Is sodium element a solid

Mia Kelly
Published Feb 27, 2026
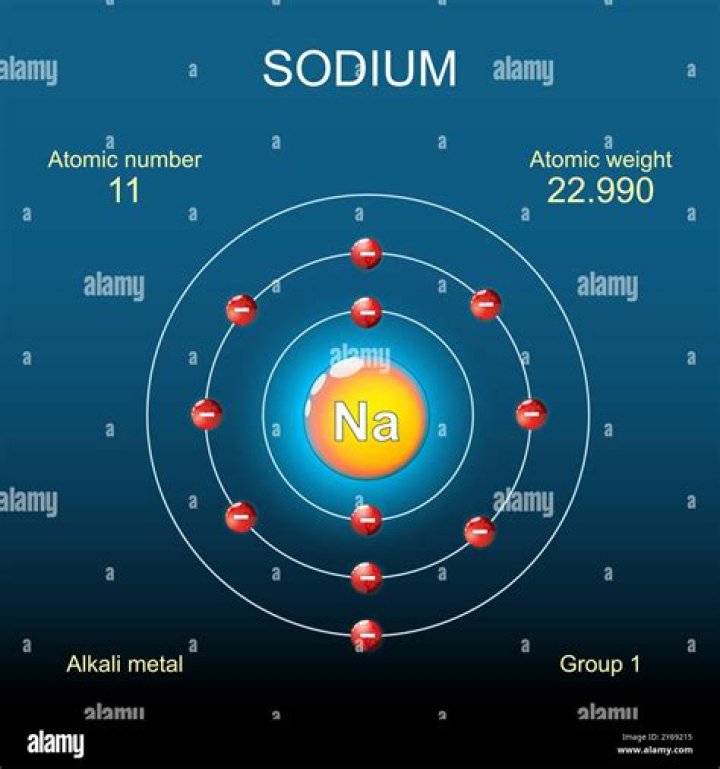
Sodium is a chemical element with symbol Na and atomic number 11. Classified as an alkali metal, Sodium is a solid at room temperature.
What state of matter is sodium?
Group197.794°C, 208.029°F, 370.944 KBlocks0.97Atomic number1122.990State at 20°CSolid23NaElectron configuration[Ne] 3s17440-23-5
Is sodium a gas?
when liquid (at m.p. ) Sodium is a chemical element with the symbol Na (from Latin natrium) and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table.
Is sodium a liquid at room temperature?
solid at room temp.What is the texture of sodium?
Chemical element, symbol: Na, atomic number: 11 and atomic weight 22,9898. It’s a soft metal, reactive and with a low melting point, with a relative density of 0,97 at 20ºC (68ºF).
Is Salt an element?
Chemically, table salt consists of two elements, sodium (Na) and chloride (Cl). Neither element occurs separately and free in nature, but are found bound together as the compound sodium chloride.
What is liquid sodium?
Liquid sodium metal Elemental sodium (Na) is a liquid metal like mercury and solder. Liquid sodium metal looks almost exactly like mercury (photo is actually mercury, from Medvedev) Sodium metal is used as a low-pressure coolant. It transfers heat extremely well and can keep a core cool without external power.
Is sodium a metal or non metal?
Sodium is a very soft silvery-white metal. Sodium is the most common alkali metal and the sixth most abundant element on Earth, comprising 2.8 percent of Earth’s crust.What gas is formed when sodium reacts with water?
The reaction of sodium and water produces hydrogen gas and heat, which is not a good combination! Sodium must be stored under mineral oil, or some other high-molecular weight hydrocarbon. Chlorine gas is toxic, and extremely irritating to the eyes and mucous membranes.
What type of reaction is sodium and water?In what way and in what form does sodium react with water? A colourless solution is formed, consisting of strongly alkalic sodium hydroxide (caustic soda) and hydrogen gas. This is an exothermic reaction.
Article first time published onDoes sodium float on water?
lithium, sodium and potassium are less dense than water so they float on the surface of the water. the metals move about the surface of the water and fizz as hydrogen gas is produced. … sodium also melts to form a silvery-grey ball on the surface of the water.
What element is a gas at room temperature?
Elemental hydrogen (H, element 1), nitrogen (N, element 7), oxygen (O, element 8), fluorine (F, element 9), and chlorine (Cl, element 17) are all gases at room temperature, and are found as diatomic molecules (H2, N2, O2, F2, Cl2).
Is hydrogen solid liquid or gas?
Hydrogen is colorless, odorless and tasteless in nature. It exists as a diatomic form i.e. in the form of gas at room temperature but at very high pressure and low temperature, it exists in the form of liquid hydrogen. And at very low temperatures i.e. at 14K, it exists in the form of solid state.
Is Neon solid liquid or gas?
Neon has the smallest temperature range (2.6 degrees C or 4.7 degrees F) for which it is a liquid, according to Chemicool. Neon, along with helium, argon, krypton and xenon, make up the group known as noble gases.
Is salt water solid or liquid?
In a chemistry class, we take solid salt NaCl (s) and dissolve it into pure liquid water H2O (l). The result is a salt water solution, denoted NaCl (aq). The solution is neither solid nor liquid.
What is the hardness of sodium?
numbersymbolMohs hardness6C10.011Na0.512Mg2.513Al2.75
Why is sodium a soft metal?
sodium $\left( \text{Na} \right)$: Sodium element is present in group 1 due to presence of one valence electron. It has a large atomic size, because of all these reasons sodium metal is a soft metal. Moreover, the metallic bonding in sodium is not strong, hence it can be easily cut.
Is sodium a pure substance or a mixture?
Ordinary table salt is called sodium chloride. It is considered a pure substance because it has a uniform and definite composition.
What is liquid sodium made of?
LBE is an eutectic alloy of lead (44.5%) and bismuth (55.5%) and is being considered instead of lead as an option for the LFR. One of the main advantages of LBE is its melting point of 123.5°C, which is significantly lower than that of lead and quite close to that of sodium.
What are the properties of liquid sodium?
Atomic number11Density (solid)m.p.0.951 g/cm³Density (liquid)m.p.0.927 g/cm³400°C0.856 g/cm³550°C0.820 g/cm³
Who makes liquid sodium?
Manufacturer, Supplier & Distributor Liquid Sodium Carbonate, 16% – HAWKINS manufactures a high purity, liquid SODIUM CARBONATE for use in a variety of industries including food and dairy, printed circuit board production and as a pH control additive in the oil industry.
Is a water an element?
Answer. Water is a compound. It contains more than one element: hydrogen and oxygen atoms are joined together; as illustrated in the video clip Elements and Compounds, above.
What are three examples of an element?
Common examples of elements are iron, copper, silver, gold, hydrogen, carbon, nitrogen, and oxygen.
Is Diamond an element?
Diamond is composed of the single element carbon, and it is the arrangement of the C atoms in the lattice that give diamond its amazing properties. Compare the structure of diamond and graphite, both composed of just carbon.
When is solid sodium added to liquid water?
When solid sodium is added to liquid water it reacts with water to produce hydrogen gas and aqueous sodium hydroxide.
Which gas is formed when sodium reacts with metal?
Reaction with air, water, and hydrogen The corrosion of solid sodium by oxygen also is accelerated by the presence of small amounts of impurities in the sodium. In ordinary air, sodium metal reacts to form a sodium hydroxide film, which can rapidly absorb carbon dioxide from the air, forming sodium bicarbonate.
Why does sodium react with water?
Its single outer electron makes the metal highly reactive and ready to combine with others at the first opportunity – such as the moment the metal hits water. According to the textbooks, these reactive electrons rip apart the surrounding water molecules to release hydrogen gas and heat.
Why is sodium named Na?
Na.” A soft, silvery white and highly reactive metal, sodium was first isolated in 1807 by Humphry Davy during the process of electrolysis of sodium hydroxide. It’s symbol and name derive from the Latin Natrium or Arabicnatrun and the Egyptian word ntry (Natrun), all of which refer to soda or sodium carbonate.
Is sodium a metal nonmetal or semimetal?
Explanation: Sodium is a soft, silvery metal and is a member of the alkali metal family about the periodic table. It is also highly reactive.
Which one is an ore of sodium?
Detailed Solution. Borax is an ore of Sodium. Its chemical formula is Na2B4O7. 10H2O.
Is sodium and water a displacement reaction?
The fast reaction between water and sodium is an intermolecular redox reaction.