Is och3 a strong activator

Christopher Lucas
Published Mar 11, 2026
Any group with decreases the rate (relative to H) is called adeactivating group. Common activating groups (not a complete list): Alkyl, NH2, NR2, OH, OCH3, SR. Common deactivating groups (not a complete list): NO2, CF3, CN, halogens, COOH, SO3H.
Is OCH3 a strong activating or deactivating?
Any group with decreases the rate (relative to H) is called adeactivating group. Common activating groups (not a complete list): Alkyl, NH2, NR2, OH, OCH3, SR. Common deactivating groups (not a complete list): NO2, CF3, CN, halogens, COOH, SO3H.
Is ch3o activating or deactivating?
As we just saw, CH3 is a perfect example of an activating group; when we substitute a hydrogen on benzene for CH3, the rate of nitration is increased. A deactivating group, on the other hand, decreases the rate of an electrophilic aromatic substitution reaction, relative to hydrogen.
Is OCH3 an activator?
Methoxy group (och3) is an electron-withdrawing group and hence is it is a deactivator.Is OCH3 more reactive than CH3?
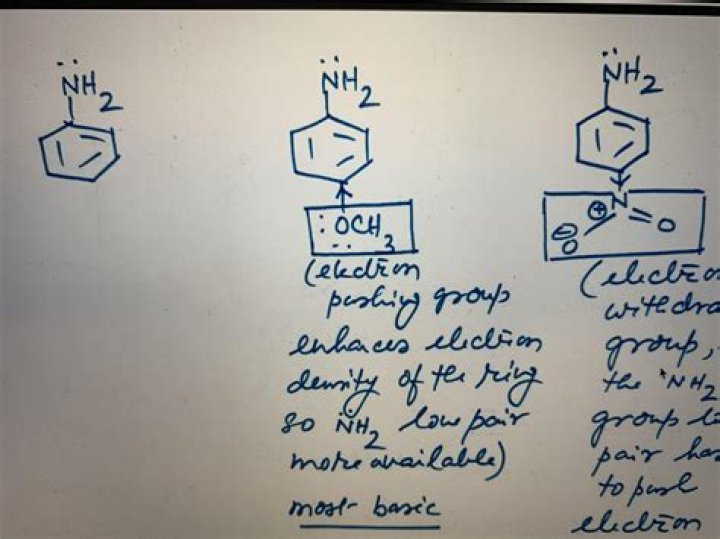
Both are EDG, OCH3 is more than CH3 due to the presence of lone pairs of electrons on oxygen. OCH3 group have +M effect by resonance, but CH3 group have +I effect and hyper conjugation. Again OCH3 group attached to the benzene to promote Ortho Para substitution in EAS, electrophilic substitution in Aromatic system.
Which group is more powerful than ch3group?
The functional group more powerful than CH₃ is –COOH, -OH, -CHO,-CN. Among all the options -CH₃(alkyl group) has the least priority and the functional group -COOH (carboxylic acids) is at the top of the list.
Does OCH3 show +R effect?
Yes, OCH3 which belongs to the is the electron-withdrawing group (methoxy group). Here, the oxygen (in OCH3) is more electronegative than carbon due to which it will show -I effect which is electron-withdrawing. On the other hand, OCH3 will lose or release electrons through resonance showing +R effect.
Why is OCH3 more electron-withdrawing than Oh?
OCH3 is more electron withdrawing group. REASON :- As methyl group is bigger than H atom . There is stearic repulsion between methyl group and lone pairs of Oxygen atom , thus leading to development of slight +ve charge on Oxygen atom. Whereas there is no such repulsion in case of H atom.Is OCH3 more stable than CH3?
CH3-CH(+)-OCH3 is more stable than CH3-CH(+)-CH3 because the mesomeric effect of O atom stabilises the carbocation to a greater extent than the inductive effect if CH3 can.
What functional group is OCH3?A methoxy group is the functional group consisting of a methyl group bound to oxygen. This alkoxy group has the formula O–CH3. On a benzene ring, the Hammett equation classifies a methoxy substituent at the para position as an electron-donating group, but as an electron-withdrawing group if at the meta position.
Article first time published onWhich is more activating OH or OCH3?
OH (hydroxy) has a better +R effect than OCH3 because The -CH3 group in -OCH3 causes steric repulsions with lone pairs, increasing bond angles. Due to which it make -O- atom of -OCH3 more electro-negative and thus affecting donor tendency.
Is OCH3 an electron withdrawing group?
Complete answer: Yes, the $OC{H_3}$ is an electron withdrawing group. The oxygen atom in the $OC{H_3}$ group is more electronegative than the carbon atom. Due to this reason, it will show the $ – I$ effect which is electron-withdrawing.
Why is NH2 more activating than Oh?
Electronegativity of oxygen is more than that of nitrogen. Thus NH2 can donate its lone pair more easily than oh group whereas OH group has more electron pair pulling tendency. Hence nh2 shows a stronger +R effect than oh 🙂 OH and NH2 have different modes of donation and withdrawal.
Is OCH3 a strong or weak base?
An alkoxide molecule has more steric hindrance and less distinct polarization, making it less attracted to the positive carbon center. So OCH3− is a weaker nucleophile than the hydroxide ion in a protic solvent though OCH3− is a stronger base.
Which is more activating OCH3 or NH2?
–NH2 will give more electron density than it takes, and thus -NH2 is an electron donating group.
Is OCH3 a meta director?
As it turns out, the ortho and para positions for the two groups are the same. The aldehyde group is electron-withdrawing and meta-directing. Okay, this one isn’t so clear. Both –OCH3 and –Ph are activating, ortho-/para-directing groups.
Is OCH3 an ether?
In the IUPAC Nomenclature system, ethers are named using the general formula “alkoxyalkane”, for example CH3–CH2–O–CH3 is methoxyethane. If the ether is part of a more-complex molecule, it is described as an alkoxy substituent, so –OCH3 would be considered a “methoxy-” group.
Is OCH3 stronger than OH?
With your question, -OCH3 is a larger molecule (more electrons from the methyl donating group) and will more easily donate electrons (think kinetics), but it is also a weaker base than -OH.
Which has more Mesomeric effect OH or OCH3?
+R effect of OCH3 is more than that of OH group. +R effect: The groups show positive mesomeric effect when they release electrons to the rest of the molecule by delocalization.
What is charge on OCH3?
O. OCH3. OH. As drawn there is no. negative charge on this.
What is the most stable carbocation?
Benzylic carbocations are so stable because they have not one, not two, but a total of 4 resonance structures. This shares the burden of charge over 4 different atoms, making it the MOST stable carbocation.
Is methoxy an electron withdrawing group?
b) -OCH3 (methoxy group) The methoxy group is electron withdrawing by the inductive effect of the oxygen atom, since the electronegativity of oxygen is 2.6.
Does CH3 show Mesomeric effect?
Since ch3 is an O – P director it thereby shows only +I effect no +R at meta but when Ch3 is attached to Ortho/Para position it shows +R >>> +I . This +R effect is only called Hyperconjugation type effect it is not actually Hyperconjugation but equally good in showing effect like it .
Why is OCH3 a good leaving group?
It is not. However, in an acidic medium, the -OCH3 will be protonated to form -OHCH3+, which is an excellent leaving group, since upon dissociation, it is simply methanol, a small, neutral molecule. There are obviously other ways to turn an alcohol into a leaving group.
Why is +M effect of OH greater than or?
In some books it is written that +M (mesomeric effect) effect of OH is less than that of OR . The reason they give is inductive effect of R (group) which sounds senseful . But at some places it is written +M of OR is less (which is correct).
What compound is OCH3?
Methylthaizolo-OCH3 | C26H22N2O3S – PubChem.
What does NaOCH3 do in organic chemistry?
Since NaOCH3 is a strong nucleophile and base, it will force a 2nd-order mechanism. It is not a bulky base, so the 2° alkyl halide will give a mixture of E2 and SN2 products. b. This bicyclic compound is locked into a single chair-flipped conformation, which has no adjacent hydrogens anti-coplanar to the bromine.
What functional group is ch3och2ch3?
Note: The above compound is categorized under the ether functional group and its name will be ethyl methyl ether. It is a colorless ether.
Why hydroxyl group is a strong activator?
Most ring activators have atoms with unshared electron pairs directly attached to a carbon atom of the benzene ring. For example, the — OH group has two pairs of unshared electrons on the oxygen atom, which will form a bond to a carbon atom of the benzene ring. Thus, the — OH group will be an activating group.
Which is more deactivating NO2 or SO3H?
Since NO2 is a strong, actually, the strongest, -R group. Hence, it decreases the electron density at ortho and para positions on a Benzene ring. SO3H is a weaker -R group as compared to NO2 and hence less deactivating.
What are the electronic effect exerted by OCH3 and CL?
The electronic effects exerted by – Cl and – OCH3 are as follows : (1) Cl being more electronegative atom it pulls the bonding electrons towards itself. This is known as negative inductive effect (- I). (2) – OCH3 is less electronegative group which repels the bonding electrons away from it.