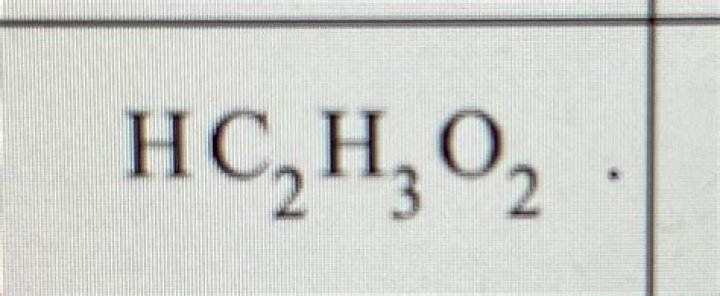
Is HC2H3O2 weak or strong

Emma Valentine
Published Mar 01, 2026
No, it is not classified as a strong acid in aqueous solutions. Strong acids are the acids that completely dissociate in water. Common strong acids are hydrochloric acid (HCl), hydrobromic acid (HBr), hydroiodic acid (HI), sulfuric acid (H2SO4), perchloric acid (HClO4), and nitric acid (HNO3).
Does HC2H3O2 have a strong electrolyte?
Acetic acid is a weak acid, and because it’s a weak acid, it would be a weak electrolyte.
Why is HC2H3O2 a weak acid?
Explanation: Acetic acid, CH3COOH , is a weak acid, because it is present in solution primarily as whole CH3COOH molecules, and very little as H+ and CH3COO− ions. Which furthermore indicates that acetic acid is weak, because strong ions ionize almost completely.
Is HC2H3O2 a weak acid or base?
Problem: Acetic acid, HC2H3O2, is a weak acid that is in equilibrium in the following given reaction: HC2H3O2(aq) + H2O(l) ⇌ H3O+(aq) + CH3CO2-(aq) a.What type of compound is HC2H3O2?
Acetic acid , systematically named ethanoic acid , is an acidic, colourless liquid and organic compound with the chemical formula CH3COOH (also written as CH3CO2H, C2H4O2, or HC2H3O2).
Is HC2H3O2 a MONOprotic acid?
As indicated by the name, a MONOprotic acid has only 1 (mono) hydrogen to give. That would make HC2H3O2 the monoprotic acid.
Is HC2H3O2 soluble?
Acetic acid is extremely soluble in water, but most of the compound dissolves into molecules, rendering it a weak electrolyte.
Is CuSO4 a strong electrolyte?
Two compounds that are strong electrolytes are the ionic compounds ZnSO4 and CuSO4. If the compound is either one of these, then it is definitely a strong electrolyte.Is HNO2 strong or weak?
Nitrous acid (HNO2), a weak acid, is very unstable and exists only in aqueous solution.
Is Ch₃cooh a strong electrolyte?CH3COOH is an acetic acid and it’s not strong acid because only few ions in solution are present to conduct electricity. Hydrochloric (HCl) is a strong electrolyte. Usually, Acetic acid are weak electrolyte.
Article first time published onWhich is the strongest electrolyte?
Strong Electrolytesstrong acidsHCl, HBr, HI, HNO3, HClO3, HClO4, and H2SO4strong basesNaOH, KOH, LiOH, Ba(OH)2, and Ca(OH)2saltsNaCl, KBr, MgCl2, and many, many moreWeak Electrolytesweak acidsHF, HC2H3O2 (acetic acid), H2CO3 (carbonic acid), H3PO4 (phosphoric acid), and many more
Is HC2H3O2 polar or nonpolar?
A 1 molar solution of acetic acid has a pH of about 2.4, meaning that only 0.4% of the molecules have donated a proton. The presence of the hydroxyl group at the carboxyl end also makes acetic acid slightly polar.
What does HC2H3O2 dissociate into?
Each of the components of the HC2H3O2 equilibrium system in Part A (the dissociation of acetic acid (HC2H3O2) in water to form a hydronium ion and acetate ion, as shown in the net ionic equation below HC2H3O2 + H2O <—> C2H3O2- + H3O+ (In order to make any observations during this reaction, methyl orange indicator …
Which is a stronger acid HCl or HC2H3O2?
HC2H3O2 is a much weaker acid than HCl so the HC2H3O2 solution would have a higher pH than the HCl solution. Therefore, the ranking from highest to lowest pH for solutions with the same solute concentrations is: NaOH > NH3 > HC2H3O2 > HCl.
Why is acetic acid a weak electrolyte?
It is a weak electrolyte because it’s ionization is very less.
What if acetic acid is a strong acid?
AcidWeak AcidsH3PO4 (phosphoric acid)H2PO−4 (dihydrogen phosphate ion)CH3COOH (acetic acid)CH3COO− (acetate ion)
Is h20 a strong electrolyte?
Weak electrolytes partially ionize in water. … Water is considered a weak electrolyte by some sources because it partly dissociates into H+ and OH– ions, but a nonelectrolyte by other sources because only a very small amount of water dissociates into ions.
Why is acetic acid soluble?
So, acetic acid will dissolve in water because it too has polar hydrogen bonding sites and can even be ionic (polar charges on different molecules), both of which will strongly attract water.
How does acetic acid dissociate in water?
Acetic acid dissociates in water because it is a proton donor with a charge stabilized anion. This spreads the resulting negative charge over a relatively large area, making it easier for acidic acid to become ionize. The electronic structure of the ionized form of acetic acid is different from what is normally shown.
How do you find the name for HC2H3O2?
HC2H3O2 is the molecular formula of acetic acid. It is weak acid. Vinegar is an aqueous solution of acetic acid, usually containing between 4% and 5% acetic acid.
Is chloric acid monoprotic?
Hydrochloric acid (HCl) and nitric acid (HNO3) are common monoprotic acids.
Is hydrofluoric acid a Diprotic acid?
H2CO3 and H2SO3 are called diprotic acids, and H3PO3 and H3PO4 are called triprotic acids. HF, HCl, HBr, and HC2H3O2 are examples of monoprotic acids. … H2SO4 is another example of a diprotic acid.
Why is HNO2 not a strong acid?
HNO2 (Nitrous acid) is a weak acid because it does not ionize 100% or only partially dissociates in an aqueous solution.
Can HNO2 act as a base?
Re: Distinguishing bronsted acids vs bases In the example you give, HNO2 + HPO4 ^2- <=> NO2^- + H2PO4^-, HNO2 is the acid, because it loses (donates) a proton to HPO4^2-. HPO4 ^2- is the base, because it accepts the H+ from HNO2. You can distinguish bronsted acids and bases by finding their roles in proton transfer.
Why is HNO2 a strong acid?
it is an acid as it releases hydronium ions as the only positively charged ion when dissolved in water. HNO2 a strong or weak acid. Because every acid reacts with water. When it loses a proton, it transfers that proton to water.
What substances are weak electrolytes?
- hydrofluoric acid, HF.
- hydrocyanic acid, HCN.
- acetic acid, HC2H3O2
- nitrous acid, HNO2
- sulfurous acid, H2SO3
- chlorous acid, HClO2
Is li2so4 a strong electrolyte?
The compounds LiOH, calcium chloride, lithium sulfate, and sodium acetate trihydrate are soluble or dissociate easily in water, and all are strong…
Is ch3coonh4 a strong or weak electrolyte?
CH3COONH4 is a strong electrolyte since in aqueous solution it dissociates completely. Sparingly soluble salts AgCl, CuSO4 are also strong electrolytes.
Is HNO2 a weak electrolyte?
Nitrous acid, HNO2(aq), is a weak electrolyte.
Is k2co3 a strong electrolyte?
Since it’s a soluble ionic salt, we can say that K2CO3 is a strong electrolyte.
Which of the is not weak electrolyte?
D is the only one listed here that is not a weak electrolyte. It is actually a strong electrolyte because it is a strong acid. It doesn’t like to stay in equilibrium. It likes to associate completely.