What causes hydrogen bond

Sophia Edwards
Published Feb 24, 2026
The reason hydrogen bonding occurs is because the electron is not shared evenly between a hydrogen atom and a negatively charged atom. Hydrogen in a bond still only has one electron, while it takes two electrons for a stable electron pair. … Any compound with polar covalent bonds has the potential to form hydrogen bonds.
What is a hydrogen bond easy definition?
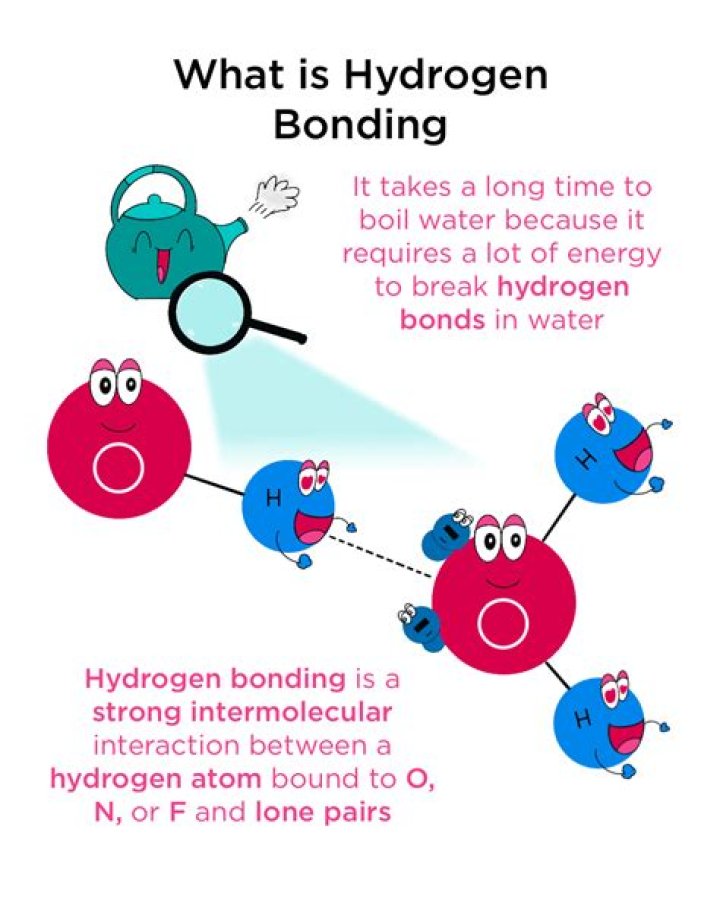
hydrogen bonding, interaction involving a hydrogen atom located between a pair of other atoms having a high affinity for electrons; such a bond is weaker than an ionic bond or covalent bond but stronger than van der Waals forces.
What is hydrogen bonding with example?
water (H2O): Water is an excellent example of hydrogen bonding. The bond is between the hydrogen of one water molecule and the oxygen atoms of another water molecule, not between the two hydrogen atoms (a common misconception). … ammonia (NH3): Hydrogen bonds form between hydrogen of one molecule and nitrogen of another.
What causes hydrogen bonding attraction between ions?
Hydrogen bonds are strong intermolecular forces created when a hydrogen atom bonded to an electronegative atom approaches a nearby electronegative atom. Greater electronegativity of the hydrogen bond acceptor will lead to an increase in hydrogen-bond strength.What type of bond leads to hydrogen bonds?
Hydrogen bonding occurs only in molecules where hydrogen is covalently bonded to one of three elements: fluorine, oxygen, or nitrogen. These three elements are so electronegative that they withdraw the majority of the electron density in the covalent bond with hydrogen, leaving the H atom very electron-deficient.
What elements can hydrogen bond with?
Hydrogen bonding occurs only in molecules where hydrogen is covalently bonded to one of three elements: fluorine, oxygen, or nitrogen. These three elements are so electronegative that they withdraw the majority of the electron density in the covalent bond with hydrogen, leaving the H atom very electron-deficient.
What are the factors responsible for the strength of hydrogen bonds Class 11?
Strength of the hydrogen bond is determined by the coulombic interaction between the lone-pair electrons of the electronegative atom of one molecule and the hydrogen atom of other molecule.
Which best describes how hydrogen bonds are created?
Hydrogen bonding occurs when a molecule contains a hydrogen atom bonded to fluorine, oxygen, or nitrogen. This hydrogen becomes partially positive in charge, while the attached atom becomes partially negative.What is hydrogen bonding explain intermolecular and intermolecular hydrogen bonding?
A hydrogen bond is an intermolecular force (IMF) that forms a special type of dipole-dipole attraction when a hydrogen atom bonded to a strongly electronegative atom exists in the vicinity of another electronegative atom with a lone pair of electrons. Intermolecular forces (IMFs) occur between molecules.
What is meant by hydrogen bond Class 11?Hydrogen bond is formed when hydrogen is attached to some electronegative element like O, N and F. It forma a special bond with them called as hydrogen bond. Types of Hydrogen bond. Intermolecular H bond. Intra-molecular H bond.
Article first time published onWhat are hydrogen bonds quizlet?
What is a hydrogen bond? A type of weak chemical bond formed when the slightly positive hydrogen atom of a polar covalent bond in one molecule is attracted to the slightly negative atom of a polar covalent bond in another molecule (or in another region of the same molecule).
Why are hydrogen bonds the strongest of the intermolecular forces?
Because it involves highly electronegative (tendency of an atom to attract electrons) e.g. oxygen and chlorine. And hydrogen has only one electron, therefore is less negative (almost positive in a sense). This causes very strong attraction between weak and strong atoms.
Where is it most likely that a hydrogen bond would form?
Hydrogen bonds occur most often between neutral molecules. The interacting atoms in these molecules carry fractional charges, and this type of hydrogen bond can be considered a type of dipole-dipole interaction.
What 3 things can hydrogen bonding cause?
- The structure of DNA.
- The specific heat capacity/ boiling point of water.
- The structure of proteins.
What is hydrogen bonding in water?
The hydrogen bond in water is a dynamic attraction between neighboring water molecules involving one hydrogen atom located between the two oxygen atoms.
How does a hydrogen bond form quizlet?
A hydrogen bond is formed between adjacent water molecules when a negative end of one water molecule is attracted to the positive end of another water molecule.
What is a hydrogen bond explain how a hydrogen bond forms quizlet?
A weak chemical bond formed when the slightly positive hydrogen atom of a polar covalent bond in one molecule is attracted to the slightly negative atom of a polar covalent bond in another molecule. …
Why do hydrogen bonds occur quizlet?
Hydrogen bonding occurs between molecules in which hydrogen atoms are bound to small, highly electronegative atoms.
Why are hydrogen bonds important?
Hydrogen bonds provide many of the critical, life-sustaining properties of water and also stabilize the structures of proteins and DNA, the building block of cells.
How is hydrogen bond responsible for high surface tension?
When many water molecules form hydrogen bonds with other water molecules, they form a lattice of water molecules, which is strong and flexible. This creates a high surface tension. … As a result, this molecule at the surface tends to be pulled into the bulk of the liquid and thus minimizes the surface area of a liquid.
Which molecule would have the strongest tendency to form hydrogen bonds?
A more basic molecule will make the hydrogen bond stronger. Nitrogen forms the strongest hydrogen bonds, whereas fluorine forms the weakest hydrogen bonds.
What is hydrogen bonding explain its types?
Hydrogen bond is a electrostatic attraction between a hydrogen atom which is bond to a more electronegative atom such as Nitrogen, Oxygen, fluorine. These are two types of hydrogen bonds :- 1) Intermolecular Hydrogen bonding :- It occurs between two separate molecules.
Which element is least likely to participate in a hydrogen bond?
The least likely to participate in a hydrogen bond is Carbon. The Hydrogen bond is an electrostatic force of attraction between a hydrogen atom with electronegative atom or group.