What are 3 uses of isotopes

Rachel Hickman
Published May 04, 2026
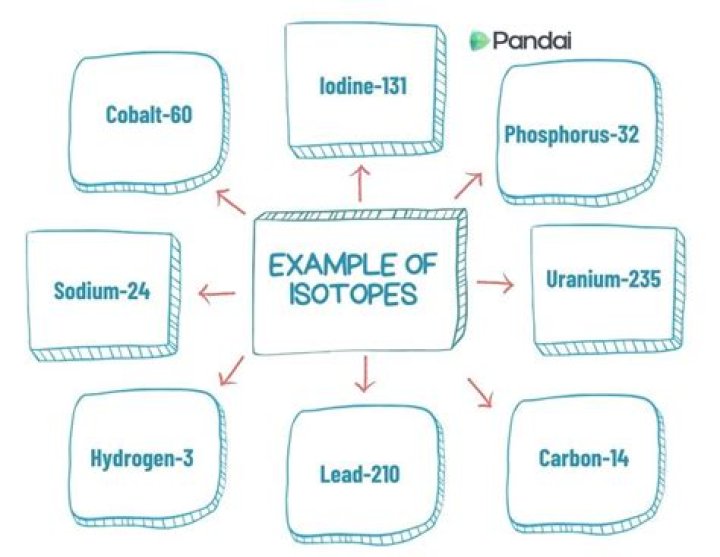
Uranium-235 is used for nuclear fission and as a fuel in nuclear reactors.An isotope of cobalt is used in the treatment of cancer.An isotope of iodine is used in the treatment of goiter.
What radioactive elements are used in medicine?
Four typical examples of radioactive tracers used in medicine are technetium-99 (9943Tc), thallium-201 (20181Tl), iodine-131 (13153I), and sodium-24 (2411Na). Damaged tissues in the heart, liver, and lungs absorb certain compounds of technetium-99 preferentially.
Which isotope is used in the treatment of Anaemia?
Iron-deficiency anemias due to malnutrition, malabsorption, blood loss, or special need are explored by dual study (oral vs intravenous) with radioactive (Fe-59, Fe-55) or stable (Fe-54, Fe-57) iron isotopes, which can guide dietary supplementation.
Are isotopes used in medical imaging?
Common isotopes that are used in nuclear imaging include: fluorine-18, gallium-67, krypton-81m, rubidium-82, nitrogen-13, technetium-99m, indium-111, iodine-123, xenon-133, and thallium-201.How do isotopes help doctors and medical technicians do their jobs?
Isotopes provide tools to do certain jobs better, easier, quicker, more simply, or more cheaply than any other method. … They are ideal tools for making measurements: a single atom can be detected using radioactive isotopes, whereas chemical methods often require a million or more atoms for detection.
What isotopes are used for diagnosing and treating illness and diseases?
The most common radioisotopes used in the medical industry are Technetium-99m, Iodine-131, and Molybdenum-99. 85% of all nuclear medical examinations use Mo/Tc generators for diagnosing problems with the liver, bones, or lungs [6].
How radioactive isotopes are used for medical diagnosis and treatment?
Radioisotopes are widely used to diagnose disease and as effective treatment tools. For diagnosis, the isotope is administered and then located in the body using a scanner of some sort. The decay product (often gamma emission) can be located and the intensity measured.
What are 5 uses of isotopes?
IsotopeUse99mTc*brain, thyroid, liver, bone marrow, lung, heart, and intestinal scanning; blood volume determination131Idiagnosis and treatment of thyroid function133Xelung imaging198Auliver disease diagnosisHow are isotopes used in biology?
Isotopes are variations of chemical elements containing different numbers of neutrons. Because isotopes are recognizable, they provide an efficient way to track biological processes during experimentation.
Is lithium an isotope?Naturally occurring lithium (3Li) is composed of two stable isotopes, lithium-6 and lithium-7, with the latter being far more abundant on Earth. Both of the natural isotopes have an unexpectedly low nuclear binding energy per nucleon (5332. 3312±0.0003 MeV for lithium-6 and 5606.
Article first time published onWhat isotopes are used in radiotherapy?
- Iodine-131. This is the most common type of radioisotope therapy. …
- Strontium-89 and Samarium-153. These radioisotopes can be used to treat some types of cancer that have spread to the bones (metastatic bone cancer). …
- Radium-223.
How radioactivity is used in medicine?
Radiation is used in monitoring the response of tumors to treatment and in distinguishing malignant tumors from benign ones. Bone and liver scans can detect cancers that have spread to these organs. Half of all people with cancer are treated with radiation, and the number of those who have been cured continues to rise.
How are medical isotopes produced?
Medical isotopes are used by medical professionals to diagnose and treat health conditions such as heart disease and cancer. The production of medical isotopes is achieved by using two overarching technologies: nuclear reactors, and particle accelerators (linear accelerators, cyclotrons).
What isotope is used in MRI?
The procedure involves the injection of a radioactive isotope (such as technetium-99m or iodine-131) into a blood vessel that supplies the cranial region. As the substance becomes localized within the brain, it decays, therewith emitting gamma rays.
What isotopes are used in CT scans?
The radioisotopes typically used in SPECT to label tracers are iodine-123, technetium-99m, xenon-133, thallium-201, and fluorine-18. These radioactive forms of natural elements will pass through your body and be detected by the scanner. Various drugs and other chemicals can be labeled with these isotopes.
What are the most common radiopharmaceuticals used in medicine?
Technetium-99m is the most extensively used radioisotope in medicine being involved in about 80% of all nuclear medicine procedures.
What is ferrous sulfate used for?
Ferrous sulfate (or sulphate) is a medicine used to treat and prevent iron deficiency anaemia. Iron helps the body to make healthy red blood cells, which carry oxygen around the body. Some things such as blood loss, pregnancy or too little iron in your diet can make your iron supply drop too low, leading to anaemia.
Which isotope is used in treating leukemia?
phosphorus P 32. A radioactive form of the element phosphorus. It is used in the laboratory to label DNA and proteins. It has also been used to treat a blood disorder called polycythemia vera and certain types of leukemia, but it is not commonly used anymore.
How is iron 59 used in medicine?
An iron isotope; a gamma and beta emitter with a half-life of 44.51 days; used as tracer in study of iron metabolism, determination of blood volume, and in blood transfusion studies.
How is uranium used in medicine?
Medicine: radio-isotopes are used for diagnosis and research. Radio-diagnosis can be used to detect disease by injecting certain radio-elements into the human body and observing their paths.
How is tritium used in medicine?
GeneralNatural abundance10−18 in hydrogenHalf-life12.32 yearsDecay products3HeIsotope mass3.01604928 u
How is iodine 131 used in medicine?
I-131 is a radioactive iodine salt that alters the mechanism of iodine absorption in the thyroid gland. Its use is particularly useful in the destruction of cells in the thyroid gland that are overactive. It can also be used in diagnostic imaging techniques used for pheochromocytoma and neuroblastoma.
How can isotopes be used in biological research and medicine?
Radioactive isotopes have many useful applications. In medicine, for example, cobalt-60 is extensively employed as a radiation source to arrest the development of cancer. Other radioactive isotopes are used as tracers for diagnostic purposes as well as in research on metabolic processes.
What is the primary use of isotopes in medicine and biological research quizlet?
Radioactive isotopes are isotopes with a spontaneously decaying nucleus, and are used in medicine and biological research. They are used for biological research in carbon dating.
Which isotope is commonly used in biology?
Carbon-12 (12C) is the most abundant of the carbon isotopes, accounting for 98.89% of carbon on Earth. Carbon-14 (14C) is unstable and only occurs in trace amounts. Unstable isotopes most commonly emit alpha particles (He2+) and electrons.
How are isotopes used in everyday life?
1) Some isotopes are used for solving chemical and medical mysteries. 2) Isotopes are also commonly used in the laboratory to investigate the steps of a chemical reaction. 3) The isotopes of uranium is used as a fuel in nuclear reactors. 4) The isotope of iodine is used in the treatment of goitre.
Why are isotopes used?
Isotopes are used to characterize human disease, to detect contraband at international borders, to sterilize medical equipment, and to power batteries for space exploration. Isotopes also enable research in agriculture, astronomy, biology, chemistry, materials science, medicine, and nuclear safety.
What are isotopes give their uses?
Uses of isotopes: An isotope of uranium acts as a fuel in nuclear reactors. In the treatment of cancer, an isotope of Cobalt is used. In the field of medical, an isotope of Iodine is used in the whole-body scan.
Is calcium an isotope?
Isotopes: Natural calcium is comprised of six isotopes: 40Ca, 42Ca, 43Ca, 44Ca, 46Ca, and 48Ca. Five stable and nineteen radioactive isotopes exist ranging from 34Ca to 57Ca. Calcium-40 is a stable isotope containing 20 neutrons.
Is Oxygen an isotope?
The element oxygen (O) is found in three naturally occurring stable isotopes, 18O, 17O, and 16O. The nucleus of each of these oxygen isotopes contains eight protons and either eight, nine, or ten neutrons, respectively.
What are the isotopes of magnesium?
Magnesium is a ubiquitous element in nature and has three naturally occurring stable isotopes, 24Mg, 25Mg and 26Mg, with relative abundance of 78.99%, 10.00% and 11.01%, respectively.