What are element families

William Taylor
Published Mar 01, 2026
Element families are elements that have the same number of valence electrons. Most element families are a single column of the periodic table, although the transition elements consist of several columns, plus the elements located below the main body of the table.
What are groups and families on the periodic table?
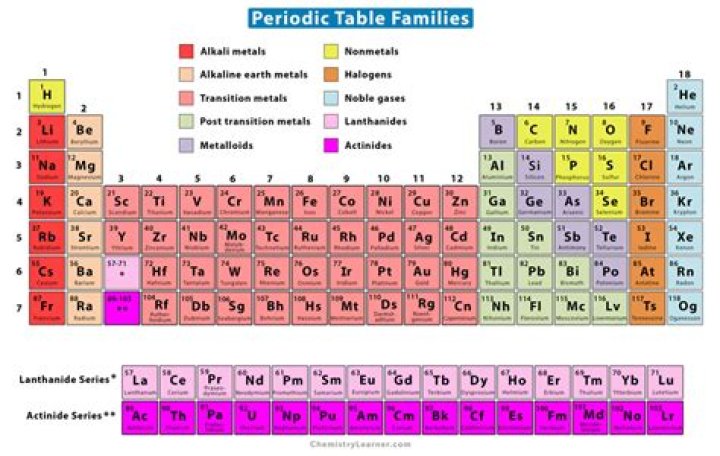
In the periodic table of elements, there are seven horizontal rows of elements. Each of these rows are called periods. The vertical columns of elements are called groups, or families.
What are the family names on the periodic table?
- Alkali metals.
- Alkaline earth metals.
- Transition metals.
- Halogens.
- Noble gases.
What does family mean in science?
noun, plural: families. (1) A taxonomic rank in the classification of organisms between genus and order. (2) A taxonomic group of one or more genera, especially sharing a common attribute.How many a families are there in the periodic table?
So why do the elements in the same family have similar properties? You can examine four families on the periodic table and look at the electron configurations for a few elements in each family. The figure below lists some important families that are given special names: The IA family is made up of the alkali metals.
Which is not a family of the periodic table?
Hydrogen is a very special element of the periodic table and doesn’t belong to any family. While hydrogen sits in Group I, it is NOT an alkali metal.
How many families of elements are there in the periodic table?
5 Element Families The five element families combine similar element groups. So, even though there are numerous columns of transition metals on the periodic table, they all belong to the same family. The transition metals also include the lanthanides and actinides, which appear the main body of the table.
What does group A and B mean on the periodic table?
Elements are arranged by reactivity in the periodic table. Elements with similar reactivity are put into the same column or group. The columns with B (IB through VIIIB) are called the transition elements. … The columns with A (IA through VIIIA) are called the main group elements.What are 3 elements in the same family?
Lithium, Hydrogen, Sodium, or Oxygen, Sulfur, Selenium They are all in the same column.
What is family Short answer?Family is defined as a group of people consisting children, parents, aunts, uncles, cousins and grandparents.
Article first time published onWhat family really means?
“Family means having someone to love you unconditionally in spite of you and your shortcomings. Family is loving and supporting one another even when it’s not easy to do so. It’s being the best person you could be so that you may inspire your love ones. … Family isn’t just important, it’s what is most important!”
What does family mean in species?
In biology, a family is a taxonomic rank, or a taxon at that rank. Each family contains one or more genera. The next important rank is that of order. Usually, the name of the family ends with a “idae” for animals, and “aceae” for plants.
What is the family name of group 2?
Alkaline earth metals – The metals of group 2: Be, Mg, Ca, Sr, Ba, Ra.
What is the second family in the periodic table?
Group 2A — The Alkaline Earth Metals. Group 2A (or IIA) of the periodic table are the alkaline earth metals: beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra). They are harder and less reactive than the alkali metals of Group 1A.
What family does neon belong to?
Group 8A (or VIIIA) of the periodic table are the noble gases or inert gases: helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). The name comes from the fact that these elements are virtually unreactive towards other elements or compounds.
How many family groups are there?
There are six specific types of family structures identified by society today.
How many families are in the periodic table list them and group number?
Each of the elements in the Periodic Table can be divided into one of nine groups. These groups share many similar properties; both chemical and physical.
What family has 7 valence electrons?
Any element in the halogen group will have seven valence electrons. These elements include fluorine, chlorine, bromine, iodine, and astatine.
What family has 6 valence electrons?
Periodic table groupValence ElectronsGroup 15 (V) (pnictogens)5Group 16 (VI) (chalcogens)6Group 17 (VII) (halogens)7Group 18 (VIII or 0) (noble gases)8**
Which family on the periodic table contains elements that won't react?
All of the elements in Group Zero are noble gases. The list includes helium, neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). Don’t think that, because these elements don’t like to react, we don’t use them. You will find noble gases all over our world.
What family of elements is in the right most column of the periodic table?
Explanation: The halogens, fluorine, chlorine, bromine, iodine, are all non-metals. Astatine is so rare that its chemistry would be limited. The Noble Gases all have a complete valence shell, and are all gases under normal conditions………
What is a family on the periodic table quizlet?
Family/Group. The vertical columns on the periodic table. Elements in the same group/family have the same number of valence electrons.
What are the names and characteristics of each family on the periodic table?
- Group 1: alkali metals, or lithium family.
- Group 2: alkaline earth metals, or beryllium family.
- Group 3: the scandium family.
- Group 4: the titanium family.
- Group 5: the vanadium family.
- Group 6: the chromium family.
- Group 7: the manganese family.
- Group 8: the iron family.
What are the 10 families in the periodic table?
Related elements, including the noble gases, halogens, alkali metals, alkaline earth metals, transition metals, lanthanides, and actinides. In addition, metals, nonmetals, and metalloids form loosely defined families. Other family designations—such as carbon family—are sometimes used.
What element is in period 4 Group 12?
zinc group element, any of the four chemical elements that constitute Group 12 (IIb) of the periodic table—namely, zinc (Zn), cadmium (Cd), mercury (Hg), and copernicium (Cn).
What is Period 3 on the periodic table?
A period 3 element is one of the chemical elements in the third row of the periodic table of the chemical elements. … The third period contains eight elements: sodium, magnesium, aluminium, silicon, phosphorus, sulfur, chlorine, and argon.
What is the only letter not appearing on the periodic table?
The only letter that doesn’t appear in the periodic table is … J! Did you know!? The letter “Q” does not appear in any official element names, but it does in temporary element names, such as ununquadium.
What is n on the periodic table?
Nitrogen – Element information, properties and uses | Periodic Table.
What is the name of Group 1 on the periodic table?
The alkali metals are six chemical elements in Group 1, the leftmost column in the periodic table. They are lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr).
What is C on the periodic table?
Carbon – Element information, properties and uses | Periodic Table.
What is a family answer Class 1?
A family is a social group of people who stay together under the same roof in a society. It comprises two or more adults like parents and grandparents and young children who are bound in a relationship by birth or blood. They are collectively known as family members.