What are turns in proteins

Sophia Edwards
Published Mar 13, 2026
Turns are the third of the three “classical” secondary structures with approximately one-third of all residues in globular proteins are contained in turns that serve to reverse the direction of the polypeptide chain. … Turns are located primarily on the protein surface and accordingly contain polar and charged residues.
What are turns and loops?
Loops and turns connect α helices and β strands. The most common types cause a change in direction of the polypeptide chain allowing it to fold back on itself to create a more compact structure. … Loops that have only 4 or 5 amino acid residues are called turns when they have internal hydrogen bonds.
What is the difference between turn and loop in protein?
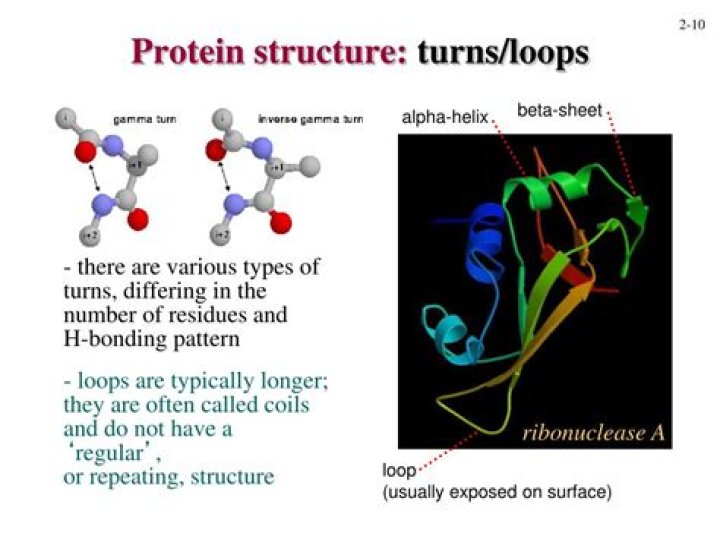
They both are degenerate. They typically connect a {helix, strand} to another {helix, strand}. The main difference is the length; turns are short and loops are longer.
What are turns in polypeptide?
Turns are a fundamental class of polypeptide structure and are defined as sites where the peptide chain reverses its overall direction. In the past 20 years, the peptide field has witnessed major development, stimulated by the discovery of a host of bioactive peptides.What are beta turns in proteins?
From Wikipedia, the free encyclopedia. β turns (also β-bends, tight turns, reverse turns, Venkatachalam turns) are the most common form of turns—a type of non-regular secondary structure in proteins that cause a change in direction of the polypeptide chain. They are very common motifs in proteins and polypeptides.
What are turns in relation to secondary structures of proteins?
Turns are the third of the three “classical” secondary structures with approximately one-third of all residues in globular proteins are contained in turns that serve to reverse the direction of the polypeptide chain. … Turns are located primarily on the protein surface and accordingly contain polar and charged residues.
What are turns in amino acids?
A turn is an element of secondary structure in proteins where the polypeptide chain reverses its overall direction.
Do beta turns contain proline?
Proline-containing beta-turns in peptides and proteins: analysis of structural data on globular proteins. Arch Biochem Biophys.How many amino acids are in a turn?
Alpha-helices have 3.6 amino acid residues per turn, ie a helix 36 amino acids long would form 10 turns.
What is an alpha turn?The alpha-turn corresponds to a chain reversal involving five amino acids and may be stabilized by a hydrogen bond between the CO group of the first residue and the NH group of the fifth (Pavone et al. … 1997).
Article first time published onWhat are reverse turns?
A reverse turn is region of the polypeptide having a hydrogen bond from one main chain carbonyl oxygen to the main chain N-H group 3 residues along the chain (ie O(i) to N(i+3)). … Reverse turns are very abundant in globular proteins and generally occur at the surface of the molecule.
Which amino acids are in reverse turns?
Three subclasses of reverse turns (Types I – III) have been recognized, All involve a four amino acid sequence in whch the carbonyl oxygen of AA-1 is H-bonded to the amino-H of AA-4 (rather than AA-5 as is found in the alpha helix). Type I and type II turns differ in the bond linking residue 2 and residue 3.
How do type I and type II beta turns differ?
Type I and II β-turns are different mainly in the orientation of the amide bond between the residues i + 1 and i + 2 on the plane of the β-turn. Type I′ and II′ β-turns are mirror images of the backbone conformations of type I and type II, respectively.
Why are glycine and proline found in turns?
Glycine and proline residues are frequently found in turn and loop structures of proteins and are believed to play an important role during chain compaction early in folding. … The altered dynamics around glycine and trans prolyl bonds can be mainly ascribed to their effects on the activation energy.
What is Loop in protein structure?
Protein loops are patternless regions which connect two regular secondary structures. They are generally located on the protein’s surface in solvent exposed areas and often play important roles, such as interacting with other biological objects. Despite the lack of patterns, loops are not completely random structures.
What amino acids turn in beta?
Gratifyingly, the turn propensities of amino acids at different positions of various protein β-turn types obtained through statistical analysis by directed evolution and phage-display correlate well with work on model peptides in showing glycine, proline, asparagine, and aspartic acid to be the most common β-turn- …
Why do turns and loops most often occur on the proteins surface?
A turn helps to stabilize abrupt directional changes in the polypeptide chain. Loops are more elaborate chain reversal structures that are rigid and well defined. Loops and turns generally lie on the surfaces of proteins so they often participate in interactions between proteins and other molecules.
Where might bends or beta turns occur?
(a) Bends or turns are most likely to occur at residues 7 and 19 because Pro residues are often (but not always) found at bends in globular folded proteins. A bend may also occur at the Thr residue (residue 4) and, assuming that this is a portion of a larger polypep- tide, at the Ile residue (residue 1).
What causes beta turns?
The Beta Turn Turns generally occur when the protein chain needs to change direction in order to connect two other elements of secondary structure. The most common is the beta turn, in which the change of direction is executed in the space of four residues.
What causes changes in protein structure?
Proteins change their shape when exposed to different pH or temperatures. The body strictly regulates pH and temperature to prevent proteins such as enzymes from denaturing. Some proteins can refold after denaturation while others cannot. Chaperone proteins help some proteins fold into the correct shape.
What causes the changes in protein structure through the three or four levels of structure?
What causes the changes in protein structure through the three or four levels of structure? … The primary structure is based on the bonds between individual amino acids while the secondary structure is based on the formation of alpha and beta pleated sheets.
What happens when protein denatures?
Denaturation involves the breaking of many of the weak linkages, or bonds (e.g., hydrogen bonds), within a protein molecule that are responsible for the highly ordered structure of the protein in its natural (native) state. Denatured proteins have a looser, more random structure; most are insoluble.
What are residues in proteins?
In biochemistry or molecular biology, a residue refers to a single unit that makes up a polymer, such as an amino acid in a polypeptide or protein. Example of usage: a polypeptide consisting of 5 amino acid residues.
How many amino acids are in one turn of an alpha helix?
An α-helix secondary structure is stabilized by hydrogen bonds between carbonyl oxygen and the amino group of every third residue in the helical turn with each helical turn consisting of 3.6 amino acid residues (Fig. 10.1A).
What are proline kinks?
Prolines in alpha helices after the first turn (4th residue) cause a kink in the helix. This kink is caused by proline being unable to complete the H-bonding chain of the helix and steric or rotamer effects that keep proline from adapting the prefered helical geometry.
Why is proline found in turns?
Proline is unique in that it is the only amino acid where the side chain is connected to the protein backbone twice, forming a five-membered nitrogen-containing ring. … For this reason, Proline can often be found in very tight turns in protein structures (i.e. where the polypeptide chain must change direction).
Is glycine found in tight turns?
What this means is that glycine can reside in parts of protein structures that are forbidden to all other amino acids (e.g. tight turns in structures).
What is bend biochemistry?
Explanation: Beta bends are part of secondary protein structures. They serve as a link between alpha helices and beta sheets. Beta bends are composed of proline and glycine, amino acids that usually are not found in alpha helices.
Which is globular protein?
Globular proteins are named for their approximately spherical shapes and are the most abundant proteins in nature. The globular proteins exist in an enormous variety of three-dimensional structures. … Hydrophilic amino acid side chains lie on the surface of the globular proteins exposed to the water.
What type of amino acids form hydrogen bonds?
The chemistry of amino acid side chains is critical to protein structure because these side chains can bond with one another to hold a length of protein in a certain shape or conformation. Charged amino acid side chains can form ionic bonds, and polar amino acids are capable of forming hydrogen bonds.
Why alpha helix is right handed?
The α-helix is a common element of protein secondary structure, formed when amino acids “wind up” to form a right-handed helix where the side-chains point out from the central coil (Fig. 3.1A,B).



