Is titanium a neutral atom

Emma Valentine
Published Mar 12, 2026
Consequently, every atom of titanium must have 22 electrons surrounding its nucleus. … An ion of titanium can have a different number of electrons because it carries a net charge, i.e. is not neutral.
What elements are neutral atoms?
NameHeliumProtons2Neutrons2Electrons2Atomic Number (Z)2
What is the atom of titanium?
atomic number22atomic weight47.867melting point1,660 °C (3,020 °F)boiling point3,287 °C (5,949 °F)density4.5 g/cm3 (20 °C)
How many electrons does a neutral atom of titanium have?
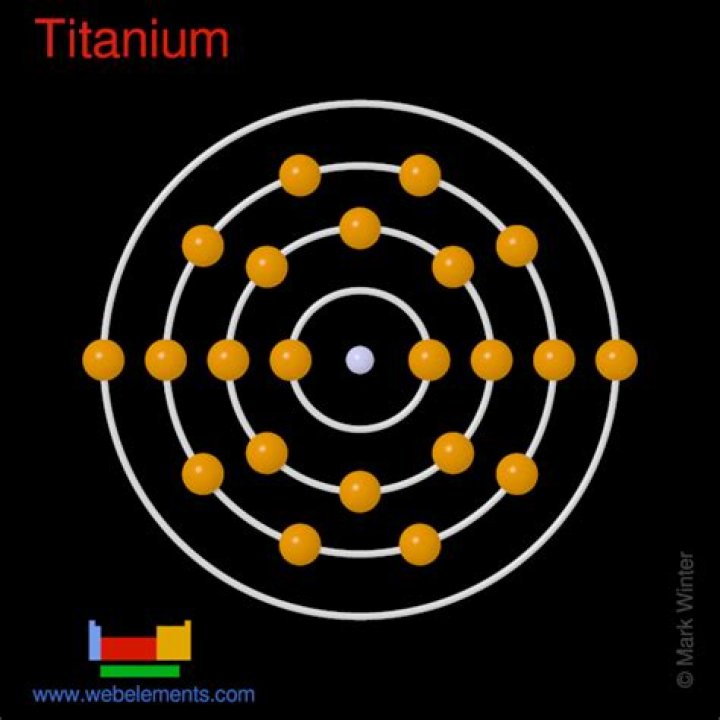
Titanium has the atomic number of 22. This means that a neutral atom of Ti has 22 electrons and 22 protons. Titanium has variable numbers of electrons…What atom is not neutral?
Explanation: If an atom is not neutral, then it will form an ion. In this case, the number of protons and electrons are not equal. If the atom loses one or more electrons, it will have more protons than electrons, and will form a positively charged ion (cation).
What is an example of a neutral element?
Review of Elements, Compounds, and Mixtures Atoms are neutral; they contain the same number of protons as electrons. … A neutral sodium atom, for example, contains 11 protons and 11 electrons. By removing an electron from this atom we get a positively charged Na+ ion that has a net charge of +1.
What is an example of a neutral atom?
Neutral atoms can be turned into positively charged ions by removing one or more electrons. A neutral sodium atom, for example, contains 11 protons and 11 electrons. … Atoms that gain extra electrons become negatively charged. A neutral chlorine atom, for example, contains 17 protons and 17 electrons.
Is titanium a solid liquid or gas?
Titanium is a chemical element with symbol Ti and atomic number 22. Classified as a transition metal, Titanium is a solid at room temperature.What is the orbital configurations of titanium?
10.2. The ground state electron configuration of ground state gaseous neutral titanium is [Ar]. 3d2. 4s2 and the term symbol is 3F2.
How is titanium found in nature?Natural abundance Titanium is the ninth most abundant element on Earth. It is almost always present in igneous rocks and the sediments derived from them. It occurs in the minerals ilmenite, rutile and sphene and is present in titanates and many iron ores.
Article first time published onHow can you identify titanium?
File some dust off it if you can. This won’t be easy because titanium is about as tough as 304 stainless steel. Sprinkle the dust into a gas flame. If it’s titanium, it will burn with a crackling sound and bright white sparks.
What does the titanium atom look like?
TitaniumAppearancesilvery grey-white metallicStandard atomic weight Ar, std(Ti)47.867(1)Titanium in the periodic table
Are all atoms neutral?
Every element has the same number of electrons orbiting the central nucleus. The negative charge from the electrons cancels the positive charge from the protons, so when you consider the whole thing, these atoms are all electrically neutral.
Is NaCl electrically neutral?
Before forming salt, both sodium (Na) and chlorine (Cl) are electrically neutral atoms. … You then have a Na+ cation and Cl- anion that combine into NaCl due to electrostatic attraction. Overall, NaCl is a neutral system (table salt doesn’t shock you when you eat it…
Is Aluminium a neutral atom?
Aluminum has (by definition) 13 protons, 13 positively charged particles. Because aluminum is neutral (as is all matter), it therefore has 13 negatively charged particles, electrons. Aluminum has an atomic mass of about 27 amu; how many neutrons are present in the aluminum nucleus?
Which three particles are neutral atoms?
Neutral atoms have equal numbers of electrons and protons. The third particle is the neutron. It has a neutral charge, also known as a charge of zero.
How do you know if a atom is neutral?
When an atom has an equal number of electrons and protons, it has an equal number of negative electric charges (the electrons) and positive electric charges (the protons). The total electric charge of the atom is therefore zero and the atom is said to be neutral.
Is sodium neutral?
We know that the atomic number of sodium is 11. This tells us that sodium has 11 protons and because it is neutral it has 11 electrons.
Is titanium a transition metal?
Titanium is a transition metal, meaning it can form bonds using electrons from more than one of its shells, or energy levels. It shares this feature with other transition metals, including gold, copper and mercury.
How many electrons shells does titanium have?
Classification:Titanium is a transition metalProtons:22Neutrons in most abundant isotope:26Electron shells:2,8,10,2Electron configuration:[Ar] 3d2 4s2
Do you fill 4s before 3d?
The Order of Filling Orbitals The aufbau principle explains how electrons fill low energy orbitals (closer to the nucleus) before they fill higher energy ones. … This means that the 4s orbital which will fill first, followed by all the 3d orbitals and then the 4p orbitals.
Why is titanium not a noble metal?
titanium, niobium, and tantalum are not included as noble metals despite the fact that they are very resistant to corrosion.
Is titanium an alloy?
Titanium alloy is an alloy consisting primarily of pure titanium with other various metals or chemical elements distributed throughout. It’s produced by mixing a specific ratio of titanium to other metals and chemical elements.
Is titanium paramagnetic or diamagnetic?
Titanium (atomic number 22) is known to be paramagnetic.
Is titanium a pure substance or mixture?
Titanium is not found as a pure element in nature, but is found in compounds as part of minerals in the Earth’s crust. It is the ninth most abundant element in the Earth’s crust.
What is the texture of titanium?
Edit. Titanium is a silvery, light, strong metal. It is odorless. Its texture is rough as well as sharp.
Why is titanium not magnetic?
It turns out that titanium is weakly magnetic (compared to other ferromagnetic materials) in the presence of an externally applied magnetic field. Titanium also exhibts the Lenz Effect but to a lesser extent that many other metals. … The result is that the moving magnet causes the metal to move without touching it.
What is pure titanium used for?
But unalloyed titanium has uses in sophisticated components due to its formability while alloys have applications in strength-requiring parts. Pure titanium has extensive uses in architecture, desalination, hydrocarbon processing, automotive components, surgical equipment, human implants and aerospace.
Why is titanium so light?
The density or “specific gravity” indicates how much mass is packed into a certain volume. … Iron (the mass of steel alloys) has only a slightly heavier atom than Titanium, but its electron shells fit more tightly together. Therefore, Titanium is more than proportionately lighter.
What is so special about titanium?
There are several things that are special about titanium. … Titanium metal is a very durable metal for engineering applications because this metal is corrosion-resistant and also this metal is very strong and very light. It is 40% lighter than steel but as strong as high-strength steel.
Do metal detectors detect titanium?
Metal detectors don’t literally detect metal material but the magnetic field that metal produces. Titanium is non-ferrous (non-iron based) so it has an extremely low magnetic field. … Therefore, the airport metal detectors are not set off.