance’s number of protons and electrons. The matter is positively charged if it contains more protons than electrons, and it is negatively charged if it contains more electrons than protons.
What are protons made of?
Protons and neutrons are composed of two types: up quarks and down quarks. Each up quark has a charge of +2/3. Each down quark has a charge of -1/3. The sum of the charges of quarks that make up a nuclear particle determines its electrical charge.
Are protons and electrons equal?
An atom contains equal numbers of protons and electrons . Since protons and electrons have equal and opposite charges , this means that atoms are neutral overall.
What is the charge of 6 protons in coulombs?
Object# of Excess Protons/ElectronsQuantity and Kind of Charge (Q) on Object in Coulombs (C)B1 x 106 excess protons+1.6 x 10-13 C
How do you find the charge of coulombs?
The unit of electric charge is a Coulomb (symbol: C). It is defined as the charge that is transported by a constant current of 1 ampere during 1 second. Hence, 1 C = 1 A * 1 s expressed in SI units. If you don’t remember what an ampere is, head to our Ohm’s law calculator.
What is a proton state the charge on a proton in coulombs and mass in grams?
A proton has a charge of +1, or 1e, which is equal to 1.602 x 10^-19 coulombs, and a mass of 1.67 x 10^-27 kg.
Are all protons the same?
All protons are identical. For example, hydrogen protons are exactly the same as protons of helium and all other elements, or pure substances. However, atoms of different elements have different numbers of protons. … The number of protons in an atom determines the electrical charge of the nucleus.
What particle proton neutron or electron has a positive charge?
The correct option is (a.) proton. Neutrons are neutral particles of an atom. Electrons are the negatively charged subatomic particles and protons are the positively charges subatomic particles of an atom.
What is the charge of 6 protons and 5 electrons?
In an atom, protons are carriers of positive charges; carriers of negative charges are electrons; protons have no charge. In our case, an atom has 6 neutrons that do not carry a charge; 6 protons, that is, a charge of +6; and 5 electrons, that is, a charge of -5. Then the charge of the carbon ion will be: +6 -5 = +1.