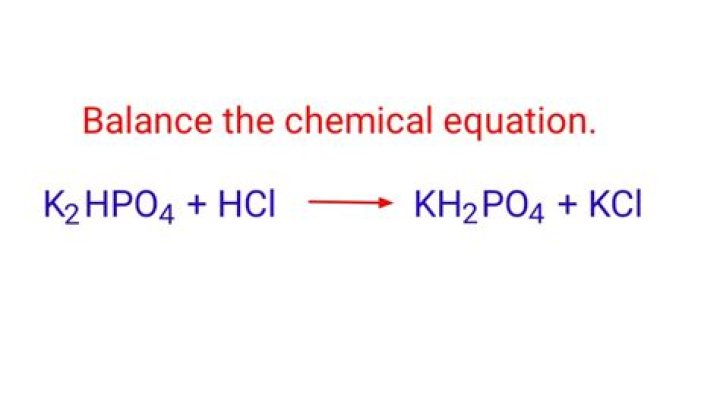
Is KH2PO4 basic or acidic

Robert Spencer
Published Feb 27, 2026
KH2PO4 will generate OH- ions when disolved in water (therefore pH is rising), because is a salt derived from a strong alkali (KOH) and a weak acid (H3PO4). This is the phenomenon occured in the alkaline hydrolysis of all salts derived from a strong alkali and a weak acid when disolved in water.
Is KH2PO4 an acid salt?
KH2PO4 – Monopotassium Phosphate Monopotassium Phosphate is the monopotassium salt of phosphoric acid with the formula KH2PO4. It is freely soluble in water and insoluble in ethanol.
What kind of compound is KH2PO4?
Monopotassium phosphate (MKP) (also, potassium dihydrogenphosphate, KDP, or monobasic potassium phosphate) is the inorganic compound with the formula KH2PO4. Together with dipotassium phosphate (K2HPO4. (H2O)x) it is often used as a fertilizer, food additive, and buffering agent.
Is KH2PO4 a pH?
ComponentAmountConcentrationKH2PO4 (mw: 136.09 g/mol)888 mg0.0065 MIs NaH2PO4 a base or acid?
NaH2PO4 is much more acidic than Na2HPO4, hence NaH2PO4 acts as an acid while Na2HPO4 acts a salt of NaH2PO4 with a strong base NaOH. Hence mixture is having a weak acid NaH2PO4 and its salt with a strong base Na2HPO4 and that’s why mixture acts as an acidic Buffer.
What are the ions in KH2PO4?
The names of K2HPO4 K 2 H P O 4 and KH2PO4 K H 2 P O 4 are potassium hydrogen phosphate and potassium dihydrogen phosphate.
Is na3po4 an acid or base?
As salts are composed of acid and base. Few are made from strong acid and weak base while few salts are composed of strong base and weak acid. So, trisodium phosphate, $ N{a_3}P{O_4} $ is slightly acidic in nature.
How do you make KH2PO4 buffer?
- Prepare 800 mL of distilled water in a suitable container.
- Add 95 g of KH2PO4 to the solution.
- Add 52.5 g of K2HPO4 to the solution.
- Adjust the pH to 6.5.
- Filter sterilize and store at room temperature.
What is the charge of KH2PO4?
Property NameProperty ValueReferenceFormal Charge0Computed by PubChem
Is KH2PO4 hygroscopic?Shelf life: Poor shelf life, hygroscopic, store in a cool, dry environment.
Article first time published onIs K2HPO4 same as KH2PO4?
The terms monobasic and dibasic refers to the number of potassium cations bound to the phosphate molecule. The key difference between KH2PO4 and K2HPO4 is that KH2PO4 is monobasic and can release a low amount of potassium, whereas K2HPO4 is dibasic and can release a high amount of potassium when used in fertilizer.
Is potassium phosphate an acid?
NamesOther names Potassium phosphate monobasic; Phosphoric acid, monopotassium salt; Potassium biphosphateIdentifiersCAS Number7778-77-0
Is CH3COONa an acid or base?
Solid sodium acetate (CH3COONa) is a salt, not an acid or a base. It is a salt formed from a weak acid (CH3COOH) and a strong base ( NaOH). However, on hydrolysis of CH3COONa in water, the resultant solution is basic.
Is NaH2PO4 an acid salt?
NaH2PO4 and NaHPO3 are acid salts. … Oxoacids are those acids that contain oxygen bonded with hydrogen and one more metal.
Is Na2HPO4 a weak acid?
Sodium dihydrogen phosphate NaH2PO4 (monobasic) and sodium hydrogen phosphate Na2HPO4 (dibasic) are a weak acid and its conjugate base pair that are mixed to make a buffer with pH 7.2.
Is Na3PO4 ionic or covalent?
Sodium phosphate is an ionic compound composed of the sodium cation and phosphate anion.
Is po43 a weak acid?
Acid Equilibria Phosphate ion is a reasonably strong base. It hydrolyzes in water to form a basic solution.
Is Na3PO4 a basic salt?
The name often used is trisodium phosphate, to distinguish it from other combinations of sodium and phosphate: NaH2PO4 and Na2HPO4. Trisodium phosphate, Na3PO4, is a very basic salt that is readily dissolved in water and found a great deal of use in cleaning products. This is an example of normal salt in chemistry .
What is the oxidation number of potassium in KH2PO4?
Looking at our formula we can conclude: Potassium is present with an oxidation state of +1. Hydrogen is present with an oxidation state of +1 each. Oxygen is present with an oxidation state of -2 each.
How do you calculate phosphorus in KH2PO4?
If you add 4.390 g KH2PO4 in 1000 ml volumetric flask and make the volume to 1L with water, you will get 1000 ppm P solution. if you dissolve 0.439 g KH2PO4 (AR Grade) in de-ionized water and make the volume to 1 litre. you will get 100 ppm solution.
What is the name of cahpo4?
PubChem CID24441StructureFind Similar StructuresChemical SafetyLaboratory Chemical Safety Summary (LCSS) DatasheetMolecular FormulaCaHPO4 or CaHO4PSynonyms7757-93-9 CALCIUM HYDROGEN PHOSPHATE Calcium phosphate dibasic Dicalcium phosphate Phosphoric acid, calcium salt (1:1) More…
How do you take mono potassium phosphate?
To apply Haifa MKP™ in combination with calcium or magnesium fertilizers, use two fertilizer tanks. If the system includes only one tank, apply the fertilizers at different times. Each kilogram of Haifa MKP™ in the tank provides 520 grams of P2O5 (230g P) and 340 grams of K2O (280g K).
What is the name for mghpo4?
Dimagnesium phosphate is a compound with formula MgHPO4. It is a Mg2+ salt of monohydrogen phosphate.
What is the molar mass of po4?
PubChem CID1061Molecular FormulaO4P-3Synonymsphosphate orthophosphate 14265-44-2 Phosphate ion O-Phosphate More…Molecular Weight94.971DatesModify 2021-12-11 Create 2004-09-16
What is the difference between mono potassium and potassium?
is that monopotassium is (chemistry) monatomic potassium while potassium is a soft, waxy, silvery reactive metal that is never found unbound in nature; an element (symbol” k) with an atomic number of 19 and atomic weight of 390983 the symbol is derived from the latin ”kalium .
What is the pKa of KH2PO4?
Non-Zwitterionic BufferCompoundFormulapKa at 20 °CImidazoleC4H4N2Potassium Phosphate, monobasicKH2PO47.21Potassium Phosphate, dibasicK2HPO47.21
Is Tripotassium a phosphate?
Tripotassium phosphate (Chemical formula: K3PO4) is an ionic salt that dissolves in water. It is utilized as an additive in various foods, due to its properties as a foaming agent, whipping agent and emulsifier. In poultry processing, it can be an antimicrobial agent in conjunction with fatty acids.
What is the correct name for k2so4?
PubChem CID24507Molecular FormulaK2SO4 or K2O4SSynonymsPOTASSIUM SULFATE 7778-80-5 Potassium sulphate Dipotassium sulfate Sulfuric acid dipotassium salt More…Molecular Weight174.26Component CompoundsCID 1118 (Sulfuric acid) CID 5462222 (Potassium)
Why is KH2PO4 and K2HPO4 a buffer?
It is a buffer, since while adding H ions to K3PO4 (or OH ions to H3PO4) solution you are forming the conjugate base/acid (K2HPO4 for K3PO4 and KH2PO4 for H3PO4) salts in the solution, which is the description of buffer.
What is the pH of 0.1 m KH2PO4?
Prepare 100mL of a 0.1M potassium phosphate buffer of pH 6.5. You have K2HPO4 and KH2PO4 in solid/salt form available to mix with water to create this buffer.
Is K3PO4 a weak base?
Tripotassium phosphate (potassium phosphate tribasic, K3PO4) is a strong inorganic base (pKa = 12.32 for the conjugate acid).