What are radioactive atoms

Robert Spencer
Published Apr 07, 2026
Radioactive atoms are atoms, which contain unstable nucleus, and which may undergo radioactive decay. The term radioactive atom is misleading term, since usually only nuclei may undergo decay and changes in electron configuration are only a result of changes in nucleus configuration.
Can radiation change atoms?
Ionizing radiation can affect the atoms in living things, so it poses a health risk by damaging tissue and DNA in genes. Ionizing radiation comes from x-ray machines, cosmic particles from outer space and radioactive elements. Radioactive elements emit ionizing radiation as their atoms undergo radioactive decay.
What is the most common cause of radioactivity in atoms?
What causes atoms to be radioactive? Atoms found in nature are either stable or unstable. An atom is stable if the forces among the particles that makeup the nucleus are balanced. An atom is unstable (radioactive) if these forces are unbalanced; if the nucleus has an excess of internal energy.
Why do atoms go through radioactive decay?
Every atom seeks to be as stable as possible. In the case of radioactive decay, instability occurs when there is an imbalance in the number of protons and neutrons in the atomic nucleus. … If the nucleus of an atom is unstable, eventually it will break apart to lose at least some of the particles that make it unstable.What happens during radioactivity?
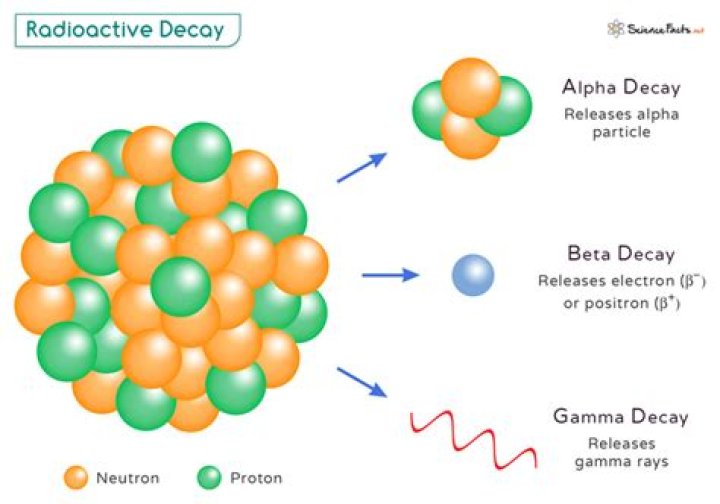
When radioactive atoms decay, they release energy in the form of ionizing radiation (alpha particles, beta particles and/or gamma rays). The energy is called ionizing radiation because it has enough energy to knock tightly bound electrons from an atom’s orbit. This causes the atom to become a charged ion.
How do atoms decay?
When a radioactive atom decays, it loses energy by emitting particles or radiation. This process changes the composition of the nucleus, often transforming it into a new element. … When it undergoes beta decay, one of its neutrons splits into a proton, an antineutrino and an electron.
What is the difference between radiation and radioactive atoms?
Radiation in the form of radiant particles or rays, is the result of a nuclear disintegration. Radioactive materials are atoms that have stored energy and may disintegrate in the future, releasing radiation.
How does radiation make other things radioactive?
Induced radioactivity, also called artificial radioactivity or man-made radioactivity, is the process of using radiation to make a previously stable material radioactive. … Neutron activation is the main form of induced radioactivity. It occurs when an atomic nucleus captures one or more free neutrons.How are electromagnetic radiation and the atom connected?
Electromagnetic radiation is made when an atom absorbs energy. The absorbed energy causes one or more electrons to change their locale within the atom. When the electron returns to its original position, an electromagnetic wave is produced. … These electrons in these atoms are then in a high energy state.
How do radioactive atoms know to decay?(An atom’s electrons and nucleons can be excited, but these excited particles quickly relax back down to the ground state. … The answer is that radioactive atoms don’t know when to decay. In fact, an individual radioactive atom does not decay at a particular, predictable time.
Article first time published onWhat happens during the radioactive decay of an element?
Elements that emit ionizing radiation are called radionuclides. When it decays, a radionuclide transforms into a different atom – a decay product. The atoms keep transforming to new decay products until they reach a stable state and are no longer radioactive.
What happens when a radioactive carbon atom decays?
C decays by a process called beta decay. During this process, an atom of 14C decays into an atom of 14N, during which one of the neutrons in the carbon atom becomes a proton. This increases the number of protons in the atom by one, creating a nitrogen atom rather than a carbon atom.
Where does radioactivity occur in an atom?
Atoms are radioactive if the protons and neutrons in the nucleus are configured in an unstable way. For low numbers of protons (Z), the number of neutrons (N) required to maintain a stable balance is roughly equal to the number of protons.
How are radioactive elements formed?
Radioactive elements are made up of atoms whose nuclei are unstable and give off atomic radiation as part of a process of attaining stability. The emission of radiation transforms radioactive atoms into another chemical element, which may be stable or may be radioactive such that it undergoes further decay.
How does metal become radioactive?
Similarly, many other materials, especially metals, are subject to neutron activation. A piece of steel containing small amounts of stable cobalt when irradiated with low energy neutrons will produce radioactive products from both the iron and the cobalt in the metal (as well as other possible species).
Why do atoms want to be stable?
Originally Answered: Why is an atom stable? All atoms are eager to become octet-in which the total number of the electrons in the outermost shell is equal to eight (since 8 is the total no. of electrons the last shell can carry) and it cannot accept electrons anymore so it becomes stable.
What are uses of radioactivity?
Today, to benefit humankind, radiation is used in medicine, academics, and industry, as well as for generating electricity. In addition, radiation has useful applications in such areas as agriculture, archaeology (carbon dating), space exploration, law enforcement, geology (including mining), and many others.
What is radioactive in simple words?
As its name implies, radioactivity is the act of emitting radiation spontaneously. This is done by an atomic nucleus that, for some reason, is unstable; it “wants” to give up some energy in order to shift to a more stable configuration.
What is the difference between radioactivity and radioactive decay explain?
Radioactivity is the term given to the breaking-up (decay) or rearrangement of an atom’s nucleus. Decay occurs naturally and spontaneously to unstable nuclei. This instability is usually caused by a mismatch between the number of protons and neutrons.
Is radioactivity and radioactive decay the same thing?
Radioactive decay (also known as nuclear decay, radioactivity, radioactive disintegration or nuclear disintegration) is the process by which an unstable atomic nucleus loses energy by radiation. A material containing unstable nuclei is considered radioactive.
Why does radioactive decay play an important role in Earth's internal heat?
Why does radioactive decay play a very important role in earth’s internal heat? Radioactive element can be found anywhere in the planet. When radioactive element decays, it produces heat. Spontaneous nuclear disintegration of radioactive elements produced thermal energy.
What happens when an atom dies?
When we die, our atoms will disassemble and move off to finds new uses elsewhere – as part of a leaf or other human being or a drop of dew. Atoms themselves, however go on practically forever. Bill Bryson’s, A Short History of Nearly Everything, 2003 Won the Aventis prize for Science books in 2004.
How do atoms form things?
Atoms form chemical bonds with other atoms when there’s an electrostatic attraction between them. This attraction results from the properties and characteristics of the atoms’ outermost electrons, which are known as valence electrons.
What happens when atoms absorb electromagnetic radiation?
When atoms absorb energy, through heating, from electricity, or by absorbing electromagnetic radiation , the electrons at a particular level can be pushed up to higher levels (at bigger distances from the nucleus). In time, they jump back down to a lower level radiating energy in the form of electromagnetic waves .
What emits electromagnetic radiation?
Electromagnetic radiation is produced whenever a charged particle, such as an electron, changes its velocity—i.e., whenever it is accelerated or decelerated. The energy of the electromagnetic radiation thus produced comes from the charged particle and is therefore lost by it.
What happens when an atom emits electromagnetic radiation?
An electron can be excited when its absorbs energy from electromagnetic radiation. … An excited electron can fall to a lower energy level. When this happens, energy is lost as electromagnetic radiation. Different changes in energy level cause the emission of different frequencies.
Can a human be radioactive?
Yes, our bodies are naturally radioactive, because we eat, drink, and breathe radioactive substances that are naturally present in the environment.
What is radiation How does it affect an object?
The main effect radiation has on matter is its ability to ionize atoms to become ions, a phenomenon known as ionization, which is very similar to the photoelectric effect. Radioactive particles or electromagnetic waves with sufficient energy collide with electrons on the atom to knock electrons off the atom.
Can humans become radioactive?
There are types of radiation where human bodies could retain radioactive particles and remain radioactive over time, but this is not the type that was seen at Chernobyl. After gamma radiation has passed through the body, the person is no longer radioactive and can’t expose other people.
Can an atom be destroyed?
No atoms are destroyed or created. The bottom line is: Matter cycles through the universe in many different forms. In any physical or chemical change, matter doesn’t appear or disappear. Atoms created in the stars (a very, very long time ago) make up every living and nonliving thing on Earth—even you.
What elements are radioactive in their most common form?
ElementMost Stable IsotopeHalf-life of Most Stable IsotopeTechnetiumTc-914.21 x 106 yearsPromethiumPm-14517.4 yearsPoloniumPo-209102 years