Is heme a prosthetic group

Mia Morrison
Published Apr 06, 2026
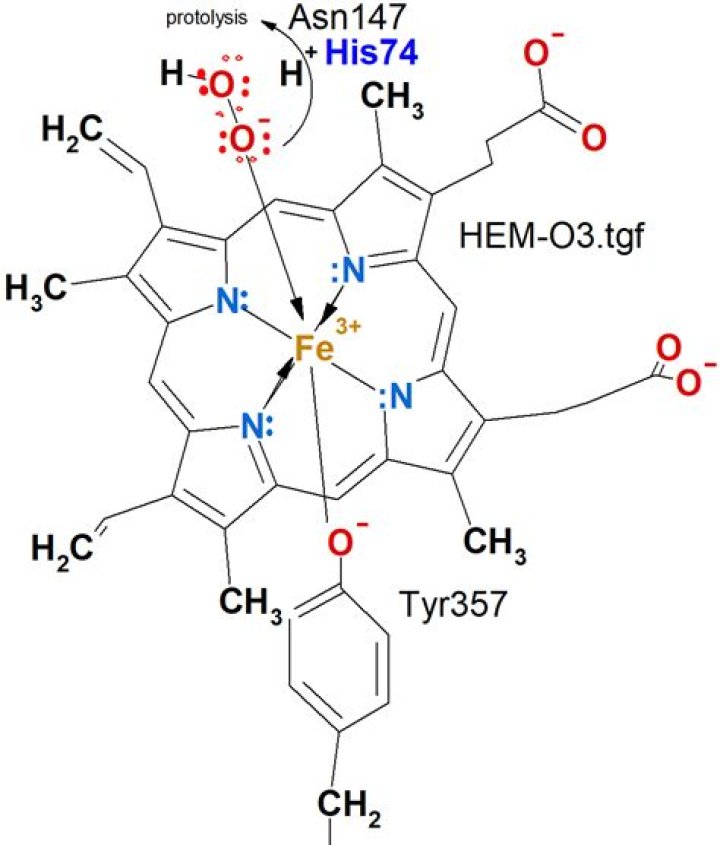
Heme group: A prosthetic group consisting of a protoporphyrin ring and a central iron (Fe) atom < >. A protoporphyrin ring is made up of four pyrrole rings linked by methene bridges. Four methyl, two vinyl, and two propionate side chains are attached.
Is heme a cofactor or coenzyme?
CofactorChemical group(s) transferredDistributionHemeElectronsBacteria, archaea and eukaryotes
What is a prosthetic group of hemoglobin?
The Heme group in hemoglobin is a prosthetic group located in the porphyrin, which is a tetramer of cyclic carbon groups. It contains an organic component called a protoporphyrin made up of four pyrrole rings and an iron atom in the ferrous state (Fe2+).
Are heme groups cofactors?
Since heme serves as an essential cofactor to several proteins involved in central metabolic processes, all organisms have established a conserved biosynthetic pathway to synthesize the cofactor. … Heme biosynthesis pathway.Why heme is a prosthetic group?
Prosthetic groupFunctionDistributionHemeOxygen binding and redox reactionsBacteria, archaea and eukaryotes
What are prosthetic groups in enzymes?
Prosthetic groups are compounds bound to enzymes (covalently or non-covalently) and their change from one form to another and back takes place in a single catalytic cycle. The term cofactors unites coenzymes and prosthetic groups.
What is cofactor and prosthetic group?
Prosthetic groups are cofactors that bind tightly to proteins or enzymes. … They can be organic or metal ions and are often attached to proteins by a covalent bond. The same cofactors can bind multiple different types of enzymes and may bind some enzymes loosely, as a coenzyme, and others tightly, as a prosthetic group.
How are cofactors different from prosthetic group?
A cofactor is a substance that is required for enzyme to be catalytically active,These include organic and inorganic substances but prosthetic group are only the cofactors that are tightly bound to the enzyme.What is an example of a prosthetic group?
Prosthetic groups assist cellular function by participating in cellular respiration and fatty acid synthesis. When bound to proteins, prosthetic groups are called holoproteins. Some examples of prosthetic groups are heme, biotin, flavin, iron sulfides, copper and ubiquinone.
What group is heme?The heme group (a component of the hemoglobin protein) is a metal complex, with iron as the central metal atom that can bind or release molecular oxygen. Both the hemoglobin protein and the heme group undergo conformational changes upon oxygenation and deoxygenation.
Article first time published onIs heme a cofactor for hemoglobin?
For example, in hemoglobin (lower left), the cofactor heme binds the oxygen that is transported from the lungs to the other tissues of the body; the second image is zoomed in on one of the heme molecules. …
What does heme mean?
(heem) The part of certain molecules that contains iron. The heme part of hemoglobin is the substance inside red blood cells that binds to oxygen in the lungs and carries it to the tissues.
Which of the following enzymes has heme as a prosthetic group?
Heme (= haem) is iron containing prosthetic group in cytochromes, haemoglobin, myoglobin, catalase and peroxidase.
What is the role of the heme prosthetic group in hemoglobin quizlet?
-Both hemoglobin and myoglobin contain a prosthetic group called heme, which contains a central iron atom. -By itself, heme is not a good oxygen carrier. It must be part of a larger protein to prevent oxidation of the iron atom. -Hemoglobin and myoglobin are heterotetramers.
Is heme a protein?
Heme proteins are strongly colored proteins, usually reddish-brown, which is due to the presence of the heme moiety. The heme moiety consists of a substituted protoporphyrin ring, containing a liganded iron atom.
What is Aprosthetic group?
Prosthetic groups are cofactors that bind tightly to proteins or enzymes. It is a specific non-polypeptide required for the biological function of some proteins. The prosthetic group may be organic (such as vitamin, sugar or lipid) or inorganic (such as metal ion), but is not composed of amino acids.
Is heme a peptide?
The heme peptide opens the protein active site and, together with the protein backbone, controls the affinity and substrate disposition during catalysis.
Which is an example of a cofactor?
Vitamins, minerals, and ATP are all examples of cofactors. ATP functions as a cofactor by transferring energy to chemical reactions.
Is NADH a prosthetic group?
To start, two electrons are carried to the first complex aboard NADH. … FMN, which is derived from vitamin B2, also called riboflavin, is one of several prosthetic groups or co-factors in the electron transport chain. A prosthetic group is a non-protein molecule required for the activity of a protein.
What is prosthetic group Class 11?
Answer: Prosthetic groups are organic compounds that are tightly bound to the apoenzyme, (anenzyme without cofaetor) by covalent or non-covalent forces, e.g., in peroxidase andcatalase, which catalyze the breakdown of. … It may beinorganic or organic in nature.
What's a cofactor in an enzyme?
Cofactor is a non-protein chemical compound that tightly and loosely binds with an enzyme or other protein molecules. Basically, cofactors are split into two groups: coenzymes and prosthetic groups (ions usually).
Are coenzymes a type of cofactor?
A coenzyme is one type of cofactor. Coenzymes are organic molecules required by some enzymes for activity. A cofactor can be either a coenzyme or an inorganic ion.
What is the prosthetic group in glycoprotein?
– The prosthetic group of glycoproteins consists of carbohydrates. Glycoproteins are found in blood plasma, egg albumin, saliva, mucus and blood group compounds. Thus, the correct answer is option D i.e., Carbohydrate. Note: Glycoproteins are proteins that have sugars bound to them.
Which one of the following is not a cofactor?
An apoenzyme is an inactive enzyme and not a cofactor.
What is prosthetic group Class 12?
A prosthetic group is a non-protein portion obtained by hydrolysis of conjugated proteins. The main function of the prosthetic group is to control the biological functions of proteins.
What is a cofactor vs coenzyme?
Coenzymes and cofactors are molecules that help an enzyme or protein to function appropriately. Coenzymes are organic molecules and quite often bind loosely to the active site of an enzyme and aid in substrate recruitment, whereas cofactors do not bind the enzyme.
Where is the heme group located?
Heme groups are embedded in the hemoglobin protein, so the tutorial gives a view of hemoglobin and its major structural features.
What are the 4 heme groups?
The hemoglobin molecule is made up of four polypeptide chains (Alpha 1, Beta 1, Alpha 2, Beta 2), noncovalently bound to each other. There are four heme-iron complexes. Each chain holds a heme group containing one Fe++ atom. The heme-iron complexes are colored red because they give hemoglobin its red color.
Why is the heme group in hemoglobin flat?
The oxygenated heme assumes a planar configuration, and the central iron atom occupies a space in the plane of the heme group (depicted by a straight red line). … Hence, when a single heme group in the hemoglobin protein becomes oxygenated, the whole protein changes its shape.
How many heme groups are there in hemoglobin?
Each hemoglobin molecule is made up of four heme groups surrounding a globin group, forming a tetrahedral structure. Heme, which accounts for only 4 percent of the weight of the molecule, is composed of a ringlike organic compound known as a porphyrin to which an iron atom is attached.
Is heme an amino acid?
Residues having heavy atoms within 4.5 Å of any non-hydrogen atoms of the heme molecule are identified as heme interacting amino acids. A protein chain is considered as heme binding if it has residue(s) as axial ligand(s) to the heme iron or has at least ten residue interactions with the heme molecule.