What causes thymine dimers

Mia Morrison
Published Apr 09, 2026
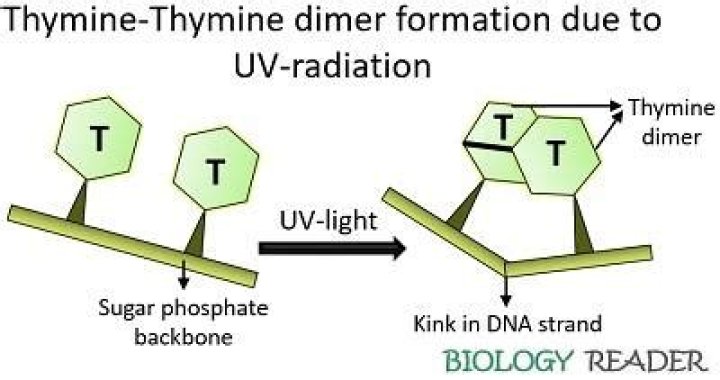
Thymine-Thymine Dimers. Thymidine Dimers are produced when adjacent thymidine residues are covalently linked by exposure to ultraviolet radiation. Covalent linkage may result in the dimer being replicated as a single base, which results in a frameshift mutation.
What is meant by thymine dimer?
A pair of abnormally chemically bonded adjacent thymine Bases in DNA, resulting from damage by ultra-violet irradiation. The cellular processes that repair this lesion often make errors that create mutations.
What's a dimer in chemistry?
noun Chemistry. a molecule composed of two identical, simpler molecules. a polymer derived from two identical monomers. Compare oligomer.
What do dimers do?
For example, dimers form in the cell membrane, where tyrosine-kinase receptors reside, and in the cytosol that contains microtubules composed of tubulin. In the nucleus, hormone receptors, acting as transcription factors, form dimers to increase stability and improve binding to DNA.Why does the formation of thymine dimers damage DNA?
Formation of thymine dimer lesion in DNA. The photon causes two consecutive bases on one strand to bind together, destroying the normal base-pairing double-strand structure in that area.
How do thymine dimers affect DNA replication?
T-T dimers cause kinks in the DNA strand that prevent both replication and transcription of that part of the DNA. Because they block DNA replication (and therefore prevent cells from reproducing), T-T dimers and other forms of UV damage cannot be inherited, and thus do not constitute mutations.
What do thymine dimers do to DNA?
The cis-syn thymine cyclobutane dimer lesion, hereafter called the thymine dimer, has traditionally been considered to be one of the more ‘bulky and destabilizing’ lesions for several reasons: it involves two nucleotides locked in a rigid, nonstandard shape; it causes anomalous migration in gels and facilitates …
Why do proteins form dimers?
(i) Dimerization occurs only when the protein concentration is higher than the dissociation constant for dimerization, forming a simple mechanism for sensing protein concentration. The dimers (mid-blue) might have higher stability and a lower surface area than the monomer (light blue).Why are dimers formed?
Carboxylic acids form dimers by hydrogen bonding of the acidic hydrogen and the carbonyl oxygen when anhydrous. For example, acetic acid forms a dimer in the gas phase, where the monomer units are held together by hydrogen bonds. Under special conditions, most OH-containing molecules form dimers, e.g. the water dimer.
What are peptides?A peptide is a short chain of amino acids. The amino acids in a peptide are connected to one another in a sequence by bonds called peptide bonds. Typically, peptides are distinguished from proteins by their shorter length, although the cut-off number of amino acids for defining a peptide and protein can be arbitrary.
Article first time published onWhy does NO2 form dimer?
Because of the presence of unpaired odd electron which is susceptible to undergo reaction. … In NO2 structure there is an unpaired electron left on Nitrogen atom, so it is reactive it dimerises to pair up its electron and gains stability as N2O4.
When a dimer is formed from two monomers what other molecule is produced?
Covalent dimers are the exact opposite. These are dimers that use covalent bonds to link monomers together. A covalent bond involves the sharing of electrons. These molecules are happy to share electrons with one another in order to link up.
What is the difference between a dimer and monomer?
is that monomer is (chemistry) a relatively small molecule which can be covalently bonded to other monomers to form a polymer while dimer is (chemistry) a molecule consisting of two identical halves, formed by joining two identical molecules, sometimes with a single atom acting as a bridge.
What can cause a thymine dimer and what are the consequences of one forming quizlet?
Ultraviolet light causes THYMINE DIMERS. Which enzyme is responsible for proofreading during replication? Bacteria can distinguish between a newly replicated DNA strand and the original template strand because the newly replicated strand is methylated, whereas the original template strand is not.
What is the main enzyme that plays a major role in formation of thymine dimer?
What is the main enzyme that plays a major role in formation of thymine dimer? Explanation: In photoreactivation DNA photolyase captures energy from light and uses it to break the covalent bond linking the adjacent pyrimidines. In this process the damaged bases are mended directly. 6.
What are the dimers of lipids?
A dimer can be modelled as a couple of lipid molecules whose headgroup lateral dipole moments have antiparallel orientation. Properties including surface pressure, chemical potentials and activity coefficients are deduced from a general expression for the free energy of the monolayer.
What does dimer mean in biology?
In biochemistry, a protein dimer is a macromolecular complex formed by two protein monomers, or single proteins, which are usually non-covalently bound. Many macromolecules, such as proteins or nucleic acids, form dimers. The word dimer has roots meaning “two parts”, di- + -mer.
What are polymers of lipids?
Lipids – polymers called diglycerides, triglycerides; monomers are glycerol and fatty acids. Proteins – polymers are known as polypeptides; monomers are amino acids.
Why are enzymes dimers?
However, dimerization provides two advantages to the thermophilic enzyme: it protects its structure against denaturation by reducing thermal fluctuations and it provides a less negative activation entropy, toning down the increase of the activation free energy with temperature.
What is the dimer of carbohydrates?
The dimers of carbohydrates are also called disaccharides as they are formed from a combination of two sugars. For instance, sucrose, which is commonly called table sugar, is the dimer of carbohydrate. It is made up of two different monomers, namely glucose and fructose.
What is a protein dimer called?
A protein dimer called tubulin, which is composed of two monomeric subunits called alpha and beta tubulin that differ from each other in only a few amino acid residues, comprises the basic structural unit of microtubules.
Where are peptide bonds formed?
A peptide bond is a chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the amino group of the other molecule, releasing a molecule of water (H2O). This is a dehydration synthesis reaction (also known as a condensation reaction), and usually occurs between amino acids.
What is difference between protein and peptide?
The basic distinguishing factors are size and structure. Peptides are smaller than proteins. Traditionally, peptides are defined as molecules that consist of between 2 and 50 amino acids, whereas proteins are made up of 50 or more amino acids.
What is the function of peptide?
Functions. Peptides are biologically and medically important molecules. They naturally occur within organisms, plus lab-synthesized compounds are active when introduced into a body. Peptides act as structural components of cells and tissues, hormones, toxins, antibiotics, and enzymes.
What is dimer in P block?
A dimerization is any chemical reaction in which two monomers react to form a dimer. For example: Dimers of carboxylic acids are often found in vapour phase. Hope this information helps you.
Why does NO2 Dimerize to form N2O4?
In the NO2 molecule the nitrogen atom has a single unpaired electron. The molecule will tend to dimerise so that this unpaired electron can be paired with another to form the N2O4 molecule. NO2 molecule is an odd electron molecule and hence paramagnetic. Due to possession of odd electron, it is coloured.
Why does NO2 Dimerise to form N2O4?
NO2 is an odd electron molecule i.e., it has one unpaired electron. Such molecules are highly reactive and tend to form a dimer by pairing of the unpaired electrons. Thus get stablity, Therefore NO2, dimerises to form N2O4 which contains a N-N single bond.
What type of reaction forms a dimer quizlet?
When water condenses out of the reaction, a dimer is created as a covalent bond forms between two monomers. In dehydration synthesis, also called a condensation reaction, a hydroxyl group (-OH) and a hydrogen atom (H) combine to form H2O, which then leaves the reaction as a product.
What is a dimer polymer?
Dimerization is a type of polymerization in which a dimer is formed from the combination of two monomer units. … We can identify a dimer as an oligomer that has a low number of repeating units and the bonds between these two monomer units can be either strong or weak, covalent or intermolecular bonds.
Is a dimer smaller than a monomer?
A dimers had less compact free energy landscapes than A monomers in both water models.
Does BCl3 form dimer?
AlCl3 forms a dimer but BCl3 does not form dimer.