What are cations formed by

William Taylor
Published Mar 12, 2026
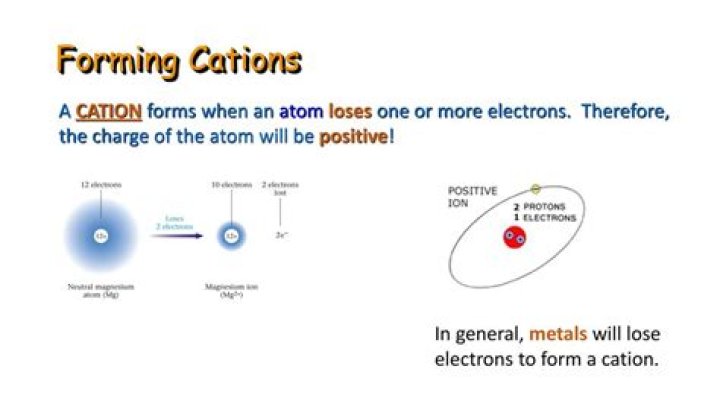
A cation is an ion with positive charge, which means it has more protons (positively-charged particles) than electrons (negatively-charged particles). Cations are formed when an atom loses one or more electrons: the loss of the negatively-charged electron(s) results in an overall positive charge.
How are ions and cations formed?
Ions. Ions are charged substances that have formed through the gain or loss of electrons. Cations form from the loss of electrons and have a positive charge while anions form through the gain of electrons and have a negative charge.
What two elements form cations?
Halogens always form anions, alkali metals and alkaline earth metals always form cations. Most other metals form cations (e.g. iron, silver, nickel), whilst most other nonmetals typically form anions (e.g. oxygen, carbon, sulfur).
Are cations only formed by metals?
Metals are not cations, metal ions are. Any substance in elemental form is an atom or a molecule. They become ions depending upon the the electronic configuration. Positive ions(cations) is they lose electrons or negative ions(anions).How is Na+ formed?
Neutral sodium atom (Na) becomes sodium cation (Na+) by releasing an electron. chlorine atom (Cl) in the chlorine molecule (Cl2) becomes chloride (Cl-) by accepting an electron. positive charge on the sodium cation is balanced by the negative charge on the chloride, so the ionic compound is neutral.
Do metalloids form cations?
Metalloids, being chemically weak nonmetals, predominately form oxyanions e.g. the borate oxyanion, BO 3 ^3-. Rarely do they form cations, and never (as far as I know) simple cations.
How are ionic compounds formed?
An ionic bond is formed by the complete transfer of some electrons from one atom to another. The atom losing one or more electrons becomes a cation—a positively charged ion. The atom gaining one or more electron becomes an anion—a negatively charged ion.
Are all transition metals cations?
Consequently, all transition-metal cations possess dn valence electron configurations, as shown in Table 23.2 for the 2+ ions of the first-row transition metals. All transition-metal cations have dn electron configurations; the ns electrons are always lost before the (n − 1)d electrons.Are cations metals or nonmetals?
First, each element that forms cations is a metal, except for one (hydrogen), while each element that forms anions is a nonmetal. This is actually one of the chemical properties of metals and nonmetals: metals tend to form cations, while nonmetals tend to form anions.
What is a cation chemistry?cation, atom or group of atoms that bears a positive electric charge.
Article first time published onWhat is a cation quizlet?
Cation. An ion or group of ions having a positive charge and characteristically moving toward the negative electrode in electrolysis.
Which elements can form an ionic bond Why?
Ionic bonds usually occur between metal and nonmetal ions. For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons. Covalent bonds usually occur between nonmetals.
How is the molecule of a substance formed?
When atoms combine by forming covalent bonds, the resulting collection of atoms is called a molecule. We can therefore say that a molecule is the simplest unit of a covalent compound.
How is chloride ion formed?
The chloride ion /ˈklɔːraɪd/ is the anion (negatively charged ion) Cl−. It is formed when the element chlorine (a halogen) gains an electron or when a compound such as hydrogen chloride is dissolved in water or other polar solvents. Chloride salts such as sodium chloride are often very soluble in water.
How are ionic compounds formed quizlet?
ionic bonds form when electrons are transferred from one atom to another atom. ions of different elements can combine by forming ionic bonds . positive ions & negative ions form when atom s lose or gain electrons.
Are ionic compounds formed by sharing electrons?
Ionic bonds form when a nonmetal and a metal exchange electrons, while covalent bonds form when electrons are shared between two nonmetals. An ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions.
Do inner transition metals form cations?
As you also have heard them as Transition metals , so the word metal also suggests that they have the tendency of loosing electrons thus gaining a positive (+ve) charge and becoming a cation. … So in the conclusion we can say they form cations and some of the transition elements have variable charges.
What do metalloids and nonmetals form?
Simply, metals lose electrons and can form only ionic bonds. Metalloids and non-metals not only form covalent bonds by sharing, but can form ionic bonds either by losing or gaining electrons. … The compounds formed by transition metals have significant covalent nature.
Do metalloids produce cations or anions?
Their chemical behavior falls between that of metals and nonmetals. For example, the pure metalloids form covalent crystals like the nonmetals, but like the metals, they generally do not form monatomic anions.
Why do metals become cations?
Metals generally lose electrons to achieve a noble gas configuration when they have one, two, or three valence electrons and thus become cations. Otherwise more energy would be required for them to gain the necessary electrons.
What element forms +4 ions?
(3)(4)(5)(6)
When ionic compounds form Why do metals tend to form cations while nonmetals form anions?
Metals tend to lose electrons and form positively charged ions called cations. Non-metals tend to gain electrons and form negatively charged ions called anions. For example, sodium metal, Na, [Ne]3s1, loses one electron to form the Na+ ion which is isoelectronic with neon.
How do cations formed by transition metals differ from those formed by metals?
Most transition metals differ from the metals of Groups 1, 2, and 13 in that they are capable of forming more than one cation with different ionic charges. It can sometimes lose two electrons to form the Fe2+ ion, while at other times it loses three electrons to form the Fe3+ ion. …
How do transition metals form cations?
We know that metals tend to lose electrons, and non-metals tend to gain electrons. Transition elements can also be called transition metals, since all of them are metals. That must mean that they form cations by losing electrons.
What are metal cations?
A metal ion is a type of atom compound that has an electric charge. Such atoms willingly lose electrons in order to build positive ions called cations. Ions are essentially enclosed by delocalized electrons which are responsible for processes like conductivity.
Why cation is called cation?
In science, a cation is an ion, or charged particle, with a positive charge. In other words, a cation has more protons than electrons. … When an ion’s charge is negative, it’s more specifically called an anion, and when it’s positive, you can use the term cation. The Greek source is kation, which means “going down.”
Which ion is cation quizlet?
Positively charged ions are called cations. Negatively charged ions are called anions. Sodium (Na +), calcium (Ca +), and potassium (K +) are cations.
What is cation example?
They are formed when a metal loses its electrons. They lose one or more than one electron and do not lose any protons. Therefore, they possess a net positive charge. Some examples of cations are Calcium (Ca2+), Potassium (K+), hydrogen (H+).
Which one of these is a cation?
Ammonium ion (NH4+) is a cation. It is a positively charged polyatomic cation.
Which two elements react together to form an ionic compound?
Whereas ionic compounds are usually formed when a metal and a nonmetal combine, covalent compounds are usually formed by a combination of nonmetals. Thus, the periodic table can help us recognize many of the compounds that are covalent.
What elements form ions?
FamilyElementIon NamePotassiumPotassium cationIIABerylliumBeryllium cationMagnesiumMagnesium cationCalciumCalcium cation