Is HClO stronger than HClO3

Mia Kelly
Published May 02, 2026
HClO3 is stronger than HClO. because as the number of oxygen atom increase the acid becomes more stronger.
Which is strongest acid HClO and HClO3?
And electronegativity of chlorine in turn is directly proportional to the oxidation state of chlorine. Thus, the order of acidic strength is HClO < HClO2 < HClO3 < HClO4 As the number of oxygen atoms attached to chlorine increases, acid strength increase. Hence, the correct option is (2).
Which one is the strongest acid HClO HClO2 HClO3 HClO4?
The HClO4 has the most oxygen atoms so it would have to be the strongest acid.
Why is HClO4 stronger than HClO3?
Greater the number of negative atoms present in the oxy-acid make the acid stronger. … In turn, the electrons of H−O bond are drawn more strongly away from the H-atom. The net effect makes it easier from the proton release and increases the acid strength.Is HClO3 a stronger acid than HClO4?
HClO4 is a stronger acid than HClO3 .
Which of the following is strongest acid Hocl?
HCIO4orHOCl3 ( C) is the strongest acid acid.
Which one is the strongest acid HClO?
The strongest acid is perchloric acid (HClO4 ).
What's the strongest acid in the world?
The world’s strongest superacid is fluoroantimonic acid. Fluoroantimonic acid is a mixture of hydrofluoric acid and antimony pentafluoride. The carbonane superacids are the strongest solo acids.Which one is stronger acid and why HClO and HClO2?
With increase in oxidation number of a particular halogen atom, the acidic character of corresponding oxoacid increases. HClO, HClO2, HClO3 and HClO4 respectively. These anions are stabilised to greater extent, it has lesser attraction for proton and therefore, will behave as weaker base.
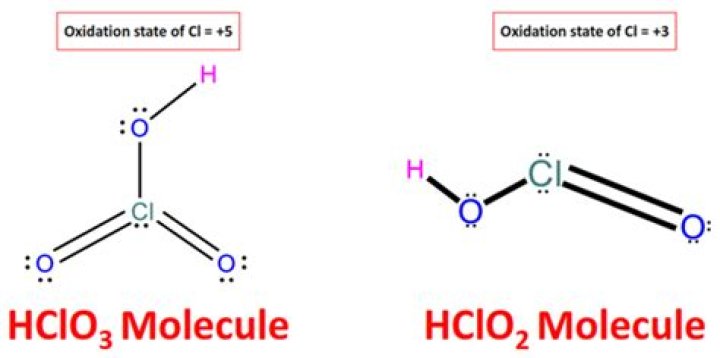
Why is HClO3 a strong acid?The strength of oxo-acids is primarily affected by the number of oxygens double-bonded to the central atom, because these electron-attracting groups weaken the O-H bonds and make the substance acidic. HClO3 and HClO4 are strong acids due to the multiple oxygens bonded to the central atom.
Article first time published onWhy is HClO a weak acid?
These acids are the only ones that fully dissociate in water. All other acids are weak acids. Since HClO is not one of these seven, and there is no -OH group present as there is in bases, HClO is a weak acid. Another way to verify that this is an acid instead of a base is by looking at the ion it dissociates into.
Is HClO a weak acid?
Hypochlorous acid (HOCl or HClO) is a weak acid that forms when chlorine dissolves in water, and itself partially dissociates, forming hypochlorite, ClO−.
Which acid is stronger HClO or HClO2?
The general rule is that the acid is stronger if it has more O atoms in a series such as this. HClO4, perchloric acid, is a very strong acid as is HClO3. HClO2 is a weak acid and HClO is even weaker.
Is HClO or HBrO a stronger acid?
In direct contrast with HCl vs. HBr , HClO is a stronger acid than HBrO , because Cl is more electronegative, which dominates over the size difference between Cl and Br due to the presence of the oxygen.
Is NH3 a strong base?
As it happens, ammonia (NH3) is not a strong acid; it is a weak base. The nitrogen of ammonia is surrounded by four pairs of electrons. Three of the pairs are occupied in binding to the three hydrogen atoms.
Is HOCl or HOBr stronger?
HOCl is the stronger acid because its Ka value is greater than the Ka value of HOBr. … HOI is a weaker acid than HOCl because the O–H bond in HOI is stronger than the O–H bond in HOCl.
Is HClO stronger than HCl?
HCl is a stronger acid because the bond between H and Cl is weaker than the bond between H and F (this is because Cl has a greater atomic radius). HClO is a stronger acid than HBrO because the resulting anion of HClO is more stable as a result of Cl having a greater electron withdrawing power than Br.
Which among the following is the strongest acid HClO3?
– Perchloric acid has three Cl=O. bonds and hence three oxygen atoms will withdraw the electron density from the negatively charged oxygen atom, so in this case the conjugate base will be most stable. Hence we can say that the acidic strength of Perchloric acid will be the highest amongst all.
Is H2CO3 strong or weak acid?
H2CO3 is a weak acid that dissociates into a proton (H+ cation) and a bicarbonate ion (HCO3- anion). This compound only partly dissociates in aqueous solutions. Furthermore, the conjugate base of carbonic acid, which is the bicarbonate ion, is a relatively good base.
Why Hof is stronger acid than HOCl?
As the O−H bond length decreases, the acidic nature of the compound increases. In HOF , the O−H bond length is 96 pm while in HOCl, the O−H bond length is 97 pm and so HOF is more acidic than HOCl.
Why is Hoi stronger than HOCl?
HOCl is a stronger acid than HOI because the Cl is far more electronegative than the I in their respective acids. The more electronegative a molecule is, the less electron density around the H, making it easier for the H to be donated to another molecule.
Is HIO stronger than HClO?
HCl is a weaker acid than HBr and HI due to the greater radii of Br and I, but why is HClO more acidic than HIO? Because Cl is more electronegative, it is more acidic.
Is HClO a strong acid?
seven strong acids: HCl, HBr, HI, HNO3, H2SO4, HClO4, & HClO3 2. … any acid that is not one of the seven strong is a weak acid (e.g. H3PO4, HNO2, H2SO3, HClO, HClO2, HF, H2S, HC2H3O2 etc.) 2. solutions of weak acids have a low concentration of H+.
Which is weaker HClO or HCl?
Acid NameFormulaKaAceticCH3COOH1.8 x 10-5HypochlorousHClO3.0 x 10-8HydrocyanicHCN4.9 x 10-10
What is the pH of HClO?
How is it acid if its pH is >7.
Can acid eat through glass?
In short, acid can dissolve glass. However, only a few acids, mainly acids containing the element fluorine, are corrosive enough to properly dissolve glass. An example of this is hydrofluoric acid, which is able to react with the compound silicon dioxide found in glass, thus dissolving it.
Can acid dissolve diamond?
Nope, diamonds do not dissolve in any acid. Matter of fact, we clean diamonds in a boiling Sulfuric acid and Potassium chloride bath. No, acids cannot dissolve diamonds, for the simple reason that a diamonds carbon atoms are too tightly packed together for the Hydrogen ions to be able to dissolve the substance.
Does acid destroy DNA?
DNA degrades rapidly in low pH and high heat, both of which are produced in a body decomposed by sulfuric acid.
Is HClO3 a strong or weak acid?
The 7 common strong acids are: HCl, HBr, HI, HNO3, HClO3, HClO4 and H2SO4 (1st proton only).
Is HClO a weak or strong electrolyte?
HBr is a strong electrolyte. HClO is a weak electrolyte.
Why is HOCl unstable?
It is a weak acid. The chlorine atom possesses oxidation state +3 in this acid. The pure substance is unstable and disproportionates to hypochlorous acid (Cl oxidation state +1) and chloric acid (chlorine oxidation state +5). Chlorite salts such as sodium chlorite are stable conjugate bases derived from this acid.



