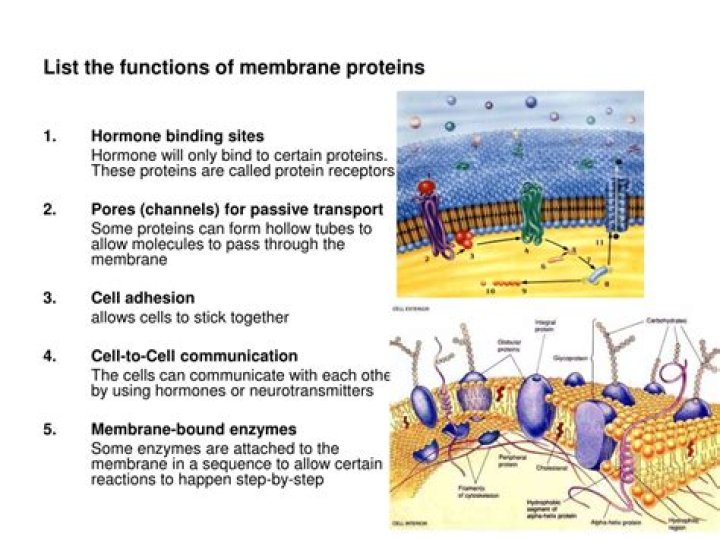
Is stearic acid an organic compound

Sophia Edwards
Published Mar 10, 2026
belongs to the class of organic compounds known as long-chain fatty acids. These are fatty acids with an aliphatic tail that contains between 13 and 21 carbon atoms.
Is stearic acid inorganic or organic?
Stearic acid, also known as stearate or 18:0, belongs to the class of organic compounds known as long-chain fatty acids.
Is stearic acid natural?
stearic acid, also called Octadecanoic Acid, one of the most common long-chain fatty acids, found in combined form in natural animal and vegetable fats.
Is stearic acid a compound or element?
Stearic acid, also known as stearate or N-octadecanoic acid, is a member of the class of compounds known as long-chain fatty acids. Long-chain fatty acids are fatty acids with an aliphatic tail that contains between 13 and 21 carbon atoms. Thus, stearic acid is considered to be a fatty acid lipid molecule.Is ethyl stearate an organic compound?
Ethyl stearate belongs to the class of organic compounds known as fatty acid esters. These are carboxylic ester derivatives of a fatty acid.
Is benzoic acid an organic compound?
benzoic acid, a white, crystalline organic compound belonging to the family of carboxylic acids, widely used as a food preservative and in the manufacture of various cosmetics, dyes, plastics, and insect repellents.
Is nacl organic or inorganic compound?
Sodium chloride is an inorganic chloride salt having sodium(1+) as the counterion. It has a role as an emetic and a flame retardant. It is an inorganic chloride and an inorganic sodium salt.
Is stearic acid covalent or ionic?
Our group determined that stearic acid, salicylic acid, and sucrose were all covalent reagents, while sodium chloride, potassium iodide, and sodium nitrate were ionic compounds.Why is acetic acid an organic compound?
It is an organic compound that is classified as a carboxylic acid because a carboxyl (-COOH) group is present in its chemical structure. Acetic acid is also known as the second simplest carboxylic acid.
Is stearic acid a lipid?NamesPreferred IUPAC name Octadecanoic acidOther names Stearic acid C18:0 (Lipid numbers)IdentifiersCAS Number57-11-4
Article first time published onIs stearic acid healthy?
Stearic acid is considered a healthy saturated fat and does not appear to raise your risk of heart disease. This held true even in a 40-day study in people whose stearic acid intake constituted up to 11% of their total calorie intake ( 9 ).
What is the source of stearic acid?
Major food sources of stearic acid for adults are meat/ poultry/fish, grain products, and milk/milk products (Table 1). Fats rich in stearic acid include cocoa butter (typically consumed as chocolate), mutton tallow, beef tallow, lard, and butter.
Is stearic acid an emulsifier?
Stearic acid is an emulsifier by itself, but can also be used with triethanolamine. When mixed, the two compounds react and become a paste called ‘triethanolamine stearate’ that helps to create a loose emulsion that is easily absorbed by the skin. Lush use a vegetable-based stearic acid, derived from palm oil.
Is stearic acid soluble in ethanol?
Stearic acid is a saturated fatty acid that can deposit on the surface in special conditions. This acid is insoluble in water and soluble in ethanol.
Is stearic acid a carboxylic acid?
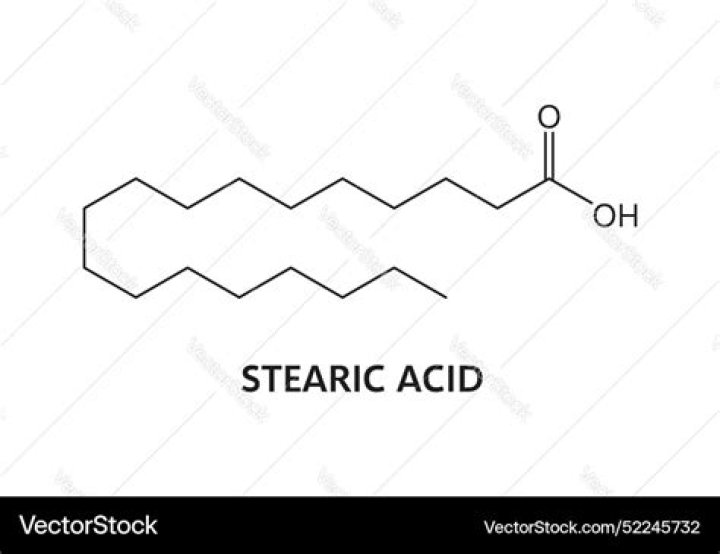
Stearic Acid is a typical example of a fatty acid, which are essentially long hydrocarbon chains containing a carboxyl group at one end and a methyl group at the other. … Stearic acid is a saturated acid, since there are no double bonds between neighbouring carbon atoms.
Is stearic acid a wax?
Stearic acid is a fatty acid that is derived from tallow or vegetable oil. Stearic is a very effective wax modifier that has been used in candle making for well over 150 years. Also referred to as stearine.
Is no2 an organic compound?
Nitro compounds are organic compounds that contain one or more nitro functional groups (−NO 2). … Nitro groups are rarely found in nature.
Is nh3 organic or inorganic?
Ammonia is an inorganic compound composed of a single nitrogen atom covalently bonded to three hydrogen atoms that is an amidase inhibitor and neurotoxin. It is both manufactured and produced naturally from bacterial processes and the breakdown of organic matter.
Is nahco3 an organic compound?
SynonymSourceBicarbonate OF sodaChEBIBicarbonate of sodabiospiderBicarbonate, sodiumMeSH
Is naphthalene an organic compound?
Naphthalene is an organic compound with formula C10H8. It is the simplest polycyclic aromatic hydrocarbon, and is a white crystalline solid with a characteristic odor that is detectable at concentrations as low as 0.08 ppm by mass.
Is phenol an organic compound?
Phenols are organic compounds which contain a hydroxyl (—OH) group attached to a carbon atom in a benzene ring. … Phenol, or hydroxybenzene, is the parent compound of the phenols, consisting of an OH group directly connected to a benzene ring.
Is aniline organic or inorganic?
Aniline is an organic compound with the formula C6H5NH2. Consisting of a phenyl group attached to an amino group, aniline is the simplest aromatic amine.
Is acetic acid organic or inorganic?
Acetic acid, also known as ethanoic acid, is an organic chemical compound best recognized for giving vinegar its sour taste and pungent smell. It is one of the simplest carboxylic acids and has the chemical formula CH3COOH.
What kind of organic compound is ethanoic acid?
Acetic acid , systematically named ethanoic acid , is an acidic, colourless liquid and organic compound with the chemical formula CH3COOH (also written as CH3CO2H, C2H4O2, or HC2H3O2).
Is glucose organic or inorganic?
An example of an organic compound is glucose (C6H12O6), which is shown in Figure 3.12. Glucose is a simple sugar that living cells use for energy. All other compounds are called inorganic compounds. Water is an example of an inorganic compound.
Is NaCl ionic or covalent?
Ionic bonds usually occur between metal and nonmetal ions. For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons.
Why is stearic acid insoluble in water?
Stearic acid has the following structure: … While the carboxylic group makes stearic acid soluble in water due to the potential hydrogen bonding and its polar character, the long hydrocarbon chain is hydrophobic and repels the water molecules to the extent that the compound is insoluble in water.
Why is stearic acid nonpolar?
Consider stearic acid, a type of lipid known as a fatty acid. It has an 18 C atom chain with a carboxylic acid group (RCO2H) at one end. The carboxyl end (shown in red) containing the two oxygens is polar, but the rest of the molecule is completely nonpolar.
Is stearic acid soluble in oil?
Stearic Acid is a waxy, colorless or white solid that exudes a mild odor. It is soluble in oil but only slightly dissolves in water, thus it floats. Stearic Acid is a long-chain fatty acid that, due to its 18-carbon chain, is also referred to as Octadecanoic Acid.
Is stearic acid saturated monounsaturated or polyunsaturated?
III. General Features of Fatty Acid Structure One system of fatty acid classification is based on the number of double bonds. Stearic acid is a typical long chain saturated fatty acid. Oleic acid is a typical monounsaturated fatty acid. Linoleic acid is a typical polyunsaturated fatty acid.
Is formic acid a lipid?
Lipid numberC1:0NameCommonFormic acidSystematicMethanoic acidSalt/Ester NameCommonFormateSystematicMethanoate